Εθνικό Κέντρο Έρευνας και Τεχνολογικής Ανάπτυξης / Ινστιτούτο Τεχνολογίας & Εφαρμογών Στερεών Καυσίμων
|
|
|
- Ευρυδίκη Κορωναίος
- 8 χρόνια πριν
- Προβολές:
Transcript
1 ΑΝΘΡΩΠΙΝΑ ΔΙΚΤΥΑ ΕΡΕΥΝΗΤΙΚΗΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΚΗΣ ΕΠΙΜΟΡΦΩΣΗΣ «ΤΕΧΝΟΛΟΓΙΚΟ ΔΥΝΑΜΙΚΟ ΓΙΑ ΤΗΝ ΜΕΙΩΣΗ ΤΩΝ ΕΚΠΟΜΠΩΝ ΔΙΟΞΕΙΔΙΟΥ ΤΟΥ ΑΝΘΡΑΚΑ ΔΥΝΑΤΟΤΗΤΕΣ ΠΡΟΟΠΤΙΚΕΣ ΤΩΝ ΕΛΛΗΝΙΚΩΝ ΕΠΙΧΕΙΡΗΣΕΩΝ» Δρ. Ν. Κούκουζας Πτολεμαΐδα, Δεκέμβριος 2007
2 Content Εθνικό Κέντρο Έρευνας και Τεχνολογικής Ανάπτυξης / Climate change effects Potential of CCS technologies CO 2 capture technologies and Storage Options Trapping mechanisms Basin-Scale Screening criteria for CO2 geological Storage Types of CO2 Geological Storage Projects CO 2 geological storage potential
3
4 The increase of atmosphere s natural warming capacity - Global warming - is caused due to the increase of human-induced GHGs.
5 Greenhouse gas effects General temperature increase Ice/glacier/pole cap melting Rise in sea level/coastal flooding Change of ocean ph/ocean life Effect on ocean currents and related large scale climate Regional cloud formation, impact on weather conditions, precipitation changes, floods Severe sectorial meteorological changes, intense storms, extreme droughts
6
7 Contribution of different gases to the anthropogenic greenhouse effect
8 The greatest increase of CO2 emissions originates from power generation and transport In Europe Energy sector is responsible for 80% of the EU-greenhouse gas (GHG) emission 93% of the EU-CO2 emission million tonnes of CO Power Generation Other Transformation Industry Transport Other Sectors Source: IEA WEO 2004
9 By 2030 energy use forecast to grow by 60% World Primary Energy Demand Fossil fuels projected to remain dominant energy source accounting for almost 90% of the growth in energy demand Source: IEA WEO 2004
10 Global CO 2 emissions grow by about 60% by Mt of CO OECD Transition economies Developing countries Source: IEA WEO 2004
11 CCS will plays a significant role in GHG mitigation 150 In the context of the long term objective of 60 per cent reduction by 2050 CCS is a medium term ( ) technology. Worldwide CO2 emissions (billion ton CO2/year) Energy efficiency Renewable energy CO2 capture and storage Fossil fuels Year Source: GESTCO project, Hendriks, Ecofys
12 Advantages of CCS technologies as a climate change mitigation option Continued use of available fossil fuel resources Sufficient storage capacity Capture technologies involve the highest costs associated with CCS BUT over the next decade the cost of capture could be reduced by %. Health, safety and environment risks for geological storage are low, and the level of risk will decline over time. Potential bridging technology from a current fossil-fuel energy-based world to a renewable energy-based one in the future.
13 Capture Transport Geological Storage
14 Overview of CO2 Capture processes
15
16 Reservoirs and Seals In general a reservoir / seal pair consist of: Porous and permeable reservoir rock that can contain (a mixture of) gas and liquid - Rocks with porosity of typically 5-30% of volume of the rock Overlain by a seal ( non permeable rock) layer Typical seal permeability is < md Typical Reservoir size is km^3
17 Porosity is the storage space in the rock for fluids and is shown by the blue spaces in this photomicrograph of a thin slice through a reservoir sandstone. Permeability is a measure of the ability of the rock to allow fluid flow. Permeability is strongly affected by the geometry of the porosity inparticular thesizeofthespacesconnecting theporesintherock
18 Suitability of basins for CO2 geological storage Sedimentary basins are suitable for CO2 storage due to the right type of porous and permeable rocks for storage and injection, such as sandstones and carbonates, and the low permeability-toimpermeable rocks needed for sealing, such as shales and evaporitic beds. Igneous and metamorphic rocks generally are not suitable for CO2 storage because they do not possess the necessary porosity and permeability that would allow injection and storage.
19 Sandstone Roughly 20% of sedimentary rocks Consists mainly of quartz, sometimes feldspar and other minerals High permeability Medium porosity Often contain liquids or gases (water, oil or gas)
20 Shale (clay rich rocks) Most abundant sedimentary rock Originates from mechanical and chemical weathering of silicate minerals Transported by water and deposited in quit environments (lakes, floodplains) Very fine particles Very low permeability High porosity Often forms a sealing barrier to liquid movement
21 Limestone Roughly 20% of sedimentary rocks Originates mainly from the precipitation and accumulation of material of biochemical origin Consists mainly of the mineral calcite Transported and deposited in water Low to medium permeability High porosity
22 Rock salt Originates from the precipitation of minerals by the evaporation of seawater Consist mainly of the mineral halite very low permeability and porosity Flows plastically (with very little fracturing) when under pressure Tends to move upwards due to low density and form salt domes (m/year) Capable to serve as a lubricant or as an impermeable seal
23 Trapping mechanisms Hydrogeological trapping - traps the carbon dioxide into flow systems for geological periods of time Phase trapping;- strips out the CO2 as an immobile liquid or gas phase Geochemical trapping - converts the carbon dioxide to aqueous species and carbonate minerals rendering it immobile.
24 Hydrodynamic (hydrogeological) trapping in saline aquifers. Carbon dioxide can be injected into deep aquifers by displacing the saline formation water CO2 is expected to migrate under the force of buoyancy towards the surface If the aquifer is well-bounded by aquitards, migration of the CO2 would be slowly The time needed for a volume of fluid to reach the surface from the deep basin is measured on a geological time scale (more than 10 5 years) Migration paths in hydrodynamic traps in deep saline aquifers: a) trapping in regional-scale systems, b) in interconnected systems through faults and fractures
25 Geologic (hydrogeological) trapping in reservoirs structural traps (anticlines, unconformities or faults) stratigraphic traps (change in type of rock layer) Folding and anticlines Folding and anticlines Faults and unconformities
26 Phase Trapping In the case of injection of CO2, the water will reach irreducible saturation as the CO2 bubble displaces it. The CO2 will reach irreducible saturation after injection ceases and the natural flow of the aquifer displaces the CO2. Irreducible saturation for gases can range from 2 to 25%. Consequently, a CO2 bubble formed around the base of an injection well will be dispersed by any natural water flow through the porous media until all of the CO2 is trapped by stripping, through retention in the form of the irreducible saturation.
27 Geochemical trapping Adsorption the preferential adsorption of gaseous CO2 onto the coal matrix because of its higher affinity to coal than that of methane; Solubility up to 30% of injected CO2 will dissolve in the formation water for an engineering time scale; Ionic Silicate or carbonate minerals present in the aquifer neutralize the acid added to the formation water by the addition of CO2. These reactions fix the CO2 as an ionic species in the formation water. Mineral - Silicate minerals present in the aquifer neutralize the acid added to the formation water by the addition of CO2. These reactions can fix the CO2 as a mineral and result in permanent trapping.
28 Contribution of trapping mechanisms to the security of CO2 geological storage
29 Basin-Scale Screening Criteria Based on: Geological characteristics Hydrodynamic and geothermal regimes Basin resources and maturity Industry maturity and infrastructure Economic and societal aspects
30 More suitable sedimentary basins
31 Basin characteristics important for storage Adequate thickness >1000 m; Strong reservoir and seal pair; Not highly faulted, fractured or located in fold belts; Strongly harmonious sequences; No volcanogenic sediments; No significant diagenesis.
32 Geological characteristics important for CO2 storage sites Adequate porosity, thickness and permeability - The presence of a storage formation of adequate porosity and thickness (for capacity) and permeability (for injectivity) is critical. Depth greater than 800 m for CO2 in supercritical state Confining unit cap - shale, salt or anhydrite beds, to ensure CO2 storage and prevent its escape into overlying, shallower units and ultimately to the surface.
33 Cold Basins: Low surface temperature and/or geothermal gradients more favorable (higher CO2 density, at shallower depths) Warms basins: High surface temperature and geothermal gradients less favorable (lower CO2 density, larger depths needed Geothermal regimes
34 Basin Maturity Fossil-energy potential (oil and gas, coals) and degree of exploration and production Mature: Rich in Energy resources, advanced production Poor: No or poor in hydrocarbon resources
35 Industry Maturity and Infrastructure Developed continental basins: Access roads, pipelines, wells Developed marine basins: Drilling and production platforms
36 Economic and Societal Aspects Cost: Affected by basin location, marine or continental, climatic conditions, transportation distances, injection depth Legal: Jurisdiction over resources, liability, regulatory regime Public Acceptance Proximity to population centers, accept or reject projects
37 Prospective geological storage areas in sedimentary basins
38 How much storage potential is available? Study Location World - Koide 92 World - van der M eer 92 World - IEA 92 Wo rld - H endriks and B lo k 93 Wo rld - H endriks and B lo k 94 World - IEA 94 Wo rld - H endriks 94 World - Hendriks & Blok 95 World - Turkenburg 97 Wo rld - IP C C 01/A rc 00 World - ECOFYS & TNO-NITG 2002 Wo rld - B ruant 02 World 1 - GEOSEQ World 2 - Beecy & Kuuskra 01 World 3 - IEA Wo rld - D o o ley and F riedman World - ECOFYS Euro pe - van der Straaten Europe - Boe et al N W Euro pe - Jo ule R epo rt Western Europe - Dooley amd Friedman Eastern Europe - Dooley and Friedman Former Soviet Union - Dooley and Combined Europe - Dooley and Friedman Western Europe - ECOFYS Eastern Europe - ECOFYS Total Europe - ECOFYS USA - Bergman & Winter )M t Simo n Sandsto ne (Ohio )M t Simo n Sandsto ne (M idwest USA M t Simo n Sandsto ne USA - Dooley and Friedman USA - ECOFYS Alberta Basin (Canada) - Total Alberta Basin (Canada) - Viking Fmn C anada - D o o ley and F riedman C anada - EC OF YS Australia - Bradshaw et al 2002 Australia/NZ - Dooley and Friedman Oceania - ECOFYS Japan - ECOFYS Japan - Dooley and Friedman Japan From Bradshaw et al., 2005: Discussion Paper on CO 2 Storage Capacity Estimation CSLF Task Force on Capacity ,000 GT CO 2 10,000 World WORLD: : , GT Europe : GT EUROPE: GT USA : GT USA: GT Canada : GT CANADA: GT Australia : GT AUSTRALIA: GT Japan : 0 80 GT JAPAN: 0-80 GT 100,000 1,000,000
39 Storage potential The potential storage capacity covers at least several decades of current global CO2 emissions (approx. 30 Gt CO2/year). The estimated range of the economic potential for CCS varies between Gt CO2, which would mean that 15-55% of the world-wide mitigation effort by 2100 could be achieved through the implementation of CCS. Gton CO Gton CO 2 2 Depleted oil fields Depleted gas fields Enhanced oil recovery Unminable coal seams >15 >73 Saline aquifers* , ,000 1 Source: (IEA GHG, 2001) 2 Source: (Edmonds, 2000)
40 European geological storage capacity 200 to up to 1550 Gt CO to 1500 Gt of CO2 in deep saline formations mainly in the North Sea. The total capacity of hydrocarbon fields is estimated at more than 40 Gt CO2, 7 Gt of which can be stored in oil reservoirs. The storage capacity of unmineable coal seams is estimated at 6 Gt CO2.
41 Enhanced oil recovery (EOR) CO2-EOR is applied on commercial scale in the USA, Canada, Turkey and Trinidad and Tobago. The majority of projects are situated in the Permian basin in the USA. Currently about 43 million tonnes/year of CO2 is being injected; the majority (~ 75%) being CO2 from geological reservoirs and the remaining part from industrial sources. CO2 injection into water flooded oil reservoirs could yield an extra of 4-12% OOIP oil production 40% of oil reserves can be recovered through primary (~25 to 30%) and secondary (~10%) processes. The use of CO2 can enhance the recovery of around 10 to 12% of the remaining oil.
42 By injecting CO2 into oil reservoirs oil is mobilized through miscible or immiscible displacement increasing oil recovery. Enhanced oil recovery (EOR) The average retention of the injected CO2 in several reservoirs is around 60% while m3 of CO2 is required to produce 1barrel of oil.
43 Enhanced oil recovery (EOR) When introduced in the reservoir CO2 interacts chemically and physically with the reservoir rock and the contained oil, creating favourable conditions that improve oil recovery such as: (i) the reduction of the capillary forces that inhibit oil flow through the pores of the reservoir by reducing the interfacial tension between oil and the reservoir rock; (ii) the expansion of the volume of the oil (oil swelling) and the subsequent reduction of its viscosity; (iii) the development of favourable complex phase changes in the oil that increase its fluidity; (iv) the maintenance of favourable mobility characteristics for oil and CO2 to improve the volume sweep (replacement) efficiency.
44 Two processes for CO2-EOR: Enhanced oil recovery (EOR) Miscible displacement where the injected gas and the hydrocarbons are completely miscible and form a single-phase fluid. Immiscible displacement occurs at pressures below a minimum miscible pressure (MMP) of the oil, in which there is less interchange of components or mixing zones between the gas injected and the reservoir fluid.
45 Miscible CO2 displacement method A schematic of a WAG miscible CO2-EOR operation
46 Immiscible CO2 displacement method CO2 is typically injected in GSGI mode at slow rates at the crest of the reservoir aiming at filling the pore volume of the reservoir rock. The injected gas creates an artificial gas cap, pushing oil simultaneously downwards and towards the rim of the reservoir where the producing wells are located. The presence of water within the reservoir reduces the effectiveness of the process as it inhibits oil flow. This process may not be effective when applied after significant water flooding.
47 Immiscible displacement technique
48 Criteria Specific to Enhanced Oil Recovery Light oil (25 to 48 API) Reservoir pressure greater than Minimum Miscibility Pressure Preferably thin net pay (<20 m) Homogeneous reservoir
49 Enhanced Gas Recovery (EGR) Using CO2 for enhanced gas recovery (EGR) is a speculative method for repressurizing depleted gas fields that can be applied to certain fields when 80-90% of the gas has been produced. CO2 EGR has not yet been applied anywhere in the world.
50 Enhanced Gas Recovery (EGR) The injected CO2 will flow in the reservoir due to pressure and gravitational effects. CO2 is denser than CH4 at all relevant pressures and temperatures and will tend to flow downwards, displacing CH4 gas and repressurizing the reservoir Given an initial pressure of 120 bar, another 5-15% of the initial gas in place could be recovered using EGR. About 1.8 GJ of gas could be recovered per tonne of CO2 stored, if a whole reservoir was filled with CO2 up to its original pressure. The potential for CO2 use for EGR might be larger than for EOR but the EGR revenues per tonne of CO2 are lower than for EOR.
51 Enhanced Coal Bed Methane Projects (ECBM) Conventional coalbed methane recovery may achieve 40-50% recovery while the recovery increases to % in the case of ECBM. ( ECBM is limited to coal seams that will not be mined The technology is only in the demonstration phase
52 Enhanced Coal Bed Methane Projects (ECBM) Coal contains micro-pores (r = nm) suitable for adsorption of gases, such as CO 2 (r = ca. 0.3 nm) Higher affinity to adsorb CO 2 than CH 4 One methane molecule can be replaced by at least two molecules of o CO 2 Ratio CO 2 /CH 4 depends on the maturity and type of coal Coal plastization and swelling can occur due to the presence of CO 2 and this reduces permeability
53 Enhanced Coal Bed Methane Projects (ECBM) Adsorption-desorption mechanism
54 Enhanced Coal Bed Methane Projects (ECBM) Adsorption of various gases on coal
55 Enhanced Coal Bed Methane Projects (ECBM) Factors affecting coal adsorption Coal rank Peat lignite bituminous coal anthracite Pore structure and size Moist content (rank dependent) Coal composition Presence of different macerals and minerals Moisture content Water molecules block adsorption sites of pore system ph change Temperature adsorption rates decrease with increasing T
56 Enhanced Coal Bed Methane Projects (ECBM) No clear whether supercritical CO2 is adsorbed by coal, occupies the pore space like a fluid with very low viscosity, or infuses into the coal matrix. Thus, CO2 is injected in gaseous phase. For hydrostatic conditions and average geothermal gradients this would correspond to depths in the m range.
57 Enhanced Coal Bed Methane Projects (ECBM) CO2 adsorption related to pressure for wet and dry coal CO2 adsorbed [mmole/g coal daf] Equilibrium Pressure [MPa]
58 Criteria Specific to Enhanced Coalbed Methane Recovery A homogeneous reservoir, laterally continuous and vertically isolated from surrounding strata; Minimally faulted and folded; At least 1-5 millidarcies (md) permeability. High methane content; Stratigraphically concentrated coal seams are preferred over multiple thin seams; A possibility to use or export methane (pipeline) and CO2 availability (local power plant, industry or pipeline).
59 CO2 storage in deep saline aquifers Saline Aquifers are defined as porous and permeable reservoir rocks that contain saline fluid in the pore spaces between the rock grains. They generally occur at depths greater than aquifers that contain potable water. Water in aquifers deep below the ground in sedimentary basins is confined by overlying and underlying aquitards and/or aquicludes, usually has a high content of dissolved solids (brackish water and brine) and may have been there for millions of years. Because of the confined character of these aquifers, they have been proposed as locations for CO2 storage.
60 CO2 storage in deep saline aquifers Storage in confined aquifers relies on trapping of the buoyant CO2 by structural (e.g. anticlines) and /or stratigraphic features, and is closely analogous to gas storage schemes in hydrocarbon fields, or indeed to natural gas storage in subsurface aquifers. In simple structural traps, volumes and migration pathways of the injected CO2 can be predicted and reservoir models constructed with a higher degree of certainty than in an unconfined aquifer.
61 CO2 storage in deep saline aquifers CO2 is injected into large aquifers without specific large structural or stratigraphic closures. The injected CO2 migrates upwards along the most permeable pathway until it reachs the impermeable cap rock. Next CO2 migrates laterally along the cap rock-reservoir reservoir boundary following the most permeable pathways where small domes and undulations trap effectively a proportion of the injected CO2, depending on the average roughness.
62 CO2 storage in deep saline aquifers As these minor structural closures are filled, the CO2 spills out and continues to migrate laterally. In unconfined aquifers over time the CO2 will likely be distributed over a large area and in low concentrations increasing thet volumes that can be stored in these small perturbations on the top reservoir surface. Because of the likely large migration path lengths, the component of storage due to dissolution is increased significantly and thus, probably in many cases with the absence of vertical migration pathways the CO2 is unlikely to reach the surface. This type of storage has been demonstrated at Sleipner
63 . Εθνικό Κέντρο Έρευνας και Τεχνολογικής Ανάπτυξης / Depleted oil and gas fields Established seal and trap.. These structures have effectively stored hydrocarbons for millions of years, proving the integrity of the reservoir seal and the permanence of the fluid trap. Existing Infrastructure.. Some of the equipment installed on the surface or underground for oil or gas recovery may be re-used for injecting and storing CO2 lowering the initial capital requirements for establishing the t CO2 storage facility. Value-Added Products: : The revenues derived from the additional oil production could offset some (or all) of the costs associated with CO2 storage. Legal barriers are less strict for the injection of CO2 into hydrocarbon reservoirs compared to aquifers. Injection of CO2 for EOR is not prohibited under the London and OSPAR treaties.
64 Advantages and disadvantages IEA, GHG, 2004
65 Maximum potential CO2 storage capacity in Prinos offshore depleted oil field compared to the annual emissions from major point sources. Oil field (10 6 t CO2) Total storage capacity (10 6 t CO2) Annual point source CO2 emissions (10 6 t CO2) Number of years storage capacity from current point sources (approx.) (GESTCO PROJECT)
66 CO2 storage capacity in saline aquifers -Greece Aquifer Position Storage capacity (Mt CO2) Prinos W. Thessaloniki W. Thessaloniki sandstone offshore 1343 onshore 459 onshore 145 Alexandria Mesohellenic basin onshore 34 onshore 360 Total 2345 (GESTCO PROJECT)
67 Conclusions With appropriate site selection informed by available subsurface information, a monitoring program to detect problems, a regulatory system, and the appropriate use of remediation methods to stop or control CO2releases if they arise, the local health, safety, and environment risks of geological storage would be comparable to risks of current activities such as natural gas storage, EOR, anddeepundergrounddisposalofacidgas. IPCC, 2005
Εθνικό Κέντρο Έρευνας και Τεχνολογικής Ανάπτυξης / Ινστιτούτο Τεχνολογίας & Εφαρµογών Στερεών Καυσίµων
 ΠΡΟΓΡΑΜΜΑ «ΑΝΘΡΩΠΙΝΑ ΙΚΤΥΑ ΕΡΕΥΝΗΤΙΚΗΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΚΗΣ ΕΠΙΜΟΡΦΩΣΗΣ-Β ΚΥΚΛΟΣ» ΕΡΓΟ «ΤΕΧΝΟΛΟΓΙΚΟ ΥΝΑΜΙΚΟ ΓΙΑ ΤΗΝ ΜΕΙΩΣΗ ΤΩΝ ΕΚΠΟΜΠΩΝ ΙΟΞΕΙ ΙΟΥ ΤΟΥ ΑΝΘΡΑΚΑ ΥΝΑΤΟΤΗΤΕΣ ΠΡΟΟΠΤΙΚΕΣ ΤΩΝ ΕΛΛΗΝΙΚΩΝ ΕΠΙΧΕΙΡΗΣΕΩΝ»
ΠΡΟΓΡΑΜΜΑ «ΑΝΘΡΩΠΙΝΑ ΙΚΤΥΑ ΕΡΕΥΝΗΤΙΚΗΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΚΗΣ ΕΠΙΜΟΡΦΩΣΗΣ-Β ΚΥΚΛΟΣ» ΕΡΓΟ «ΤΕΧΝΟΛΟΓΙΚΟ ΥΝΑΜΙΚΟ ΓΙΑ ΤΗΝ ΜΕΙΩΣΗ ΤΩΝ ΕΚΠΟΜΠΩΝ ΙΟΞΕΙ ΙΟΥ ΤΟΥ ΑΝΘΡΑΚΑ ΥΝΑΤΟΤΗΤΕΣ ΠΡΟΟΠΤΙΚΕΣ ΤΩΝ ΕΛΛΗΝΙΚΩΝ ΕΠΙΧΕΙΡΗΣΕΩΝ»
ΤΕΙ ΚΑΒΑΛΑΣ ΣΧΟΛΗ ΤΕΧΝΟΛΟΓΙΚΩΝ ΕΦΑΡΜΟΓΩΝ ΤΜΗΜΑ ΗΛΕΚΤΡΟΛΟΓΙΑΣ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ
 ΤΕΙ ΚΑΒΑΛΑΣ ΣΧΟΛΗ ΤΕΧΝΟΛΟΓΙΚΩΝ ΕΦΑΡΜΟΓΩΝ ΤΜΗΜΑ ΗΛΕΚΤΡΟΛΟΓΙΑΣ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΜΕΛΕΤΗ ΦΩΤΟΒΟΛΤΑΙΚΟΥ ΠΑΡΚΟΥ ΜΕ ΟΙΚΙΣΚΟΥΣ ΓΙΑ ΠΑΡΑΓΩΓΗ ΗΛΕΚΤΡΙΚΗΣ ΕΝΕΡΓΕΙΑΣ ΜΕΣΗΣ ΤΑΣΗΣ STUDY PHOTOVOLTAIC PARK WITH SUBSTATIONS
ΤΕΙ ΚΑΒΑΛΑΣ ΣΧΟΛΗ ΤΕΧΝΟΛΟΓΙΚΩΝ ΕΦΑΡΜΟΓΩΝ ΤΜΗΜΑ ΗΛΕΚΤΡΟΛΟΓΙΑΣ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΜΕΛΕΤΗ ΦΩΤΟΒΟΛΤΑΙΚΟΥ ΠΑΡΚΟΥ ΜΕ ΟΙΚΙΣΚΟΥΣ ΓΙΑ ΠΑΡΑΓΩΓΗ ΗΛΕΚΤΡΙΚΗΣ ΕΝΕΡΓΕΙΑΣ ΜΕΣΗΣ ΤΑΣΗΣ STUDY PHOTOVOLTAIC PARK WITH SUBSTATIONS
EU energy policy Strategies for renewable energy sources in Cyprus
 EU energy policy Strategies for renewable energy sources in Cyprus Dr. Andreas Poullikkas Electricity Authority of Cyprus 0 Contents Future energy systems Strategies for RES Towards 2020 Post 2020 - Towards
EU energy policy Strategies for renewable energy sources in Cyprus Dr. Andreas Poullikkas Electricity Authority of Cyprus 0 Contents Future energy systems Strategies for RES Towards 2020 Post 2020 - Towards
Assalamu `alaikum wr. wb.
 LUMP SUM Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. LUMP SUM Lump sum lump sum lump sum. lump sum fixed price lump sum lump
LUMP SUM Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. LUMP SUM Lump sum lump sum lump sum. lump sum fixed price lump sum lump
ΠΟΛΤΣΔΥΝΔΗΟ ΚΡΖΣΖ ΣΜΖΜΑ ΜΖΥΑΝΗΚΧΝ ΟΡΤΚΣΧΝ ΠΟΡΧΝ
 ΠΟΛΤΣΔΥΝΔΗΟ ΚΡΖΣΖ ΣΜΖΜΑ ΜΖΥΑΝΗΚΧΝ ΟΡΤΚΣΧΝ ΠΟΡΧΝ ΓΗΠΛΧΜΑΣΗΚΖ ΔΡΓΑΗΑ ΔΦΑΡΜΟΓΔ ΓΔΧΘΔΡΜΗΑ ΥΑΜΖΛΖ ΔΝΘΑΛΠΗΑ ΣΖΝ ΠΔΡΗΟΥΖ ΑΛΔΞΑΝΓΡΔΗΑ Ν. ΖΜΑΘΗΑ ΓΗΑΜΑΝΣΟΠΟΤΛΟ Η. ΦΧΣΗΟ ΔΞΔΣΑΣΗΚΖ ΔΠΗΣΡΟΠΖ: ΘΔΟΓΧΡΟ ΜΑΡΚΟΠΟΤΛΟ Καζεγεηήο
ΠΟΛΤΣΔΥΝΔΗΟ ΚΡΖΣΖ ΣΜΖΜΑ ΜΖΥΑΝΗΚΧΝ ΟΡΤΚΣΧΝ ΠΟΡΧΝ ΓΗΠΛΧΜΑΣΗΚΖ ΔΡΓΑΗΑ ΔΦΑΡΜΟΓΔ ΓΔΧΘΔΡΜΗΑ ΥΑΜΖΛΖ ΔΝΘΑΛΠΗΑ ΣΖΝ ΠΔΡΗΟΥΖ ΑΛΔΞΑΝΓΡΔΗΑ Ν. ΖΜΑΘΗΑ ΓΗΑΜΑΝΣΟΠΟΤΛΟ Η. ΦΧΣΗΟ ΔΞΔΣΑΣΗΚΖ ΔΠΗΣΡΟΠΖ: ΘΔΟΓΧΡΟ ΜΑΡΚΟΠΟΤΛΟ Καζεγεηήο
1, +,*+* + +-,, -*, * : Key words: global warming, snowfall, snowmelt, snow water equivalent. Ohmura,,**0,**
 1, +,*+* + +-,, + : /+* m,1+ m, -*, * +3132* : Key words: global warming, snowfall, snowmelt, snow water equivalent + IPCC,,**+ Inoue and,**2 Yokoyama,**- Ohmura,,**0,**0 +331 +332 + +2- **+, ++,* 14 1,
1, +,*+* + +-,, + : /+* m,1+ m, -*, * +3132* : Key words: global warming, snowfall, snowmelt, snow water equivalent + IPCC,,**+ Inoue and,**2 Yokoyama,**- Ohmura,,**0,**0 +331 +332 + +2- **+, ++,* 14 1,
ΑΚΑ ΗΜΙΑ ΕΜΠΟΡΙΚΟΥ ΝΑΥΤΙΚΟΥ ΜΑΚΕ ΟΝΙΑΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΘΕΜΑ : ΧΗΜΙΚΑ ΠΡΟΣΘΕΤΑ ΠΟΥ ΠΡΟΟΡΙΖΟΝΤΑΙ ΓΙΑ ΤΟ ΝΕΡΟ ΤΟΥ ΑΤΜΟΛΕΒΗΤΑ
 ΑΚΑ ΗΜΙΑ ΕΜΠΟΡΙΚΟΥ ΝΑΥΤΙΚΟΥ ΜΑΚΕ ΟΝΙΑΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΘΕΜΑ : ΧΗΜΙΚΑ ΠΡΟΣΘΕΤΑ ΠΟΥ ΠΡΟΟΡΙΖΟΝΤΑΙ ΓΙΑ ΤΟ ΝΕΡΟ ΤΟΥ ΑΤΜΟΛΕΒΗΤΑ ΣΠΟΥ ΑΣΤΗΣ : ΑΓΟΡΑΣΤΟΣ ΧΡΥΣΟΒΑΛΑΝΤΗΣ ΕΠΙΒΛΕΠΟΥΣΑ ΚΑΘΗΓΗΤΡΙΑ :
ΑΚΑ ΗΜΙΑ ΕΜΠΟΡΙΚΟΥ ΝΑΥΤΙΚΟΥ ΜΑΚΕ ΟΝΙΑΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΘΕΜΑ : ΧΗΜΙΚΑ ΠΡΟΣΘΕΤΑ ΠΟΥ ΠΡΟΟΡΙΖΟΝΤΑΙ ΓΙΑ ΤΟ ΝΕΡΟ ΤΟΥ ΑΤΜΟΛΕΒΗΤΑ ΣΠΟΥ ΑΣΤΗΣ : ΑΓΟΡΑΣΤΟΣ ΧΡΥΣΟΒΑΛΑΝΤΗΣ ΕΠΙΒΛΕΠΟΥΣΑ ΚΑΘΗΓΗΤΡΙΑ :
ΗΜΟΣΙΑ ΕΠΙΧΕΙΡΗΣΗ ΗΛΕΚΤΡΙΣΜΟΥ Α.Ε. Νίκος Περδικάρης. ιεύθυνση Περιβάλλοντος Παραγωγής
 ΗΜΟΣΙΑ ΕΠΙΧΕΙΡΗΣΗ ΗΛΕΚΤΡΙΣΜΟΥ Α.Ε. SIXTH FRAMEWORK PROGRAMME PRIORITY [6.1] SUSTAINABLE ENERGY SYSTEMS Νίκος Περδικάρης Χηµικός Μηχ/κός ΕΜΠ, MSc. ιεύθυνση Περιβάλλοντος Παραγωγής Σεµινάριο ΕΗ: «Τεχνολογικό
ΗΜΟΣΙΑ ΕΠΙΧΕΙΡΗΣΗ ΗΛΕΚΤΡΙΣΜΟΥ Α.Ε. SIXTH FRAMEWORK PROGRAMME PRIORITY [6.1] SUSTAINABLE ENERGY SYSTEMS Νίκος Περδικάρης Χηµικός Μηχ/κός ΕΜΠ, MSc. ιεύθυνση Περιβάλλοντος Παραγωγής Σεµινάριο ΕΗ: «Τεχνολογικό
O ρόλος των τεχνολογιών CCS ως τεχνολογική επιλογή αντιµετώπισης της κλιµατικής αλλαγής
 ΠΡΟΓΡΑΜΜΑ «ΑΝΘΡΩΠΙΝΑ ΙΚΤΥΑ ΕΡΕΥΝΗΤΙΚΗΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΚΗΣ ΕΠΙΜΟΡΦΩΣΗΣ-Β ΚΥΚΛΟΣ» ΕΡΓΟ «ΤΕΧΝΟΛΟΓΙΚΟ ΥΝΑΜΙΚΟ ΓΙΑ ΤΗΝ ΜΕΙΩΣΗ ΤΩΝ ΕΚΠΟΜΠΩΝ ΙΟΞΕΙ ΙΟΥ ΤΟΥ ΑΝΘΡΑΚΑ ΥΝΑΤΟΤΗΤΕΣ ΠΡΟΟΠΤΙΚΕΣ ΤΩΝ ΕΛΛΗΝΙΚΩΝ ΕΠΙΧΕΙΡΗΣΕΩΝ»
ΠΡΟΓΡΑΜΜΑ «ΑΝΘΡΩΠΙΝΑ ΙΚΤΥΑ ΕΡΕΥΝΗΤΙΚΗΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΚΗΣ ΕΠΙΜΟΡΦΩΣΗΣ-Β ΚΥΚΛΟΣ» ΕΡΓΟ «ΤΕΧΝΟΛΟΓΙΚΟ ΥΝΑΜΙΚΟ ΓΙΑ ΤΗΝ ΜΕΙΩΣΗ ΤΩΝ ΕΚΠΟΜΠΩΝ ΙΟΞΕΙ ΙΟΥ ΤΟΥ ΑΝΘΡΑΚΑ ΥΝΑΤΟΤΗΤΕΣ ΠΡΟΟΠΤΙΚΕΣ ΤΩΝ ΕΛΛΗΝΙΚΩΝ ΕΠΙΧΕΙΡΗΣΕΩΝ»
(1) Describe the process by which mercury atoms become excited in a fluorescent tube (3)
 Q1. (a) A fluorescent tube is filled with mercury vapour at low pressure. In order to emit electromagnetic radiation the mercury atoms must first be excited. (i) What is meant by an excited atom? (1) (ii)
Q1. (a) A fluorescent tube is filled with mercury vapour at low pressure. In order to emit electromagnetic radiation the mercury atoms must first be excited. (i) What is meant by an excited atom? (1) (ii)
Mean bond enthalpy Standard enthalpy of formation Bond N H N N N N H O O O
 Q1. (a) Explain the meaning of the terms mean bond enthalpy and standard enthalpy of formation. Mean bond enthalpy... Standard enthalpy of formation... (5) (b) Some mean bond enthalpies are given below.
Q1. (a) Explain the meaning of the terms mean bond enthalpy and standard enthalpy of formation. Mean bond enthalpy... Standard enthalpy of formation... (5) (b) Some mean bond enthalpies are given below.
ΠΟΛΥΤΕΧΝΕΙΟ ΚΡΗΤΗΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΕΡΙΒΑΛΛΟΝΤΟΣ
 ΠΟΛΥΤΕΧΝΕΙΟ ΚΡΗΤΗΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΕΡΙΒΑΛΛΟΝΤΟΣ Τομέας Περιβαλλοντικής Υδραυλικής και Γεωπεριβαλλοντικής Μηχανικής (III) Εργαστήριο Γεωπεριβαλλοντικής Μηχανικής TECHNICAL UNIVERSITY OF CRETE SCHOOL of
ΠΟΛΥΤΕΧΝΕΙΟ ΚΡΗΤΗΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΕΡΙΒΑΛΛΟΝΤΟΣ Τομέας Περιβαλλοντικής Υδραυλικής και Γεωπεριβαλλοντικής Μηχανικής (III) Εργαστήριο Γεωπεριβαλλοντικής Μηχανικής TECHNICAL UNIVERSITY OF CRETE SCHOOL of
ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΣΧΟΛΗ ΠΟΛΙΤΙΚΩΝ ΜΗΧΑΝΙΚΩΝ. «Θεσμικό Πλαίσιο Φωτοβολταïκών Συστημάτων- Βέλτιστη Απόδοση Μέσω Τρόπων Στήριξης»
 ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΣΧΟΛΗ ΠΟΛΙΤΙΚΩΝ ΜΗΧΑΝΙΚΩΝ ΤΟΜΕΑΣ ΑΝΘΡΩΠΙΣΤΙΚΩΝ & ΚΟΙΝΩΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΚΑΙ ΔΙΚΑΙΟΥ «Θεσμικό Πλαίσιο Φωτοβολταïκών Συστημάτων- Βέλτιστη Απόδοση Μέσω Τρόπων Στήριξης» Διπλωματική
ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΣΧΟΛΗ ΠΟΛΙΤΙΚΩΝ ΜΗΧΑΝΙΚΩΝ ΤΟΜΕΑΣ ΑΝΘΡΩΠΙΣΤΙΚΩΝ & ΚΟΙΝΩΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΚΑΙ ΔΙΚΑΙΟΥ «Θεσμικό Πλαίσιο Φωτοβολταïκών Συστημάτων- Βέλτιστη Απόδοση Μέσω Τρόπων Στήριξης» Διπλωματική
the total number of electrons passing through the lamp.
 1. A 12 V 36 W lamp is lit to normal brightness using a 12 V car battery of negligible internal resistance. The lamp is switched on for one hour (3600 s). For the time of 1 hour, calculate (i) the energy
1. A 12 V 36 W lamp is lit to normal brightness using a 12 V car battery of negligible internal resistance. The lamp is switched on for one hour (3600 s). For the time of 1 hour, calculate (i) the energy
Potential Dividers. 46 minutes. 46 marks. Page 1 of 11
 Potential Dividers 46 minutes 46 marks Page 1 of 11 Q1. In the circuit shown in the figure below, the battery, of negligible internal resistance, has an emf of 30 V. The pd across the lamp is 6.0 V and
Potential Dividers 46 minutes 46 marks Page 1 of 11 Q1. In the circuit shown in the figure below, the battery, of negligible internal resistance, has an emf of 30 V. The pd across the lamp is 6.0 V and
«ΑΓΡΟΤΟΥΡΙΣΜΟΣ ΚΑΙ ΤΟΠΙΚΗ ΑΝΑΠΤΥΞΗ: Ο ΡΟΛΟΣ ΤΩΝ ΝΕΩΝ ΤΕΧΝΟΛΟΓΙΩΝ ΣΤΗΝ ΠΡΟΩΘΗΣΗ ΤΩΝ ΓΥΝΑΙΚΕΙΩΝ ΣΥΝΕΤΑΙΡΙΣΜΩΝ»
 I ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΣΧΟΛΗ ΝΟΜΙΚΩΝ ΟΙΚΟΝΟΜΙΚΩΝ ΚΑΙ ΠΟΛΙΤΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΤΜΗΜΑ ΟΙΚΟΝΟΜΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΗΝ «ΔΙΟΙΚΗΣΗ ΚΑΙ ΟΙΚΟΝΟΜΙΑ» ΚΑΤΕΥΘΥΝΣΗ: ΟΙΚΟΝΟΜΙΚΗ
I ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΣΧΟΛΗ ΝΟΜΙΚΩΝ ΟΙΚΟΝΟΜΙΚΩΝ ΚΑΙ ΠΟΛΙΤΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΤΜΗΜΑ ΟΙΚΟΝΟΜΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΗΝ «ΔΙΟΙΚΗΣΗ ΚΑΙ ΟΙΚΟΝΟΜΙΑ» ΚΑΤΕΥΘΥΝΣΗ: ΟΙΚΟΝΟΜΙΚΗ
Strain gauge and rosettes
 Strain gauge and rosettes Introduction A strain gauge is a device which is used to measure strain (deformation) on an object subjected to forces. Strain can be measured using various types of devices classified
Strain gauge and rosettes Introduction A strain gauge is a device which is used to measure strain (deformation) on an object subjected to forces. Strain can be measured using various types of devices classified
Figure 3 Three observations (Vp, Vs and density isosurfaces) intersecting in the PLF space. Solutions exist at the two indicated points.
 φ φ φ φ Figure 1 Resampling of a rock-physics model from velocity-porosity to lithology-porosity space. C i are model results for various clay contents. φ ρ ρ δ Figure 2 Bulk modulus constraint cube in
φ φ φ φ Figure 1 Resampling of a rock-physics model from velocity-porosity to lithology-porosity space. C i are model results for various clay contents. φ ρ ρ δ Figure 2 Bulk modulus constraint cube in
Isotopic and Geochemical Study of Travertine and Hot Springs Occurring Along the Yumoto Fault at North Coast of the Oga Peninsula, Akita Prefecture
 J. Hot Spring Sci. /2 **2 * + + + + + +3 ++ * * + 3 Isotopic and Geochemical Study of Travertine and Hot Springs Occurring Along the Yumoto Fault at North Coast of the Oga Peninsula Akita Prefecture +
J. Hot Spring Sci. /2 **2 * + + + + + +3 ++ * * + 3 Isotopic and Geochemical Study of Travertine and Hot Springs Occurring Along the Yumoto Fault at North Coast of the Oga Peninsula Akita Prefecture +
ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙ ΕΥΤΙΚΟ Ι ΡΥΜΑ ΚΡΗΤΗΣ ΣΧΟΛΗ ΙΟΙΚΗΣΗΣ ΚΑΙ ΟΙΚΟΝΟΜΙΑΣ ΤΜΗΜΑ ΙΟΙΚΗΣΗΣ ΕΠΙΧΕΙΡΗΣΕΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ
 ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙ ΕΥΤΙΚΟ Ι ΡΥΜΑ ΚΡΗΤΗΣ ΣΧΟΛΗ ΙΟΙΚΗΣΗΣ ΚΑΙ ΟΙΚΟΝΟΜΙΑΣ ΤΜΗΜΑ ΙΟΙΚΗΣΗΣ ΕΠΙΧΕΙΡΗΣΕΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ Το franchising ( δικαιόχρηση ) ως µέθοδος ανάπτυξης των επιχειρήσεων λιανικού εµπορίου
ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙ ΕΥΤΙΚΟ Ι ΡΥΜΑ ΚΡΗΤΗΣ ΣΧΟΛΗ ΙΟΙΚΗΣΗΣ ΚΑΙ ΟΙΚΟΝΟΜΙΑΣ ΤΜΗΜΑ ΙΟΙΚΗΣΗΣ ΕΠΙΧΕΙΡΗΣΕΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ Το franchising ( δικαιόχρηση ) ως µέθοδος ανάπτυξης των επιχειρήσεων λιανικού εµπορίου
Μελέτη των μεταβολών των χρήσεων γης στο Ζαγόρι Ιωαννίνων 0
 Μελέτη των μεταβολών των χρήσεων γης στο Ζαγόρι Ιωαννίνων 0 ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΔΙΕΠΙΣΤΗΜΟΝΙΚΟ - ΔΙΑΤΜΗΜΑΤΙΚΟ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ (Δ.Π.Μ.Σ.) "ΠΕΡΙΒΑΛΛΟΝ ΚΑΙ ΑΝΑΠΤΥΞΗ" 2 η ΚΑΤΕΥΘΥΝΣΗ
Μελέτη των μεταβολών των χρήσεων γης στο Ζαγόρι Ιωαννίνων 0 ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΔΙΕΠΙΣΤΗΜΟΝΙΚΟ - ΔΙΑΤΜΗΜΑΤΙΚΟ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ (Δ.Π.Μ.Σ.) "ΠΕΡΙΒΑΛΛΟΝ ΚΑΙ ΑΝΑΠΤΥΞΗ" 2 η ΚΑΤΕΥΘΥΝΣΗ
Démographie spatiale/spatial Demography
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΙΑΣ Démographie spatiale/spatial Demography Session 1: Introduction to spatial demography Basic concepts Michail Agorastakis Department of Planning & Regional Development Άδειες Χρήσης
ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΙΑΣ Démographie spatiale/spatial Demography Session 1: Introduction to spatial demography Basic concepts Michail Agorastakis Department of Planning & Regional Development Άδειες Χρήσης
Phys460.nb Solution for the t-dependent Schrodinger s equation How did we find the solution? (not required)
 Phys460.nb 81 ψ n (t) is still the (same) eigenstate of H But for tdependent H. The answer is NO. 5.5.5. Solution for the tdependent Schrodinger s equation If we assume that at time t 0, the electron starts
Phys460.nb 81 ψ n (t) is still the (same) eigenstate of H But for tdependent H. The answer is NO. 5.5.5. Solution for the tdependent Schrodinger s equation If we assume that at time t 0, the electron starts
ΠΟΙΟΤΗΤΑ ΑΤΜΟΣΦΑΙΡΙΚΟΥ ΠΕΡΙΒΑΛΛΟΝΤΟΣ
 ΠΟΙΟΤΗΤΑ ΑΤΜΟΣΦΑΙΡΙΚΟΥ ΠΕΡΙΒΑΛΛΟΝΤΟΣ Κωνσταντινή Σαµαρά - Κωνσταντίνου Εργαστήριο Ελέγχου Ρύπανσης Περιβάλλοντος, Τµήµα Χηµείας, ΑΠΘ ΠΕΡΙΛΗΨΗ Παρότι υπήρξε σηµαντική βελτίωση της ποιότητας του αέρα στις
ΠΟΙΟΤΗΤΑ ΑΤΜΟΣΦΑΙΡΙΚΟΥ ΠΕΡΙΒΑΛΛΟΝΤΟΣ Κωνσταντινή Σαµαρά - Κωνσταντίνου Εργαστήριο Ελέγχου Ρύπανσης Περιβάλλοντος, Τµήµα Χηµείας, ΑΠΘ ΠΕΡΙΛΗΨΗ Παρότι υπήρξε σηµαντική βελτίωση της ποιότητας του αέρα στις
Σπουδάστρια Δακανάλη Νικολέτα Α.Μ. 5567. "Πώς η εξέλιξη της τεχνολογίας επηρεάζει την απόδοση των επιχειρήσεων" ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ
 ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙΔΕΥΤΙΚΟ ΙΔΡΥΜΑ ΑΝ. ΜΑΚΕΔΟΝΙΑΣ ΚΑΙ ΘΡΑΚΗΣ ΣΧΟΛΗ ΔΙΟΙΚΗΣΗΣ ΚΑΙ ΟΙΚΟΝΟΜΙΑΣ ΤΜΗΜΑ ΔΙΟΙΚΗΣΗΣ ΕΠΙΧΕΙΡΗΣΕΩΝ Σπουδάστρια Δακανάλη Νικολέτα Α.Μ. 5567 "Πώς η εξέλιξη της τεχνολογίας επηρεάζει την
ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙΔΕΥΤΙΚΟ ΙΔΡΥΜΑ ΑΝ. ΜΑΚΕΔΟΝΙΑΣ ΚΑΙ ΘΡΑΚΗΣ ΣΧΟΛΗ ΔΙΟΙΚΗΣΗΣ ΚΑΙ ΟΙΚΟΝΟΜΙΑΣ ΤΜΗΜΑ ΔΙΟΙΚΗΣΗΣ ΕΠΙΧΕΙΡΗΣΕΩΝ Σπουδάστρια Δακανάλη Νικολέτα Α.Μ. 5567 "Πώς η εξέλιξη της τεχνολογίας επηρεάζει την
A global biofuels outlook: U.S. RIN markets and EU proposals. Gerard Wynn Energy and climate columnist Thomson Reuters
 1η Ενότητα: «Το Παρόν και το Μέλλον των Βιοκαυσίμων στην Ελλάδα και την Ευρώπη» A global biofuels outlook: U.S. RIN markets and EU proposals Gerard Wynn Energy and climate columnist Thomson Reuters Global
1η Ενότητα: «Το Παρόν και το Μέλλον των Βιοκαυσίμων στην Ελλάδα και την Ευρώπη» A global biofuels outlook: U.S. RIN markets and EU proposals Gerard Wynn Energy and climate columnist Thomson Reuters Global
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΓΕΩΤΕΧΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΚΑΙ ΔΙΑΧΕΙΡΗΣΗΣ ΠΕΡΙΒΑΛΛΟΝΤΟΣ. Πτυχιακή εργασία ΑΝΑΠΤΥΞΗ ΔΕΙΚΤΩΝ ΠΟΙΟΤΗΤΑΣ ΕΔΑΦΟΥΣ
 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΓΕΩΤΕΧΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΚΑΙ ΔΙΑΧΕΙΡΗΣΗΣ ΠΕΡΙΒΑΛΛΟΝΤΟΣ Πτυχιακή εργασία ΑΝΑΠΤΥΞΗ ΔΕΙΚΤΩΝ ΠΟΙΟΤΗΤΑΣ ΕΔΑΦΟΥΣ [Μαρία Μαρκουλλή] Λεμεσός 2015 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΓΕΩΤΕΧΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΚΑΙ ΔΙΑΧΕΙΡΗΣΗΣ ΠΕΡΙΒΑΛΛΟΝΤΟΣ Πτυχιακή εργασία ΑΝΑΠΤΥΞΗ ΔΕΙΚΤΩΝ ΠΟΙΟΤΗΤΑΣ ΕΔΑΦΟΥΣ [Μαρία Μαρκουλλή] Λεμεσός 2015 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ
ΔΘΝΗΚΖ ΥΟΛΖ ΓΖΜΟΗΑ ΓΗΟΗΚΖΖ
 Ε ΔΘΝΗΚΖ ΥΟΛΖ ΓΖΜΟΗΑ ΓΗΟΗΚΖΖ Κ ΔΚΠΑΗΓΔΤΣΗΚΖ ΔΗΡΑ ΣΜΖΜΑ : Σνπξηζηηθήο Οηθνλνκίαο θαη Αλάπηπμεο (ΣΟΑ) ΣΔΛΗΚΖ ΔΡΓΑΗΑ Θέκα: Σνπξηζκφο θαη Οηθνλνκηθή Κξίζε Δπηβιέπσλ : Νηνχβαο Λνπθάο πνπδάζηξηα : Σζαγθαξάθε
Ε ΔΘΝΗΚΖ ΥΟΛΖ ΓΖΜΟΗΑ ΓΗΟΗΚΖΖ Κ ΔΚΠΑΗΓΔΤΣΗΚΖ ΔΗΡΑ ΣΜΖΜΑ : Σνπξηζηηθήο Οηθνλνκίαο θαη Αλάπηπμεο (ΣΟΑ) ΣΔΛΗΚΖ ΔΡΓΑΗΑ Θέκα: Σνπξηζκφο θαη Οηθνλνκηθή Κξίζε Δπηβιέπσλ : Νηνχβαο Λνπθάο πνπδάζηξηα : Σζαγθαξάθε
ΟΙΚΟΝΟΜΟΤΕΧΝΙΚΗ ΑΝΑΛΥΣΗ ΕΝΟΣ ΕΝΕΡΓΕΙΑΚΑ ΑΥΤΟΝΟΜΟΥ ΝΗΣΙΟΥ ΜΕ Α.Π.Ε
 Τμήμα Ηλεκτρονικών Μηχανικών Τ.Ε. ΟΙΚΟΝΟΜΟΤΕΧΝΙΚΗ ΑΝΑΛΥΣΗ ΕΝΟΣ ΕΝΕΡΓΕΙΑΚΑ ΑΥΤΟΝΟΜΟΥ ΝΗΣΙΟΥ ΜΕ Α.Π.Ε Πτυχιακή Εργασία Φοιτητής: Γεμενής Κωνσταντίνος ΑΜ: 30931 Επιβλέπων Καθηγητής Κοκκόσης Απόστολος Λέκτορας
Τμήμα Ηλεκτρονικών Μηχανικών Τ.Ε. ΟΙΚΟΝΟΜΟΤΕΧΝΙΚΗ ΑΝΑΛΥΣΗ ΕΝΟΣ ΕΝΕΡΓΕΙΑΚΑ ΑΥΤΟΝΟΜΟΥ ΝΗΣΙΟΥ ΜΕ Α.Π.Ε Πτυχιακή Εργασία Φοιτητής: Γεμενής Κωνσταντίνος ΑΜ: 30931 Επιβλέπων Καθηγητής Κοκκόσης Απόστολος Λέκτορας
HOMEWORK 4 = G. In order to plot the stress versus the stretch we define a normalized stretch:
 HOMEWORK 4 Problem a For the fast loading case, we want to derive the relationship between P zz and λ z. We know that the nominal stress is expressed as: P zz = ψ λ z where λ z = λ λ z. Therefore, applying
HOMEWORK 4 Problem a For the fast loading case, we want to derive the relationship between P zz and λ z. We know that the nominal stress is expressed as: P zz = ψ λ z where λ z = λ λ z. Therefore, applying
[1] P Q. Fig. 3.1
![[1] P Q. Fig. 3.1 [1] P Q. Fig. 3.1](/thumbs/79/80362156.jpg) 1 (a) Define resistance....... [1] (b) The smallest conductor within a computer processing chip can be represented as a rectangular block that is one atom high, four atoms wide and twenty atoms long. One
1 (a) Define resistance....... [1] (b) The smallest conductor within a computer processing chip can be represented as a rectangular block that is one atom high, four atoms wide and twenty atoms long. One
ΧΑΡΑΚΤΗΡΙΣΤΙΚΑ ΑΠΟΔΟΣΗΣ ΜΗΧΑΝΩΝ ΕΣΩΤΕΡΙΚΗΣ ΚΑΥΣΗΣ ΜΕ ΧΡΗΣΗ ΒΙΟΚΑΥΣΙΜΩΝ
 ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙΔΕΥΤΙΚΟ ΙΔΡΥΜΑ ΚΑΒΑΛΑΣ ΣΧΟΛΗ ΤΕΧΝΟΛΟΓΙΚΩΝ ΕΦΑΡΜΟΓΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΧΑΡΑΚΤΗΡΙΣΤΙΚΑ ΑΠΟΔΟΣΗΣ ΜΗΧΑΝΩΝ ΕΣΩΤΕΡΙΚΗΣ ΚΑΥΣΗΣ ΜΕ ΧΡΗΣΗ ΒΙΟΚΑΥΣΙΜΩΝ ΒΑΣΙΛΟΠΟΥΛΟΣ ΦΩΤΗΣ Π. ΜΑΝΟΥΣΟΣ ΝΙΚΟΛΑΟΣ Α. ΕΠΙΒΛΕΠΟΥΣΑ:
ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙΔΕΥΤΙΚΟ ΙΔΡΥΜΑ ΚΑΒΑΛΑΣ ΣΧΟΛΗ ΤΕΧΝΟΛΟΓΙΚΩΝ ΕΦΑΡΜΟΓΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΧΑΡΑΚΤΗΡΙΣΤΙΚΑ ΑΠΟΔΟΣΗΣ ΜΗΧΑΝΩΝ ΕΣΩΤΕΡΙΚΗΣ ΚΑΥΣΗΣ ΜΕ ΧΡΗΣΗ ΒΙΟΚΑΥΣΙΜΩΝ ΒΑΣΙΛΟΠΟΥΛΟΣ ΦΩΤΗΣ Π. ΜΑΝΟΥΣΟΣ ΝΙΚΟΛΑΟΣ Α. ΕΠΙΒΛΕΠΟΥΣΑ:
CH 3 CH 2 COOCH 2 CH 2 CH 3 + H 2 O
 Q1.An experiment was carried out to determine the equilibrium constant, K c, for the following reaction. CH 3 CH 2 COOH + CH 3 CH 2 CH 2 OH CH 3 CH 2 COOCH 2 CH 2 CH 3 + H 2 O A student added measured
Q1.An experiment was carried out to determine the equilibrium constant, K c, for the following reaction. CH 3 CH 2 COOH + CH 3 CH 2 CH 2 OH CH 3 CH 2 COOCH 2 CH 2 CH 3 + H 2 O A student added measured
Πανεπιστήμιο Πειραιώς Τμήμα Πληροφορικής Πρόγραμμα Μεταπτυχιακών Σπουδών «Πληροφορική»
 Πανεπιστήμιο Πειραιώς Τμήμα Πληροφορικής Πρόγραμμα Μεταπτυχιακών Σπουδών «Πληροφορική» Μεταπτυχιακή Διατριβή Τίτλος Διατριβής Επίκαιρα Θέματα Ηλεκτρονικής Διακυβέρνησης Ονοματεπώνυμο Φοιτητή Σταμάτιος
Πανεπιστήμιο Πειραιώς Τμήμα Πληροφορικής Πρόγραμμα Μεταπτυχιακών Σπουδών «Πληροφορική» Μεταπτυχιακή Διατριβή Τίτλος Διατριβής Επίκαιρα Θέματα Ηλεκτρονικής Διακυβέρνησης Ονοματεπώνυμο Φοιτητή Σταμάτιος
ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΕΙΡΑΙΩΣ ΤΜΗΜΑ ΝΑΥΤΙΛΙΑΚΩΝ ΣΠΟΥΔΩΝ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΗ ΝΑΥΤΙΛΙΑ
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΕΙΡΑΙΩΣ ΤΜΗΜΑ ΝΑΥΤΙΛΙΑΚΩΝ ΣΠΟΥΔΩΝ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΗ ΝΑΥΤΙΛΙΑ ΕΝΑΛΛΑΚΤΙΚΕΣ ΠΗΓΕΣ ΕΝΕΡΓΕΙΑΣ ΣΤΙΣ ΘΑΛΑΣΣΙΕΣ ΜΕΤΑΦΟΡΕΣ. ΠΕΡΙΒΑΛΛΟΝΤΙΚΑ ΟΦΕΛΗ ΤΗΣ ΝΕΑΣ ΤΕΧΝΟΛΟΓΙΑΣ. ΟΙΚΟΝΟΜΙΚΑ
ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΕΙΡΑΙΩΣ ΤΜΗΜΑ ΝΑΥΤΙΛΙΑΚΩΝ ΣΠΟΥΔΩΝ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΗ ΝΑΥΤΙΛΙΑ ΕΝΑΛΛΑΚΤΙΚΕΣ ΠΗΓΕΣ ΕΝΕΡΓΕΙΑΣ ΣΤΙΣ ΘΑΛΑΣΣΙΕΣ ΜΕΤΑΦΟΡΕΣ. ΠΕΡΙΒΑΛΛΟΝΤΙΚΑ ΟΦΕΛΗ ΤΗΣ ΝΕΑΣ ΤΕΧΝΟΛΟΓΙΑΣ. ΟΙΚΟΝΟΜΙΚΑ
ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΤΜΗΜΑ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ ΥΠΟΛΟΓΙΣΤΩΝ ΤΟΜΕΑΣ ΣΥΣΤΗΜΑΤΩΝ ΗΛΕΚΤΡΙΚΗΣ ΕΝΕΡΓΕΙΑΣ ΕΡΓΑΣΤΗΡΙΟ ΥΨΗΛΩΝ ΤΑΣΕΩΝ
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΤΜΗΜΑ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ ΥΠΟΛΟΓΙΣΤΩΝ ΤΟΜΕΑΣ ΣΥΣΤΗΜΑΤΩΝ ΗΛΕΚΤΡΙΚΗΣ ΕΝΕΡΓΕΙΑΣ ΕΡΓΑΣΤΗΡΙΟ ΥΨΗΛΩΝ ΤΑΣΕΩΝ Διπλωµατική Εργασία Της Φοιτήτριας του Τµήµατος Ηλεκτρολόγων
ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΤΜΗΜΑ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ ΥΠΟΛΟΓΙΣΤΩΝ ΤΟΜΕΑΣ ΣΥΣΤΗΜΑΤΩΝ ΗΛΕΚΤΡΙΚΗΣ ΕΝΕΡΓΕΙΑΣ ΕΡΓΑΣΤΗΡΙΟ ΥΨΗΛΩΝ ΤΑΣΕΩΝ Διπλωµατική Εργασία Της Φοιτήτριας του Τµήµατος Ηλεκτρολόγων
Capacitors - Capacitance, Charge and Potential Difference
 Capacitors - Capacitance, Charge and Potential Difference Capacitors store electric charge. This ability to store electric charge is known as capacitance. A simple capacitor consists of 2 parallel metal
Capacitors - Capacitance, Charge and Potential Difference Capacitors store electric charge. This ability to store electric charge is known as capacitance. A simple capacitor consists of 2 parallel metal
Food production and climate change
 Food production and climate change Charalampos Theopemptou Commissioner for the Environment http://perivallon.eu Ακάμας Χαράλαμπος Θεοπέμπτου - Επίτροπος Περιβάλλοντος 2 The Med area Χαράλαμπος Θεοπέμπτου
Food production and climate change Charalampos Theopemptou Commissioner for the Environment http://perivallon.eu Ακάμας Χαράλαμπος Θεοπέμπτου - Επίτροπος Περιβάλλοντος 2 The Med area Χαράλαμπος Θεοπέμπτου
ΣΕΝΑΡΙΟ ΑΝΑΦΟΡΑΣ ΣΕΝΑΡΙΟ ΤΩΝ ΜΗΧΑΝΙΚΩΝ ΣΕΝΑΡΙΟ ΣΥΝΕΡΓΑΣΙΑΣ
 ΑΝΑΠΤΥΞΗ ΚΑΙ ΕΦΑΡΜΟΓΗ ΠΟΛΙΤΙΚΗΣ ΟΛΟΚΛΗΡΩΜΕΝΗΣ ΔΙΑΧΕΙΡΙΣΗΣ ΥΔΑΤΙΚΩΝ ΠΟΡΩΝ ΣΕ ΜΙΑ ΥΔΡΟΛΟΓΙΚΗ ΛΕΚΑΝΗ ΜΕ ΤΗΝ ΕΦΑΡΜΟΓΗ ΜΙΑΣ ΔΗΜΟΣΙΑΣ ΚΟΙΝΩΝΙΚΗΣ ΣΥΜΦΩΝΙΑΣ ΣΤΗ ΒΑΣΗ ΤΩΝ ΑΡΧΩΝ ΤΗΣ AGENDA 21 ΚΑΙ ΤΩΝ ΚΑΤΕΥΘΥΝΣΕΩΝ
ΑΝΑΠΤΥΞΗ ΚΑΙ ΕΦΑΡΜΟΓΗ ΠΟΛΙΤΙΚΗΣ ΟΛΟΚΛΗΡΩΜΕΝΗΣ ΔΙΑΧΕΙΡΙΣΗΣ ΥΔΑΤΙΚΩΝ ΠΟΡΩΝ ΣΕ ΜΙΑ ΥΔΡΟΛΟΓΙΚΗ ΛΕΚΑΝΗ ΜΕ ΤΗΝ ΕΦΑΡΜΟΓΗ ΜΙΑΣ ΔΗΜΟΣΙΑΣ ΚΟΙΝΩΝΙΚΗΣ ΣΥΜΦΩΝΙΑΣ ΣΤΗ ΒΑΣΗ ΤΩΝ ΑΡΧΩΝ ΤΗΣ AGENDA 21 ΚΑΙ ΤΩΝ ΚΑΤΕΥΘΥΝΣΕΩΝ
EE512: Error Control Coding
 EE512: Error Control Coding Solution for Assignment on Finite Fields February 16, 2007 1. (a) Addition and Multiplication tables for GF (5) and GF (7) are shown in Tables 1 and 2. + 0 1 2 3 4 0 0 1 2 3
EE512: Error Control Coding Solution for Assignment on Finite Fields February 16, 2007 1. (a) Addition and Multiplication tables for GF (5) and GF (7) are shown in Tables 1 and 2. + 0 1 2 3 4 0 0 1 2 3
ΕΦΑΡΜΟΓΗ ΕΥΤΕΡΟΒΑΘΜΙΑ ΕΠΕΞΕΡΓΑΣΜΕΝΩΝ ΥΓΡΩΝ ΑΠΟΒΛΗΤΩΝ ΣΕ ΦΥΣΙΚΑ ΣΥΣΤΗΜΑΤΑ ΚΛΙΝΗΣ ΚΑΛΑΜΙΩΝ
 ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙ ΕΥΤΙΚΟ Ι ΡΥΜΑ ΚΡΗΤΗΣ ΤΜΗΜΑ ΦΥΣΙΚΩΝ ΠΟΡΩΝ ΚΑΙ ΠΕΡΙΒΑΛΛΟΝΤΟΣ ΕΦΑΡΜΟΓΗ ΕΥΤΕΡΟΒΑΘΜΙΑ ΕΠΕΞΕΡΓΑΣΜΕΝΩΝ ΥΓΡΩΝ ΑΠΟΒΛΗΤΩΝ ΣΕ ΦΥΣΙΚΑ ΣΥΣΤΗΜΑΤΑ ΚΛΙΝΗΣ ΚΑΛΑΜΙΩΝ ΕΠΙΜΕΛΕΙΑ: ΑΡΜΕΝΑΚΑΣ ΜΑΡΙΝΟΣ ΧΑΝΙΑ
ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙ ΕΥΤΙΚΟ Ι ΡΥΜΑ ΚΡΗΤΗΣ ΤΜΗΜΑ ΦΥΣΙΚΩΝ ΠΟΡΩΝ ΚΑΙ ΠΕΡΙΒΑΛΛΟΝΤΟΣ ΕΦΑΡΜΟΓΗ ΕΥΤΕΡΟΒΑΘΜΙΑ ΕΠΕΞΕΡΓΑΣΜΕΝΩΝ ΥΓΡΩΝ ΑΠΟΒΛΗΤΩΝ ΣΕ ΦΥΣΙΚΑ ΣΥΣΤΗΜΑΤΑ ΚΛΙΝΗΣ ΚΑΛΑΜΙΩΝ ΕΠΙΜΕΛΕΙΑ: ΑΡΜΕΝΑΚΑΣ ΜΑΡΙΝΟΣ ΧΑΝΙΑ
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΓΕΩΤΕΧΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΚΑΙ ΔΙΑΧΕΙΡΙΣΗΣ ΠΕΡΙΒΑΛΛΟΝΤΟΣ. Πτυχιακή εργασία
 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΓΕΩΤΕΧΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΚΑΙ ΔΙΑΧΕΙΡΙΣΗΣ ΠΕΡΙΒΑΛΛΟΝΤΟΣ Πτυχιακή εργασία ΑΝΑΛΥΣΗ ΚΟΣΤΟΥΣ-ΟΦΕΛΟΥΣ ΓΙΑ ΤΗ ΔΙΕΙΣΔΥΣΗ ΤΩΝ ΑΝΑΝΕΩΣΙΜΩΝ ΠΗΓΩΝ ΕΝΕΡΓΕΙΑΣ ΣΤΗΝ ΚΥΠΡΟ ΜΕΧΡΙ ΤΟ 2030
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΓΕΩΤΕΧΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΚΑΙ ΔΙΑΧΕΙΡΙΣΗΣ ΠΕΡΙΒΑΛΛΟΝΤΟΣ Πτυχιακή εργασία ΑΝΑΛΥΣΗ ΚΟΣΤΟΥΣ-ΟΦΕΛΟΥΣ ΓΙΑ ΤΗ ΔΙΕΙΣΔΥΣΗ ΤΩΝ ΑΝΑΝΕΩΣΙΜΩΝ ΠΗΓΩΝ ΕΝΕΡΓΕΙΑΣ ΣΤΗΝ ΚΥΠΡΟ ΜΕΧΡΙ ΤΟ 2030
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΓΕΩΤΕΧΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΚΑΙ ΔΙΑΧΕΙΡΙΣΗΣ ΠΕΡΙΒΑΛΛΟΝΤΟΣ. Πτυχιακή εργασία
 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΓΕΩΤΕΧΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΚΑΙ ΔΙΑΧΕΙΡΙΣΗΣ ΠΕΡΙΒΑΛΛΟΝΤΟΣ Πτυχιακή εργασία ΥΔΡΟΠΟΝΙΚΗ ΚΑΛΛΙΕΡΓΕΙΑ ΔΥΟΣΜΟΥ ΣΕ ΔΙΑΦΟΡΕΤΙΚΑ ΘΡΕΠΤΙΚΑ ΔΙΑΛΥΜΑΤΑ ΕΡΑΤΩ ΝΙΚΟΛΑΪΔΟΥ Λεμεσός 2014
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΓΕΩΤΕΧΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΚΑΙ ΔΙΑΧΕΙΡΙΣΗΣ ΠΕΡΙΒΑΛΛΟΝΤΟΣ Πτυχιακή εργασία ΥΔΡΟΠΟΝΙΚΗ ΚΑΛΛΙΕΡΓΕΙΑ ΔΥΟΣΜΟΥ ΣΕ ΔΙΑΦΟΡΕΤΙΚΑ ΘΡΕΠΤΙΚΑ ΔΙΑΛΥΜΑΤΑ ΕΡΑΤΩ ΝΙΚΟΛΑΪΔΟΥ Λεμεσός 2014
ΚΥΠΡΙΑΚΗ ΕΤΑΙΡΕΙΑ ΠΛΗΡΟΦΟΡΙΚΗΣ CYPRUS COMPUTER SOCIETY ΠΑΓΚΥΠΡΙΟΣ ΜΑΘΗΤΙΚΟΣ ΔΙΑΓΩΝΙΣΜΟΣ ΠΛΗΡΟΦΟΡΙΚΗΣ 19/5/2007
 Οδηγίες: Να απαντηθούν όλες οι ερωτήσεις. Αν κάπου κάνετε κάποιες υποθέσεις να αναφερθούν στη σχετική ερώτηση. Όλα τα αρχεία που αναφέρονται στα προβλήματα βρίσκονται στον ίδιο φάκελο με το εκτελέσιμο
Οδηγίες: Να απαντηθούν όλες οι ερωτήσεις. Αν κάπου κάνετε κάποιες υποθέσεις να αναφερθούν στη σχετική ερώτηση. Όλα τα αρχεία που αναφέρονται στα προβλήματα βρίσκονται στον ίδιο φάκελο με το εκτελέσιμο
Η ΝΟΜΟΘΕΣΙΑ ΤΗΣ Ε.Ε. ΣΧΕΤΙΚΑ ΜΕ ΤΗΝ ΑΤΜΟΣΦΑΙΡΙΚΗ ΡΥΠΑΝΣΗ ΑΠΟ ΠΛΟΙΑ ΚΑΙ ΟΙ ΠΡΟΚΛΗΣΕΙΣ ΣΤΗΝ ΕΦΑΡΜΟΓΗ ΤΗΣ
 Σχολή Γεωτεχνικών Επιστημών και Διαχείρισης Περιβάλλοντος Πτυχιακή εργασία Η ΝΟΜΟΘΕΣΙΑ ΤΗΣ Ε.Ε. ΣΧΕΤΙΚΑ ΜΕ ΤΗΝ ΑΤΜΟΣΦΑΙΡΙΚΗ ΡΥΠΑΝΣΗ ΑΠΟ ΠΛΟΙΑ ΚΑΙ ΟΙ ΠΡΟΚΛΗΣΕΙΣ ΣΤΗΝ ΕΦΑΡΜΟΓΗ ΤΗΣ Ελένη Άσπρου Λεμεσός, Μάιος
Σχολή Γεωτεχνικών Επιστημών και Διαχείρισης Περιβάλλοντος Πτυχιακή εργασία Η ΝΟΜΟΘΕΣΙΑ ΤΗΣ Ε.Ε. ΣΧΕΤΙΚΑ ΜΕ ΤΗΝ ΑΤΜΟΣΦΑΙΡΙΚΗ ΡΥΠΑΝΣΗ ΑΠΟ ΠΛΟΙΑ ΚΑΙ ΟΙ ΠΡΟΚΛΗΣΕΙΣ ΣΤΗΝ ΕΦΑΡΜΟΓΗ ΤΗΣ Ελένη Άσπρου Λεμεσός, Μάιος
AΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΠΟΛΥΤΕΧΝΙΚΗ ΣΧΟΛΗ ΤΜΗΜΑ ΠΟΛΙΤΙΚΩΝ ΜΗΧΑΝΙΚΩΝ
 AΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΠΟΛΥΤΕΧΝΙΚΗ ΣΧΟΛΗ ΤΜΗΜΑ ΠΟΛΙΤΙΚΩΝ ΜΗΧΑΝΙΚΩΝ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΕΙΔΙΚΕΥΣΗΣ ΠΡΟΣΤΑΣΙΑ ΠΕΡΙΒΑΛΛΟΝΤΟΣ ΚΑΙ ΒΙΩΣΙΜΗ ΑΝΑΠΤΥΞΗ ΔΙΕΡΕΥΝΗΣΗ ΤΩΝ ΠΙΕΣΕΩΝ ΣΤΟ ΠΕΡΙΒΑΛΛΟΝ
AΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΠΟΛΥΤΕΧΝΙΚΗ ΣΧΟΛΗ ΤΜΗΜΑ ΠΟΛΙΤΙΚΩΝ ΜΗΧΑΝΙΚΩΝ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΕΙΔΙΚΕΥΣΗΣ ΠΡΟΣΤΑΣΙΑ ΠΕΡΙΒΑΛΛΟΝΤΟΣ ΚΑΙ ΒΙΩΣΙΜΗ ΑΝΑΠΤΥΞΗ ΔΙΕΡΕΥΝΗΣΗ ΤΩΝ ΠΙΕΣΕΩΝ ΣΤΟ ΠΕΡΙΒΑΛΛΟΝ
Το μελλοντικό Ευρωπαϊκό Ενεργειακό σύστημα: Υλοποίηση των ενεργειακών στόχων για το 2030
 DEPARTMENT OF ELECTRICAL ENGINEERING, COMPUTER ENGINEERING AND INFORMATICS Το μελλοντικό Ευρωπαϊκό Ενεργειακό σύστημα: Υλοποίηση των ενεργειακών στόχων για το 2030 Dr. Andreas Poullikkas Chair and Associate
DEPARTMENT OF ELECTRICAL ENGINEERING, COMPUTER ENGINEERING AND INFORMATICS Το μελλοντικό Ευρωπαϊκό Ενεργειακό σύστημα: Υλοποίηση των ενεργειακών στόχων για το 2030 Dr. Andreas Poullikkas Chair and Associate
ΣΧΕΔΙΑΣΜΟΣ ΠΑΡΑΒΟΛΙΚΟΥ ΣΥΛΛΕΚΤΗ ΓΙΑ ΠΑΡΑΓΩΓΗ ΘΕΡΜΟΤΗΤΑΣ ΣΕ ΒΙΟΜΗΧΑΝΙΕΣ ΠΑΝΑΓΙΩΤΗΣ ΚΤΙΣΤΗΣ
 Μεταπτυχιακή διατριβή ΣΧΕΔΙΑΣΜΟΣ ΠΑΡΑΒΟΛΙΚΟΥ ΣΥΛΛΕΚΤΗ ΓΙΑ ΠΑΡΑΓΩΓΗ ΘΕΡΜΟΤΗΤΑΣ ΣΕ ΒΙΟΜΗΧΑΝΙΕΣ ΠΑΝΑΓΙΩΤΗΣ ΚΤΙΣΤΗΣ Λεμεσός, Μάιος 2017 i ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΜΗΧΑΝΙΚΗΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ ΤΜΗΜΑ
Μεταπτυχιακή διατριβή ΣΧΕΔΙΑΣΜΟΣ ΠΑΡΑΒΟΛΙΚΟΥ ΣΥΛΛΕΚΤΗ ΓΙΑ ΠΑΡΑΓΩΓΗ ΘΕΡΜΟΤΗΤΑΣ ΣΕ ΒΙΟΜΗΧΑΝΙΕΣ ΠΑΝΑΓΙΩΤΗΣ ΚΤΙΣΤΗΣ Λεμεσός, Μάιος 2017 i ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΜΗΧΑΝΙΚΗΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ ΤΜΗΜΑ
4 th SE European CODE Workshop 10 th 11 th of March 2011, Thessaloniki, Greece
 4 th SE European CODE Workshop 10 th 11 th of March 2011, Thessaloniki, Greece SESSION II: Best case Cogeneration project(s) in Greece Dimitris Miras Head of Thermo-electric Power Projects Dpt, ITA Group
4 th SE European CODE Workshop 10 th 11 th of March 2011, Thessaloniki, Greece SESSION II: Best case Cogeneration project(s) in Greece Dimitris Miras Head of Thermo-electric Power Projects Dpt, ITA Group
Conductivity Logging for Thermal Spring Well
 /.,**. 25 +,1- **-- 0/2,,,1- **-- 0/2, +,, +/., +0 /,* Conductivity Logging for Thermal Spring Well Koji SATO +, Tadashi TAKAYA,, Tadashi CHIBA, + Nihon Chika Kenkyuusho Co. Ltd., 0/2,, Hongo, Funabashi,
/.,**. 25 +,1- **-- 0/2,,,1- **-- 0/2, +,, +/., +0 /,* Conductivity Logging for Thermal Spring Well Koji SATO +, Tadashi TAKAYA,, Tadashi CHIBA, + Nihon Chika Kenkyuusho Co. Ltd., 0/2,, Hongo, Funabashi,
Biodiesel quality and EN 14214:2012
 3η Ενότητα: «Αγορά Βιοκαυσίμων στην Ελλάδα: Τάσεις και Προοπτικές» Biodiesel quality and EN 14214:2012 Dr. Hendrik Stein Pilot Plant Manager, ASG Analytik Content Introduction Development of the Biodiesel
3η Ενότητα: «Αγορά Βιοκαυσίμων στην Ελλάδα: Τάσεις και Προοπτικές» Biodiesel quality and EN 14214:2012 Dr. Hendrik Stein Pilot Plant Manager, ASG Analytik Content Introduction Development of the Biodiesel
ΓΕΩΜΕΣΡΙΚΗ ΣΕΚΜΗΡΙΩΗ ΣΟΤ ΙΕΡΟΤ ΝΑΟΤ ΣΟΤ ΣΙΜΙΟΤ ΣΑΤΡΟΤ ΣΟ ΠΕΛΕΝΔΡΙ ΣΗ ΚΤΠΡΟΤ ΜΕ ΕΦΑΡΜΟΓΗ ΑΤΣΟΜΑΣΟΠΟΙΗΜΕΝΟΤ ΤΣΗΜΑΣΟ ΨΗΦΙΑΚΗ ΦΩΣΟΓΡΑΜΜΕΣΡΙΑ
 ΕΘΝΙΚΟ ΜΕΣΟΒΙΟ ΠΟΛΤΣΕΧΝΕΙΟ ΣΜΗΜΑ ΑΓΡΟΝΟΜΩΝ-ΣΟΠΟΓΡΑΦΩΝ ΜΗΧΑΝΙΚΩΝ ΣΟΜΕΑ ΣΟΠΟΓΡΑΦΙΑ ΕΡΓΑΣΗΡΙΟ ΦΩΣΟΓΡΑΜΜΕΣΡΙΑ ΓΕΩΜΕΣΡΙΚΗ ΣΕΚΜΗΡΙΩΗ ΣΟΤ ΙΕΡΟΤ ΝΑΟΤ ΣΟΤ ΣΙΜΙΟΤ ΣΑΤΡΟΤ ΣΟ ΠΕΛΕΝΔΡΙ ΣΗ ΚΤΠΡΟΤ ΜΕ ΕΦΑΡΜΟΓΗ ΑΤΣΟΜΑΣΟΠΟΙΗΜΕΝΟΤ
ΕΘΝΙΚΟ ΜΕΣΟΒΙΟ ΠΟΛΤΣΕΧΝΕΙΟ ΣΜΗΜΑ ΑΓΡΟΝΟΜΩΝ-ΣΟΠΟΓΡΑΦΩΝ ΜΗΧΑΝΙΚΩΝ ΣΟΜΕΑ ΣΟΠΟΓΡΑΦΙΑ ΕΡΓΑΣΗΡΙΟ ΦΩΣΟΓΡΑΜΜΕΣΡΙΑ ΓΕΩΜΕΣΡΙΚΗ ΣΕΚΜΗΡΙΩΗ ΣΟΤ ΙΕΡΟΤ ΝΑΟΤ ΣΟΤ ΣΙΜΙΟΤ ΣΑΤΡΟΤ ΣΟ ΠΕΛΕΝΔΡΙ ΣΗ ΚΤΠΡΟΤ ΜΕ ΕΦΑΡΜΟΓΗ ΑΤΣΟΜΑΣΟΠΟΙΗΜΕΝΟΤ
Αγγλική Τουριστική Ορολογία
 ΕΛΛΗΝΙΚΗ ΔΗΜΟΚΡΑΤΙΑ Ανώτατο Εκπαιδευτικό Ίδρυμα Πειραιά Τεχνολογικού Τομέα Αγγλική Τουριστική Ορολογία Ενότητα 3: Exercises Κουτσογιάννη Ευαγγελία Τμήμα Διοίκηση Επιχειρήσεων Άδειες Χρήσης Το παρόν εκπαιδευτικό
ΕΛΛΗΝΙΚΗ ΔΗΜΟΚΡΑΤΙΑ Ανώτατο Εκπαιδευτικό Ίδρυμα Πειραιά Τεχνολογικού Τομέα Αγγλική Τουριστική Ορολογία Ενότητα 3: Exercises Κουτσογιάννη Ευαγγελία Τμήμα Διοίκηση Επιχειρήσεων Άδειες Χρήσης Το παρόν εκπαιδευτικό
Παρουζίαζη ζηην Ημερίδα ΥΝΕΡΓΕΙΑ 15 Ιανουαρίου A success story: Waste management in UK. Γπ. Θάνορ Μποςπηζάλαρ
 Παρουζίαζη ζηην Ημερίδα ΥΝΕΡΓΕΙΑ 15 Ιανουαρίου 2016 A success story: Waste management in UK Γπ. Θάνορ Μποςπηζάλαρ Ηνυμένο Βαζίλειο Πληθυσμός (εκ. πολίτες) Πληθςζμόρ και πποβλέτειρ Γείκηηρ αειθόπος διασείπιζηρ
Παρουζίαζη ζηην Ημερίδα ΥΝΕΡΓΕΙΑ 15 Ιανουαρίου 2016 A success story: Waste management in UK Γπ. Θάνορ Μποςπηζάλαρ Ηνυμένο Βαζίλειο Πληθυσμός (εκ. πολίτες) Πληθςζμόρ και πποβλέτειρ Γείκηηρ αειθόπος διασείπιζηρ
ΠΑΝΔΠΗΣΖΜΗΟ ΠΑΣΡΩΝ ΓΗΑΣΜΖΜΑΣΗΚΟ ΠΡΟΓΡΑΜΜΑ ΜΔΣΑΠΣΤΥΗΑΚΩΝ ΠΟΤΓΩΝ «ΤΣΖΜΑΣΑ ΔΠΔΞΔΡΓΑΗΑ ΖΜΑΣΩΝ ΚΑΗ ΔΠΗΚΟΗΝΩΝΗΩΝ» ΣΜΖΜΑ ΜΖΥΑΝΗΚΩΝ Ζ/Τ ΚΑΗ ΠΛΖΡΟΦΟΡΗΚΖ
 ΠΑΝΔΠΗΣΖΜΗΟ ΠΑΣΡΩΝ ΓΗΑΣΜΖΜΑΣΗΚΟ ΠΡΟΓΡΑΜΜΑ ΜΔΣΑΠΣΤΥΗΑΚΩΝ ΠΟΤΓΩΝ «ΤΣΖΜΑΣΑ ΔΠΔΞΔΡΓΑΗΑ ΖΜΑΣΩΝ ΚΑΗ ΔΠΗΚΟΗΝΩΝΗΩΝ» ΣΜΖΜΑ ΜΖΥΑΝΗΚΩΝ Ζ/Τ ΚΑΗ ΠΛΖΡΟΦΟΡΗΚΖ ΣΜΖΜΑ ΖΛΔΚΣΡΟΛΟΓΩΝ ΜΖΥΑΝΗΚΩΝ ΚΑΗ ΣΔΥΝΟΛΟΓΗΑ ΤΠΟΛΟΓΗΣΩΝ ΣΜΖΜΑ
ΠΑΝΔΠΗΣΖΜΗΟ ΠΑΣΡΩΝ ΓΗΑΣΜΖΜΑΣΗΚΟ ΠΡΟΓΡΑΜΜΑ ΜΔΣΑΠΣΤΥΗΑΚΩΝ ΠΟΤΓΩΝ «ΤΣΖΜΑΣΑ ΔΠΔΞΔΡΓΑΗΑ ΖΜΑΣΩΝ ΚΑΗ ΔΠΗΚΟΗΝΩΝΗΩΝ» ΣΜΖΜΑ ΜΖΥΑΝΗΚΩΝ Ζ/Τ ΚΑΗ ΠΛΖΡΟΦΟΡΗΚΖ ΣΜΖΜΑ ΖΛΔΚΣΡΟΛΟΓΩΝ ΜΖΥΑΝΗΚΩΝ ΚΑΗ ΣΔΥΝΟΛΟΓΗΑ ΤΠΟΛΟΓΗΣΩΝ ΣΜΖΜΑ
Instruction Execution Times
 1 C Execution Times InThisAppendix... Introduction DL330 Execution Times DL330P Execution Times DL340 Execution Times C-2 Execution Times Introduction Data Registers This appendix contains several tables
1 C Execution Times InThisAppendix... Introduction DL330 Execution Times DL330P Execution Times DL340 Execution Times C-2 Execution Times Introduction Data Registers This appendix contains several tables
ΕΛΛΗΝΙΚΗ ΔΗΜΟΚΡΑΤΙΑ Ανώτατο Εκπαιδευτικό Ίδρυμα Πειραιά Τεχνολογικού Τομέα. Ξένη Ορολογία. Ενότητα 6: Working Capital
 ΕΛΛΗΝΙΚΗ ΔΗΜΟΚΡΑΤΙΑ Ανώτατο Εκπαιδευτικό Ίδρυμα Πειραιά Τεχνολογικού Τομέα Ξένη Ορολογία Ενότητα 6: Working Capital Ευαγγελία Κουτσογιάννη Τμήμα Λογιστικής & Χρηματοοικονομικής Άδειες Χρήσης Το παρόν εκπαιδευτικό
ΕΛΛΗΝΙΚΗ ΔΗΜΟΚΡΑΤΙΑ Ανώτατο Εκπαιδευτικό Ίδρυμα Πειραιά Τεχνολογικού Τομέα Ξένη Ορολογία Ενότητα 6: Working Capital Ευαγγελία Κουτσογιάννη Τμήμα Λογιστικής & Χρηματοοικονομικής Άδειες Χρήσης Το παρόν εκπαιδευτικό
Figure 1 T / K Explain, in terms of molecules, why the first part of the graph in Figure 1 is a line that slopes up from the origin.
 Q1.(a) Figure 1 shows how the entropy of a molecular substance X varies with temperature. Figure 1 T / K (i) Explain, in terms of molecules, why the entropy is zero when the temperature is zero Kelvin.
Q1.(a) Figure 1 shows how the entropy of a molecular substance X varies with temperature. Figure 1 T / K (i) Explain, in terms of molecules, why the entropy is zero when the temperature is zero Kelvin.
ΙΕΥΘΥΝΤΗΣ: Καθηγητής Γ. ΧΡΥΣΟΛΟΥΡΗΣ Ι ΑΚΤΟΡΙΚΗ ΙΑΤΡΙΒΗ
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΠΟΛΥΤΕΧΝΙΚΗ ΣΧΟΛΗ ΤΜΗΜΑ ΜΗΧΑΝΟΛΟΓΩΝ ΚΑΙ ΑΕΡΟΝΑΥΠΗΓΩΝ ΜΗΧΑΝΙΚΩΝ ΕΡΓΑΣΤΗΡΙΟ ΣΥΣΤΗΜΑΤΩΝ ΠΑΡΑΓΩΓΗΣ & ΑΥΤΟΜΑΤΙΣΜΟΥ / ΥΝΑΜΙΚΗΣ & ΘΕΩΡΙΑΣ ΜΗΧΑΝΩΝ ΙΕΥΘΥΝΤΗΣ: Καθηγητής Γ. ΧΡΥΣΟΛΟΥΡΗΣ Ι ΑΚΤΟΡΙΚΗ
ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΠΟΛΥΤΕΧΝΙΚΗ ΣΧΟΛΗ ΤΜΗΜΑ ΜΗΧΑΝΟΛΟΓΩΝ ΚΑΙ ΑΕΡΟΝΑΥΠΗΓΩΝ ΜΗΧΑΝΙΚΩΝ ΕΡΓΑΣΤΗΡΙΟ ΣΥΣΤΗΜΑΤΩΝ ΠΑΡΑΓΩΓΗΣ & ΑΥΤΟΜΑΤΙΣΜΟΥ / ΥΝΑΜΙΚΗΣ & ΘΕΩΡΙΑΣ ΜΗΧΑΝΩΝ ΙΕΥΘΥΝΤΗΣ: Καθηγητής Γ. ΧΡΥΣΟΛΟΥΡΗΣ Ι ΑΚΤΟΡΙΚΗ
Μεταπτυχιακή διατριβή. Ανδρέας Παπαευσταθίου
 Σχολή Γεωτεχνικών Επιστημών και Διαχείρισης Περιβάλλοντος Μεταπτυχιακή διατριβή Κτίρια σχεδόν μηδενικής ενεργειακής κατανάλωσης :Αξιολόγηση συστημάτων θέρμανσης -ψύξης και ΑΠΕ σε οικιστικά κτίρια στην
Σχολή Γεωτεχνικών Επιστημών και Διαχείρισης Περιβάλλοντος Μεταπτυχιακή διατριβή Κτίρια σχεδόν μηδενικής ενεργειακής κατανάλωσης :Αξιολόγηση συστημάτων θέρμανσης -ψύξης και ΑΠΕ σε οικιστικά κτίρια στην
AKAΔΗΜΙΑ ΕΜΠΟΡΙΚΟΥ ΝΑΥΤΙΚΟΥ ΜΑΚΕΔΟΝΙΑΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΘΕΜΑ: Η ΧΡΗΣΗ ΒΙΟΚΑΥΣΙΜΩΝ ΣΤΗΝ ΝΑΥΤΙΛΙΑ ΠΛΕΟΝΕΚΤΗΜΑΤΑ-ΜΕΙΟΝΕΚΤΗΜΑΤΑ ΠΡΟΟΠΤΙΚΕΣ
 AKAΔΗΜΙΑ ΕΜΠΟΡΙΚΟΥ ΝΑΥΤΙΚΟΥ ΜΑΚΕΔΟΝΙΑΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΘΕΜΑ: Η ΧΡΗΣΗ ΒΙΟΚΑΥΣΙΜΩΝ ΣΤΗΝ ΝΑΥΤΙΛΙΑ ΠΛΕΟΝΕΚΤΗΜΑΤΑ-ΜΕΙΟΝΕΚΤΗΜΑΤΑ ΠΡΟΟΠΤΙΚΕΣ ΣΠΟΥΔΑΣΤΕΣ : ΜΟΤΣΚΑΛΙΔΗΣ ΒΑΛΕΡΙΟΣ, ΠΕΛΕΚΑΝΟΣ ΙΩΑΝΝΗΣ
AKAΔΗΜΙΑ ΕΜΠΟΡΙΚΟΥ ΝΑΥΤΙΚΟΥ ΜΑΚΕΔΟΝΙΑΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΘΕΜΑ: Η ΧΡΗΣΗ ΒΙΟΚΑΥΣΙΜΩΝ ΣΤΗΝ ΝΑΥΤΙΛΙΑ ΠΛΕΟΝΕΚΤΗΜΑΤΑ-ΜΕΙΟΝΕΚΤΗΜΑΤΑ ΠΡΟΟΠΤΙΚΕΣ ΣΠΟΥΔΑΣΤΕΣ : ΜΟΤΣΚΑΛΙΔΗΣ ΒΑΛΕΡΙΟΣ, ΠΕΛΕΚΑΝΟΣ ΙΩΑΝΝΗΣ
Σπανό Ιωάννη Α.Μ. 148
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΔΙΑΤΜΗΜΑΤΙΚΟ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΙΣ ΠΕΡΙΒΑΛΛΟΝΤΙΚΕΣ ΕΠΙΣΤΗΜΕΣ Ηλεκτροχημική εναπόθεση και μελέτη των ιδιοτήτων, λεπτών υμενίων μεταβατικών μετάλλων, για παραγωγή H2 Διπλωματική
ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΔΙΑΤΜΗΜΑΤΙΚΟ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΙΣ ΠΕΡΙΒΑΛΛΟΝΤΙΚΕΣ ΕΠΙΣΤΗΜΕΣ Ηλεκτροχημική εναπόθεση και μελέτη των ιδιοτήτων, λεπτών υμενίων μεταβατικών μετάλλων, για παραγωγή H2 Διπλωματική
ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΤΜΗΜΑ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ ΥΠΟΛΟΓΙΣΤΩΝ ΤΟΜΕΑΣ ΣΥΣΤΗΜΑΤΩΝ ΗΛΕΚΤΡΙΚΗΣ ΕΝΕΡΓΕΙΑΣ
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΤΜΗΜΑ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ ΥΠΟΛΟΓΙΣΤΩΝ ΤΟΜΕΑΣ ΣΥΣΤΗΜΑΤΩΝ ΗΛΕΚΤΡΙΚΗΣ ΕΝΕΡΓΕΙΑΣ ιπλωµατική Εργασία του φοιτητή του τµήµατος Ηλεκτρολόγων Μηχανικών και Τεχνολογίας Ηλεκτρονικών
ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΤΜΗΜΑ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ ΥΠΟΛΟΓΙΣΤΩΝ ΤΟΜΕΑΣ ΣΥΣΤΗΜΑΤΩΝ ΗΛΕΚΤΡΙΚΗΣ ΕΝΕΡΓΕΙΑΣ ιπλωµατική Εργασία του φοιτητή του τµήµατος Ηλεκτρολόγων Μηχανικών και Τεχνολογίας Ηλεκτρονικών
ΠΡΟΣΟΜΟΙΩΣΗ ΤΗΣ ΥΠΟΓΕΙΑΣ ΡΟΗΣ ΚΑΙ ΜΕΤΑΦΟΡΑΣ ΝΙΤΡΙΚΩΝ ΣΤΟΝ ΠΑΡΑΚΤΙΟ ΥΔΡΟΦΟΡΕΑ ΤΟΥ ΠΛΑΤΥ ΠΟΤΑΜΟΥ ΣΤΟ ΝΟΜΟ ΡΕΘΥΜΝΟΥ
 ΠΟΛΥΤΕΧΝΕΙΟ ΚΡΗΤΗΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΕΡΙΒΑΛΛΟΝΤΟΣ Τομέας Περιβαλλοντικής Υδραυλικής και Γεωπεριβαλλοντικής Μηχανικής (ΙΙΙ) Εργαστήριο Γεωπεριβαλλοντικής Μηχανικής SCHOOL of ENVIRONMENTAL ENGINEERING Division
ΠΟΛΥΤΕΧΝΕΙΟ ΚΡΗΤΗΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΕΡΙΒΑΛΛΟΝΤΟΣ Τομέας Περιβαλλοντικής Υδραυλικής και Γεωπεριβαλλοντικής Μηχανικής (ΙΙΙ) Εργαστήριο Γεωπεριβαλλοντικής Μηχανικής SCHOOL of ENVIRONMENTAL ENGINEERING Division
Enthalpy data for the reacting species are given in the table below. The activation energy decreases when the temperature is increased.
 Q1.This question is about the reaction given below. O(g) + H 2O(g) O 2(g) + H 2(g) Enthalpy data for the reacting species are given in the table below. Substance O(g) H 2O(g) O 2(g) H 2(g) ΔH / kj mol
Q1.This question is about the reaction given below. O(g) + H 2O(g) O 2(g) + H 2(g) Enthalpy data for the reacting species are given in the table below. Substance O(g) H 2O(g) O 2(g) H 2(g) ΔH / kj mol
Study of urban housing development projects: The general planning of Alexandria City
 Paper published at Alexandria Engineering Journal, vol, No, July, Study of urban housing development projects: The general planning of Alexandria City Hisham El Shimy Architecture Department, Faculty of
Paper published at Alexandria Engineering Journal, vol, No, July, Study of urban housing development projects: The general planning of Alexandria City Hisham El Shimy Architecture Department, Faculty of
ΠΕΡΙΕΧΟΜΕΝΑ. Κεφάλαιο 1: Κεφάλαιο 2: Κεφάλαιο 3:
 4 Πρόλογος Η παρούσα διπλωµατική εργασία µε τίτλο «ιερεύνηση χωρικής κατανοµής µετεωρολογικών µεταβλητών. Εφαρµογή στον ελληνικό χώρο», ανατέθηκε από το ιεπιστηµονικό ιατµηµατικό Πρόγραµµα Μεταπτυχιακών
4 Πρόλογος Η παρούσα διπλωµατική εργασία µε τίτλο «ιερεύνηση χωρικής κατανοµής µετεωρολογικών µεταβλητών. Εφαρµογή στον ελληνικό χώρο», ανατέθηκε από το ιεπιστηµονικό ιατµηµατικό Πρόγραµµα Μεταπτυχιακών
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΓΕΩΤΕΧΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΚΑΙ ΔΙΑΧΕΙΡΙΣΗΣ ΠΕΡΙΒΑΛΛΟΝΤΟΣ ΤΜΗΜΑ ΕΠΙΣΤΗΜΗΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ ΠΕΡΙΒΑΛΛΟΝΤΟΣ
 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΓΕΩΤΕΧΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΚΑΙ ΔΙΑΧΕΙΡΙΣΗΣ ΠΕΡΙΒΑΛΛΟΝΤΟΣ ΤΜΗΜΑ ΕΠΙΣΤΗΜΗΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ ΠΕΡΙΒΑΛΛΟΝΤΟΣ ΗΛΕΚΤΡΟΚΙΝΗΣΗ ΚΑΙ ΑΝΑΛΥΣΗ ΤΗΣ VEHICLE TO GRID (V2G) ΛΕΙΤΟΥΡΓΙΑΣ Μεταπτυχιακή
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΓΕΩΤΕΧΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΚΑΙ ΔΙΑΧΕΙΡΙΣΗΣ ΠΕΡΙΒΑΛΛΟΝΤΟΣ ΤΜΗΜΑ ΕΠΙΣΤΗΜΗΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ ΠΕΡΙΒΑΛΛΟΝΤΟΣ ΗΛΕΚΤΡΟΚΙΝΗΣΗ ΚΑΙ ΑΝΑΛΥΣΗ ΤΗΣ VEHICLE TO GRID (V2G) ΛΕΙΤΟΥΡΓΙΑΣ Μεταπτυχιακή
6.1. Dirac Equation. Hamiltonian. Dirac Eq.
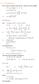 6.1. Dirac Equation Ref: M.Kaku, Quantum Field Theory, Oxford Univ Press (1993) η μν = η μν = diag(1, -1, -1, -1) p 0 = p 0 p = p i = -p i p μ p μ = p 0 p 0 + p i p i = E c 2 - p 2 = (m c) 2 H = c p 2
6.1. Dirac Equation Ref: M.Kaku, Quantum Field Theory, Oxford Univ Press (1993) η μν = η μν = diag(1, -1, -1, -1) p 0 = p 0 p = p i = -p i p μ p μ = p 0 p 0 + p i p i = E c 2 - p 2 = (m c) 2 H = c p 2
Μετρήσεις ηλιοφάνειας στην Κύπρο
 Πτυχιακή εργασία Μετρήσεις ηλιοφάνειας στην Κύπρο Ιωσήφ Μικαίος Λεμεσός, Μάιος 2018 1 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΓΕΩΤΕΧΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΚΑΙ ΔΙΑΧΕΙΡΙΣΗΣ ΠΕΡΙΒΑΛΛΟΝΤΟΣ ΤΜΗΜΑ ΕΠΙΣΤΗΜΗΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ
Πτυχιακή εργασία Μετρήσεις ηλιοφάνειας στην Κύπρο Ιωσήφ Μικαίος Λεμεσός, Μάιος 2018 1 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΓΕΩΤΕΧΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΚΑΙ ΔΙΑΧΕΙΡΙΣΗΣ ΠΕΡΙΒΑΛΛΟΝΤΟΣ ΤΜΗΜΑ ΕΠΙΣΤΗΜΗΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ
ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΕΙΡΑΙΑ ΤΜΗΜΑ ΝΑΥΤΙΛΙΑΚΩΝ ΣΠΟΥΔΩΝ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΗΝ ΝΑΥΤΙΛΙΑ
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΕΙΡΑΙΑ ΤΜΗΜΑ ΝΑΥΤΙΛΙΑΚΩΝ ΣΠΟΥΔΩΝ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΗΝ ΝΑΥΤΙΛΙΑ ΝΟΜΙΚΟ ΚΑΙ ΘΕΣΜΙΚΟ ΦΟΡΟΛΟΓΙΚΟ ΠΛΑΙΣΙΟ ΚΤΗΣΗΣ ΚΑΙ ΕΚΜΕΤΑΛΛΕΥΣΗΣ ΠΛΟΙΟΥ ΔΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ που υποβλήθηκε στο
ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΕΙΡΑΙΑ ΤΜΗΜΑ ΝΑΥΤΙΛΙΑΚΩΝ ΣΠΟΥΔΩΝ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΗΝ ΝΑΥΤΙΛΙΑ ΝΟΜΙΚΟ ΚΑΙ ΘΕΣΜΙΚΟ ΦΟΡΟΛΟΓΙΚΟ ΠΛΑΙΣΙΟ ΚΤΗΣΗΣ ΚΑΙ ΕΚΜΕΤΑΛΛΕΥΣΗΣ ΠΛΟΙΟΥ ΔΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ που υποβλήθηκε στο
 ΑΚΑ ΗΜΙΑ ΕΜΠΟΡΙΚΟΥ ΝΑΥΤΙΚΟΥ ΜΑΚΕ ΟΝΙΑΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΟΝΟΜΑΤΕΠΩΝΥΜΟ: ΓΕΩΡΓΙΟΣ ΙΩΑΝΝΟΥ Α.Μ.: 4674 ΟΝΟΜΑΤΕΠΩΝΥΜΟ ΚΑΘΗΓΗΤΡΙΑΣ: ρ.ελευθερια ΜΠΑΚΟΓΙΑΝΝΗ ΘΕΜΑ: ΟΙ ΕΠΙΠΤΩΣΕΙΣ ΤΗΣ ΠΑΡΟΥΣΙΑΣ ΧΗΜΙΚΩΝ
ΑΚΑ ΗΜΙΑ ΕΜΠΟΡΙΚΟΥ ΝΑΥΤΙΚΟΥ ΜΑΚΕ ΟΝΙΑΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΟΝΟΜΑΤΕΠΩΝΥΜΟ: ΓΕΩΡΓΙΟΣ ΙΩΑΝΝΟΥ Α.Μ.: 4674 ΟΝΟΜΑΤΕΠΩΝΥΜΟ ΚΑΘΗΓΗΤΡΙΑΣ: ρ.ελευθερια ΜΠΑΚΟΓΙΑΝΝΗ ΘΕΜΑ: ΟΙ ΕΠΙΠΤΩΣΕΙΣ ΤΗΣ ΠΑΡΟΥΣΙΑΣ ΧΗΜΙΚΩΝ
Approximation of distance between locations on earth given by latitude and longitude
 Approximation of distance between locations on earth given by latitude and longitude Jan Behrens 2012-12-31 In this paper we shall provide a method to approximate distances between two points on earth
Approximation of distance between locations on earth given by latitude and longitude Jan Behrens 2012-12-31 In this paper we shall provide a method to approximate distances between two points on earth
Γιπλυμαηική Δπγαζία. «Ανθπυποκενηπικόρ ζσεδιαζμόρ γέθςπαρ πλοίος» Φοςζιάνηρ Αθανάζιορ. Δπιβλέπυν Καθηγηηήρ: Νηθφιανο Π. Βεληίθνο
 ΔΘΝΙΚΟ ΜΔΣΟΒΙΟ ΠΟΛΤΣΔΥΝΔΙΟ ΥΟΛΗ ΝΑΤΠΗΓΩΝ ΜΗΥΑΝΟΛΟΓΩΝ ΜΗΥΑΝΙΚΩΝ Γιπλυμαηική Δπγαζία «Ανθπυποκενηπικόρ ζσεδιαζμόρ γέθςπαρ πλοίος» Φοςζιάνηρ Αθανάζιορ Δπιβλέπυν Καθηγηηήρ: Νηθφιανο Π. Βεληίθνο Σπιμελήρ Δξεηαζηική
ΔΘΝΙΚΟ ΜΔΣΟΒΙΟ ΠΟΛΤΣΔΥΝΔΙΟ ΥΟΛΗ ΝΑΤΠΗΓΩΝ ΜΗΥΑΝΟΛΟΓΩΝ ΜΗΥΑΝΙΚΩΝ Γιπλυμαηική Δπγαζία «Ανθπυποκενηπικόρ ζσεδιαζμόρ γέθςπαρ πλοίος» Φοςζιάνηρ Αθανάζιορ Δπιβλέπυν Καθηγηηήρ: Νηθφιανο Π. Βεληίθνο Σπιμελήρ Δξεηαζηική
(Biomass utilization for electric energy production)
 ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙΔΕΥΤΙΚΟ ΙΔΡΥΜΑ T.Ε.I. ΠΕΙΡΑΙΑ ΣΧΟΛΗ: ΤΕΧΝΟΛΟΓΙΚΩΝ ΕΦΑΡΜΟΓΩΝ ΤΜΗΜΑ: ΗΛΕΚΤΡΟΛΟΓΙΑΣ Επιβλέπων: ΠΕΤΡΟΣ Γ. ΒΕΡΝΑΔΟΣ, Ομότιμος Καθηγητής Συνεπιβλέπουσα: ΕΡΙΕΤΤΑ Ι. ΖΟΥΝΤΟΥΡΙΔΟΥ, Παν. Υπότροφος
ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙΔΕΥΤΙΚΟ ΙΔΡΥΜΑ T.Ε.I. ΠΕΙΡΑΙΑ ΣΧΟΛΗ: ΤΕΧΝΟΛΟΓΙΚΩΝ ΕΦΑΡΜΟΓΩΝ ΤΜΗΜΑ: ΗΛΕΚΤΡΟΛΟΓΙΑΣ Επιβλέπων: ΠΕΤΡΟΣ Γ. ΒΕΡΝΑΔΟΣ, Ομότιμος Καθηγητής Συνεπιβλέπουσα: ΕΡΙΕΤΤΑ Ι. ΖΟΥΝΤΟΥΡΙΔΟΥ, Παν. Υπότροφος
Πτυχιακή εργασία. Παραγωγή Βιοντίζελ από Χρησιμοποιημένα Έλαια
 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΤΜΗΜΑ ΕΠΙΣΤΗΜΗΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ ΠΕΡΙΒΑΛΛΟΝΤΟΣ Πτυχιακή εργασία Παραγωγή Βιοντίζελ από Χρησιμοποιημένα Έλαια Ελένη Χριστοδούλου Λεμεσός 2014 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΤΜΗΜΑ ΕΠΙΣΤΗΜΗΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ ΠΕΡΙΒΑΛΛΟΝΤΟΣ Πτυχιακή εργασία Παραγωγή Βιοντίζελ από Χρησιμοποιημένα Έλαια Ελένη Χριστοδούλου Λεμεσός 2014 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ
Math 6 SL Probability Distributions Practice Test Mark Scheme
 Math 6 SL Probability Distributions Practice Test Mark Scheme. (a) Note: Award A for vertical line to right of mean, A for shading to right of their vertical line. AA N (b) evidence of recognizing symmetry
Math 6 SL Probability Distributions Practice Test Mark Scheme. (a) Note: Award A for vertical line to right of mean, A for shading to right of their vertical line. AA N (b) evidence of recognizing symmetry
ΔΘΝΙΚΗ ΥΟΛΗ ΓΗΜΟΙΑ ΓΙΟΙΚΗΗ ΚΑ ΔΚΠΑΙΓΔΤΣΙΚΗ ΔΙΡΑ ΣΔΛΙΚΗ ΔΡΓΑΙΑ
 Ε ΔΘΝΙΚΗ ΥΟΛΗ ΓΗΜΟΙΑ ΓΙΟΙΚΗΗ ΚΑ ΔΚΠΑΙΓΔΤΣΙΚΗ ΔΙΡΑ ΣΜΗΜΑ ΓΔΝΙΚΗ ΓΙΟΙΚΗΗ ΣΔΛΙΚΗ ΔΡΓΑΙΑ Θέκα: Η Γηνίθεζε Αιιαγώλ (Change Management) ζην Γεκόζην Σνκέα: Η πεξίπησζε ηεο εθαξκνγήο ηνπ ύγρξνλνπ Γεκνζηνλνκηθνύ
Ε ΔΘΝΙΚΗ ΥΟΛΗ ΓΗΜΟΙΑ ΓΙΟΙΚΗΗ ΚΑ ΔΚΠΑΙΓΔΤΣΙΚΗ ΔΙΡΑ ΣΜΗΜΑ ΓΔΝΙΚΗ ΓΙΟΙΚΗΗ ΣΔΛΙΚΗ ΔΡΓΑΙΑ Θέκα: Η Γηνίθεζε Αιιαγώλ (Change Management) ζην Γεκόζην Σνκέα: Η πεξίπησζε ηεο εθαξκνγήο ηνπ ύγρξνλνπ Γεκνζηνλνκηθνύ
EPL 603 TOPICS IN SOFTWARE ENGINEERING. Lab 5: Component Adaptation Environment (COPE)
 EPL 603 TOPICS IN SOFTWARE ENGINEERING Lab 5: Component Adaptation Environment (COPE) Performing Static Analysis 1 Class Name: The fully qualified name of the specific class Type: The type of the class
EPL 603 TOPICS IN SOFTWARE ENGINEERING Lab 5: Component Adaptation Environment (COPE) Performing Static Analysis 1 Class Name: The fully qualified name of the specific class Type: The type of the class
ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙΔΕΥΤΙΚΟ ΙΔΡΥΜΑ ΚΡΗΤΗΣ ΣΧΟΛΗ ΔΙΟΙΚΗΣΗΣ ΚΑΙ ΟΙΚΟΝΟΜΙΑΣ ΤΜΗΜΑ ΛΟΓΙΣΤΙΚΗΣ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ
 ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙΔΕΥΤΙΚΟ ΙΔΡΥΜΑ ΚΡΗΤΗΣ ΣΧΟΛΗ ΔΙΟΙΚΗΣΗΣ ΚΑΙ ΟΙΚΟΝΟΜΙΑΣ ΤΜΗΜΑ ΛΟΓΙΣΤΙΚΗΣ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΛΟΓΙΣΤΙΚΗ ΚΑΙ ΦΟΡΟΛΟΓΙΑ Ο.Ε. ΕΙΣΗΓΗΤΡΙΑ ΚΑΘΗΓΗΤΡΙΑ: κ. ΟΥΡΑΝΟΥ ΕΡΜΙΟΝΗ ΣΠΟΥΔΑΣΤΡΙΕΣ: ΔΕΜΕΤΖΟΥ ΑΓΛΑΪΑ
ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙΔΕΥΤΙΚΟ ΙΔΡΥΜΑ ΚΡΗΤΗΣ ΣΧΟΛΗ ΔΙΟΙΚΗΣΗΣ ΚΑΙ ΟΙΚΟΝΟΜΙΑΣ ΤΜΗΜΑ ΛΟΓΙΣΤΙΚΗΣ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΛΟΓΙΣΤΙΚΗ ΚΑΙ ΦΟΡΟΛΟΓΙΑ Ο.Ε. ΕΙΣΗΓΗΤΡΙΑ ΚΑΘΗΓΗΤΡΙΑ: κ. ΟΥΡΑΝΟΥ ΕΡΜΙΟΝΗ ΣΠΟΥΔΑΣΤΡΙΕΣ: ΔΕΜΕΤΖΟΥ ΑΓΛΑΪΑ
ΜΑΡΙΟΛΑΚΟΣ Η., ΦΟΥΝΤΟΥΛΗΣ Ι., ΣΠΥΡΙΔΩΝΟΣ Ε., ΑΝΔΡΕΑΔΑΚΗΣ Ε., ΚΑΠΟΥΡΑΝΗ, Ε.
 ΔΗΜΟΣΙΕΥΣΗ Νο 95 ΜΑΡΙΟΛΑΚΟΣ Η., ΦΟΥΝΤΟΥΛΗΣ Ι., ΣΠΥΡΙΔΩΝΟΣ Ε., ΑΝΔΡΕΑΔΑΚΗΣ Ε., ΚΑΠΟΥΡΑΝΗ, Ε. (2003). Το πρόβλημα του νερού στη Θεσσαλία και προτάσεις για την αντιμετώπισή του στα πλαίσια της αειφόρου ανάπτυξης.
ΔΗΜΟΣΙΕΥΣΗ Νο 95 ΜΑΡΙΟΛΑΚΟΣ Η., ΦΟΥΝΤΟΥΛΗΣ Ι., ΣΠΥΡΙΔΩΝΟΣ Ε., ΑΝΔΡΕΑΔΑΚΗΣ Ε., ΚΑΠΟΥΡΑΝΗ, Ε. (2003). Το πρόβλημα του νερού στη Θεσσαλία και προτάσεις για την αντιμετώπισή του στα πλαίσια της αειφόρου ανάπτυξης.
«Έντυπο και ψηφιακό βιβλίο στη σύγχρονη εποχή: τάσεις στην παγκόσμια βιομηχανία».
 ΕΛΛΗΝΙΚΗ ΔΗΜΟΚΡΑΤΙΑ ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙΔΕΥΤΙΚΟ ΙΔΡΥΜΑ ΙΟΝΙΩΝ ΝΗΣΩΝ ΤΜΗΜΑ ΔΗΜΟΣΙΩΝ ΣΧΕΣΕΩΝ ΚΑΙ ΕΠΙΚΟΙΝΩΝΙΑΣ Ταχ. Δ/νση : ΑΤΕΙ Ιονίων Νήσων- Λεωφόρος Αντώνη Τρίτση Αργοστόλι- Κεφαλληνίας, Ελλάδα 28100, +30
ΕΛΛΗΝΙΚΗ ΔΗΜΟΚΡΑΤΙΑ ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙΔΕΥΤΙΚΟ ΙΔΡΥΜΑ ΙΟΝΙΩΝ ΝΗΣΩΝ ΤΜΗΜΑ ΔΗΜΟΣΙΩΝ ΣΧΕΣΕΩΝ ΚΑΙ ΕΠΙΚΟΙΝΩΝΙΑΣ Ταχ. Δ/νση : ΑΤΕΙ Ιονίων Νήσων- Λεωφόρος Αντώνη Τρίτση Αργοστόλι- Κεφαλληνίας, Ελλάδα 28100, +30
ΚΑΘΟΡΙΣΜΟΣ ΠΑΡΑΓΟΝΤΩΝ ΠΟΥ ΕΠΗΡΕΑΖΟΥΝ ΤΗΝ ΠΑΡΑΓΟΜΕΝΗ ΙΣΧΥ ΣΕ Φ/Β ΠΑΡΚΟ 80KWp
 ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΣΧΟΛΗ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΜΗΧΑΝΙΚΩΝ ΥΠΟΛΟΓΙΣΤΩΝ ΤΟΜΕΑΣ ΣΥΣΤΗΜΑΤΩΝ ΜΕΤΑΔΟΣΗΣ ΠΛΗΡΟΦΟΡΙΑΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ ΥΛΙΚΩΝ ΚΑΘΟΡΙΣΜΟΣ ΠΑΡΑΓΟΝΤΩΝ ΠΟΥ ΕΠΗΡΕΑΖΟΥΝ ΤΗΝ ΠΑΡΑΓΟΜΕΝΗ ΙΣΧΥ
ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΣΧΟΛΗ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΜΗΧΑΝΙΚΩΝ ΥΠΟΛΟΓΙΣΤΩΝ ΤΟΜΕΑΣ ΣΥΣΤΗΜΑΤΩΝ ΜΕΤΑΔΟΣΗΣ ΠΛΗΡΟΦΟΡΙΑΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ ΥΛΙΚΩΝ ΚΑΘΟΡΙΣΜΟΣ ΠΑΡΑΓΟΝΤΩΝ ΠΟΥ ΕΠΗΡΕΑΖΟΥΝ ΤΗΝ ΠΑΡΑΓΟΜΕΝΗ ΙΣΧΥ
Block Ciphers Modes. Ramki Thurimella
 Block Ciphers Modes Ramki Thurimella Only Encryption I.e. messages could be modified Should not assume that nonsensical messages do no harm Always must be combined with authentication 2 Padding Must be
Block Ciphers Modes Ramki Thurimella Only Encryption I.e. messages could be modified Should not assume that nonsensical messages do no harm Always must be combined with authentication 2 Padding Must be
GREECE BULGARIA 6 th JOINT MONITORING
 GREECE BULGARIA 6 th JOINT MONITORING COMMITTEE BANSKO 26-5-2015 «GREECE BULGARIA» Timeline 02 Future actions of the new GR-BG 20 Programme June 2015: Re - submission of the modified d Programme according
GREECE BULGARIA 6 th JOINT MONITORING COMMITTEE BANSKO 26-5-2015 «GREECE BULGARIA» Timeline 02 Future actions of the new GR-BG 20 Programme June 2015: Re - submission of the modified d Programme according
Faculty of Management and. Economics. Master s Thesis. Πορεία Ναύλων και Οικονομία: Παράγοντες που επηρεάζουν τους Δείκτες Ναύλων της Ναυτιλίας
 Faculty of Management and Economics Master s Thesis Πορεία Ναύλων και Οικονομία: Παράγοντες που επηρεάζουν τους Δείκτες Ναύλων της Ναυτιλίας ΓΙΩΡΚΑΣ ΑΝΤΡΕΑΣ ΕΠΙΒΛΕΠΩΝ ΚΑΘΗΓΗΤΗΣ : ΔΗΜΗΤΡΗΣ ΤΣΟΥΚΝΙΔΗΣ Limassol,
Faculty of Management and Economics Master s Thesis Πορεία Ναύλων και Οικονομία: Παράγοντες που επηρεάζουν τους Δείκτες Ναύλων της Ναυτιλίας ΓΙΩΡΚΑΣ ΑΝΤΡΕΑΣ ΕΠΙΒΛΕΠΩΝ ΚΑΘΗΓΗΤΗΣ : ΔΗΜΗΤΡΗΣ ΤΣΟΥΚΝΙΔΗΣ Limassol,
Calculating the propagation delay of coaxial cable
 Your source for quality GNSS Networking Solutions and Design Services! Page 1 of 5 Calculating the propagation delay of coaxial cable The delay of a cable or velocity factor is determined by the dielectric
Your source for quality GNSS Networking Solutions and Design Services! Page 1 of 5 Calculating the propagation delay of coaxial cable The delay of a cable or velocity factor is determined by the dielectric
ΜΕΤΑΠΤΥΧΙΑΚΗ ΕΡΕΥΝΗΤΙΚΗ ΔΙΑΤΡΙΒΗ
 ΓΕΩΠΟΝΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΑΘΗΝΩΝ ΤΜΗΜΑ ΕΠΙΣΤΗΜΗΣ & ΤΕΧΝΟΛΟΓΙΑΣ ΤΡΟΦΙΜΩΝ ΕΡΓΑΣΤΗΡΙΟ ΠΟΙΟΤΙΚΟΥ ΕΛΕΓΧΟΥ & ΥΓΙΕΙΝΗΣ ΤΡΟΦΙΜΩΝ ΚΑΙ ΠΟΤΩΝ Π.Μ.Σ. «ΕΠΙΣΤΗΜΗ & ΤΕΧΝΟΛΟΓΙΑ ΤΡΟΦΙΜΩΝ & ΔΙΑΤΡΟΦΗ ΤΟΥ ΑΝΘΡΩΠΟΥ» Η ΕΠΙΔΡΑΣΗ
ΓΕΩΠΟΝΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΑΘΗΝΩΝ ΤΜΗΜΑ ΕΠΙΣΤΗΜΗΣ & ΤΕΧΝΟΛΟΓΙΑΣ ΤΡΟΦΙΜΩΝ ΕΡΓΑΣΤΗΡΙΟ ΠΟΙΟΤΙΚΟΥ ΕΛΕΓΧΟΥ & ΥΓΙΕΙΝΗΣ ΤΡΟΦΙΜΩΝ ΚΑΙ ΠΟΤΩΝ Π.Μ.Σ. «ΕΠΙΣΤΗΜΗ & ΤΕΧΝΟΛΟΓΙΑ ΤΡΟΦΙΜΩΝ & ΔΙΑΤΡΟΦΗ ΤΟΥ ΑΝΘΡΩΠΟΥ» Η ΕΠΙΔΡΑΣΗ
Επίδραση της Συμβολαιακής Γεωργίας στην Χρηματοοικονομική Διοίκηση των Επιχειρήσεων Τροφίμων. Ιωάννης Γκανάς
 ΓΕΩΠΟΝΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΑΘΗΝΩΝ ΤΜΗΜΑ ΑΓΡΟΤΙΚΗΣ ΟΙΚΟΝΟΜΙΑΣ ΚΑΙ ΑΝΑΠΤΥΞΗΣ ΤΜΗΜΑ ΕΠΙΣΤΗΜΗΣ ΤΡΟΦΙΜΩΝ & ΔΙΑΤΡΟΦΗΣ ΤΟΥ ΑΝΘΡΩΠΟΥ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΟΡΓΑΝΩΣΗ ΚΑΙ ΔΙΟΙΚΗΣΗ ΕΠΙΧΕΙΡΗΣΕΩΝ ΤΡΟΦΙΜΩΝ & ΓΕΩΡΓΙΑΣ
ΓΕΩΠΟΝΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΑΘΗΝΩΝ ΤΜΗΜΑ ΑΓΡΟΤΙΚΗΣ ΟΙΚΟΝΟΜΙΑΣ ΚΑΙ ΑΝΑΠΤΥΞΗΣ ΤΜΗΜΑ ΕΠΙΣΤΗΜΗΣ ΤΡΟΦΙΜΩΝ & ΔΙΑΤΡΟΦΗΣ ΤΟΥ ΑΝΘΡΩΠΟΥ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΟΡΓΑΝΩΣΗ ΚΑΙ ΔΙΟΙΚΗΣΗ ΕΠΙΧΕΙΡΗΣΕΩΝ ΤΡΟΦΙΜΩΝ & ΓΕΩΡΓΙΑΣ
IMES DISCUSSION PAPER SERIES
 IMES DISCUSSION PAPER SERIES Will a Growth Miracle Reduce Debt in Japan? Selahattin mrohorolu and Nao Sudo Discussion Paper No. 2011-E-1 INSTITUTE FOR MONETARY AND ECONOMIC STUDIES BANK OF JAPAN 2-1-1
IMES DISCUSSION PAPER SERIES Will a Growth Miracle Reduce Debt in Japan? Selahattin mrohorolu and Nao Sudo Discussion Paper No. 2011-E-1 INSTITUTE FOR MONETARY AND ECONOMIC STUDIES BANK OF JAPAN 2-1-1
Main source: "Discrete-time systems and computer control" by Α. ΣΚΟΔΡΑΣ ΨΗΦΙΑΚΟΣ ΕΛΕΓΧΟΣ ΔΙΑΛΕΞΗ 4 ΔΙΑΦΑΝΕΙΑ 1
 Main source: "Discrete-time systems and computer control" by Α. ΣΚΟΔΡΑΣ ΨΗΦΙΑΚΟΣ ΕΛΕΓΧΟΣ ΔΙΑΛΕΞΗ 4 ΔΙΑΦΑΝΕΙΑ 1 A Brief History of Sampling Research 1915 - Edmund Taylor Whittaker (1873-1956) devised a
Main source: "Discrete-time systems and computer control" by Α. ΣΚΟΔΡΑΣ ΨΗΦΙΑΚΟΣ ΕΛΕΓΧΟΣ ΔΙΑΛΕΞΗ 4 ΔΙΑΦΑΝΕΙΑ 1 A Brief History of Sampling Research 1915 - Edmund Taylor Whittaker (1873-1956) devised a
Μεταπτυχιακή διατριβή
 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΓΕΩΤΕΧΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΚΑΙ ΔΙΑΧΕΙΡΙΣΗΣΠΕΡΙΒΑΛΛΟΝΤΟΣ Μεταπτυχιακή διατριβή ΟΡΥΚΤΑ ΚΑΥΣΙΜΑ ΣΤΟΝ ΠΑΓΚΟΣΜΙΟ ΧΩΡΟ ΜΙΑ ΕΝΕΡΓΕΙΑΚΗ ΕΠΙΣΚΟΠΗΣΗ ΤΟΥ 2013 ΜΕ ΙΔΙΑΙΤΕΡΗ ΑΝΑΦΟΡΑ
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΓΕΩΤΕΧΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΚΑΙ ΔΙΑΧΕΙΡΙΣΗΣΠΕΡΙΒΑΛΛΟΝΤΟΣ Μεταπτυχιακή διατριβή ΟΡΥΚΤΑ ΚΑΥΣΙΜΑ ΣΤΟΝ ΠΑΓΚΟΣΜΙΟ ΧΩΡΟ ΜΙΑ ΕΝΕΡΓΕΙΑΚΗ ΕΠΙΣΚΟΠΗΣΗ ΤΟΥ 2013 ΜΕ ΙΔΙΑΙΤΕΡΗ ΑΝΑΦΟΡΑ
þÿ ³¹µ¹½ º±¹ ±ÃÆ»µ¹± ÃÄ ÇÎÁ
 Neapolis University HEPHAESTUS Repository School of Economic Sciences and Business http://hephaestus.nup.ac.cy Master Degree Thesis 2014 þÿ ³¹µ¹½ º±¹ ±ÃÆ»µ¹± ÃÄ ÇÎÁ þÿµá³±ã ±Â Äɽ ½ à º ¼µ ɽ : Georgiou,
Neapolis University HEPHAESTUS Repository School of Economic Sciences and Business http://hephaestus.nup.ac.cy Master Degree Thesis 2014 þÿ ³¹µ¹½ º±¹ ±ÃÆ»µ¹± ÃÄ ÇÎÁ þÿµá³±ã ±Â Äɽ ½ à º ¼µ ɽ : Georgiou,
ΔΗΜΙΟΥΡΓΙΑ ΔΥΝΑΜΙΚΟΥ ΜΟΝΤΕΛΟΥ ΑΝΟΔΙΚΗΣ ΠΡΟΣΕΓΓΙΣΗΣ ΓΙΑ ΤΗΝ ΕΚΤΙΜΗΣΗ ΤΗΣ ΖΗΤΗΣΗΣ ΕΝΕΡΓΕΙΑΣ ΣΤΟΝ ΟΙΚΙΣΤΙΚΟ ΤΟΜΕΑ ΤΗΣ ΚΥΠΡΟΥ
 Σχολή Γεωτεχνικών Επιστημών & Διαχείρισης Περιβάλλοντος Μεταπτυχιακή διατριβή ΔΗΜΙΟΥΡΓΙΑ ΔΥΝΑΜΙΚΟΥ ΜΟΝΤΕΛΟΥ ΑΝΟΔΙΚΗΣ ΠΡΟΣΕΓΓΙΣΗΣ ΓΙΑ ΤΗΝ ΕΚΤΙΜΗΣΗ ΤΗΣ ΖΗΤΗΣΗΣ ΕΝΕΡΓΕΙΑΣ ΣΤΟΝ ΟΙΚΙΣΤΙΚΟ ΤΟΜΕΑ ΤΗΣ ΚΥΠΡΟΥ Γαλάτεια
Σχολή Γεωτεχνικών Επιστημών & Διαχείρισης Περιβάλλοντος Μεταπτυχιακή διατριβή ΔΗΜΙΟΥΡΓΙΑ ΔΥΝΑΜΙΚΟΥ ΜΟΝΤΕΛΟΥ ΑΝΟΔΙΚΗΣ ΠΡΟΣΕΓΓΙΣΗΣ ΓΙΑ ΤΗΝ ΕΚΤΙΜΗΣΗ ΤΗΣ ΖΗΤΗΣΗΣ ΕΝΕΡΓΕΙΑΣ ΣΤΟΝ ΟΙΚΙΣΤΙΚΟ ΤΟΜΕΑ ΤΗΣ ΚΥΠΡΟΥ Γαλάτεια
The Simply Typed Lambda Calculus
 Type Inference Instead of writing type annotations, can we use an algorithm to infer what the type annotations should be? That depends on the type system. For simple type systems the answer is yes, and
Type Inference Instead of writing type annotations, can we use an algorithm to infer what the type annotations should be? That depends on the type system. For simple type systems the answer is yes, and
ΚΛΙΜΑΤΙΚΕΣ ΑΛΛΑΓΕΣ IPCC WGII AR5. Summary for Policymakers. WGII AR5 Phase I Report Launch 1 31 March 2014
 ΚΛΙΜΑΤΙΚΕΣ ΑΛΛΑΓΕΣ IPCC WGII AR5 Summary for Policymakers WGII AR5 Phase I Report Launch 1 31 March 2014 Climate Change 2014: Impacts, Adaptation, and Vulnerability SUMMARY FOR POLICYMAKERS ΚΛΙΜΑΤΙΚΕΣ
ΚΛΙΜΑΤΙΚΕΣ ΑΛΛΑΓΕΣ IPCC WGII AR5 Summary for Policymakers WGII AR5 Phase I Report Launch 1 31 March 2014 Climate Change 2014: Impacts, Adaptation, and Vulnerability SUMMARY FOR POLICYMAKERS ΚΛΙΜΑΤΙΚΕΣ
ΠΡΟΣΤΑΣΙΑ ΑΤΜΟΣΦΑΙΡΙΚΗΣ ΔΙΑΒΡΩΣΗΣ ΑΛΟΥΜΙΝΙΟΥ/ΑΝΟΔΙΩΣΗ Al
 Σχολή Μηχανικής και Τεχνολογίας Πτυχιακή εργασία ΠΡΟΣΤΑΣΙΑ ΑΤΜΟΣΦΑΙΡΙΚΗΣ ΔΙΑΒΡΩΣΗΣ ΑΛΟΥΜΙΝΙΟΥ/ΑΝΟΔΙΩΣΗ Al Ανδρέας Παπαχριστοδούλου Λεμεσός, Μάιος 2017 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΜΗΧΑΝΙΚΗΣ ΚΑΙ
Σχολή Μηχανικής και Τεχνολογίας Πτυχιακή εργασία ΠΡΟΣΤΑΣΙΑ ΑΤΜΟΣΦΑΙΡΙΚΗΣ ΔΙΑΒΡΩΣΗΣ ΑΛΟΥΜΙΝΙΟΥ/ΑΝΟΔΙΩΣΗ Al Ανδρέας Παπαχριστοδούλου Λεμεσός, Μάιος 2017 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΜΗΧΑΝΙΚΗΣ ΚΑΙ
Study of In-vehicle Sound Field Creation by Simultaneous Equation Method
 Study of In-vehicle Sound Field Creation by Simultaneous Equation Method Kensaku FUJII Isao WAKABAYASI Tadashi UJINO Shigeki KATO Abstract FUJITSU TEN Limited has developed "TOYOTA remium Sound System"
Study of In-vehicle Sound Field Creation by Simultaneous Equation Method Kensaku FUJII Isao WAKABAYASI Tadashi UJINO Shigeki KATO Abstract FUJITSU TEN Limited has developed "TOYOTA remium Sound System"
ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ
 ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ Μελέτη των υλικών των προετοιμασιών σε υφασμάτινο υπόστρωμα, φορητών έργων τέχνης (17ος-20ος αιώνας). Διερεύνηση της χρήσης της τεχνικής της Ηλεκτρονικής Μικροσκοπίας
ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ Μελέτη των υλικών των προετοιμασιών σε υφασμάτινο υπόστρωμα, φορητών έργων τέχνης (17ος-20ος αιώνας). Διερεύνηση της χρήσης της τεχνικής της Ηλεκτρονικής Μικροσκοπίας
k A = [k, k]( )[a 1, a 2 ] = [ka 1,ka 2 ] 4For the division of two intervals of confidence in R +
[a 1, a 2 ] = [ka 1,ka 2 ] 4For the division of two intervals of confidence in R + k A = [k, k]( )[a 1, a 2 ] = [ka 1,ka 2 ] 4For the division of two intervals of confidence in R +](/thumbs/73/69566903.jpg) Chapter 3. Fuzzy Arithmetic 3- Fuzzy arithmetic: ~Addition(+) and subtraction (-): Let A = [a and B = [b, b in R If x [a and y [b, b than x+y [a +b +b Symbolically,we write A(+)B = [a (+)[b, b = [a +b
Chapter 3. Fuzzy Arithmetic 3- Fuzzy arithmetic: ~Addition(+) and subtraction (-): Let A = [a and B = [b, b in R If x [a and y [b, b than x+y [a +b +b Symbolically,we write A(+)B = [a (+)[b, b = [a +b
