Supporting Information. for. Angew. Chem. Int. Ed. Z Wiley-VCH 2003
|
|
|
- Ευπραξία Κυπραίος
- 7 χρόνια πριν
- Προβολές:
Transcript
1 Supporting Information for Angew. Chem. Int. Ed. Z52857 Wiley-VC Weinheim, Germany
2 Palladium Catalyzed DYKAT of Vinyl Epoxide: Enantioselective Total Synthesis and Assignment of Configuration of (+)-Broussonetine G Barry M. Trost *, Daniel B. orne, Michael J. Woltering Department of Chemistry, Stanford University, Stanford, California, Characterization Data (S)--(1-ydroxymethyl-2-propenyl)phthalimide 1 4 mp C (PE/Et 2 ) [lit. 1 mp 58 C]; [α] D 25 = (c=1.02, C 2 Cl 2 ); R f =0.23 (1:1 PE:Et 2 ); IR (thin film) 3464, 1774, 1710, 1468, 1388, 1360, 1064, 1027, 719 cm -1 ; 1 MR (300 Mz, CDCl 3 ) δ 7.85 (m, 2), 7.74 (m, 2), 6.17 (ddd, J=17.1, 10.5, 6.6 z, 1), 5.29 (d, J=10.5 z, 1), 5.28 (d, J=17.1 z, 1), 4.94 (m, 1), 4.66 (m, 1, ), 4.15 (ddd, J=11.7, 8.7, 7.8 z, 1 ), 3.98 (ddd, J=11.7, 4.5, 3.9 z, 1) ppm; 13 C MR (75 MZ, CDCl 3 ) δ 168.5, 134.0, 131.9, 131.6, 123.3, 118.8, 62.6, 55.9 ppm. Enantiomeric excess was determined to be 98% ee by chiral PLC (Chiralpak D, 90:10 heptane:ipr, 1.0 ml/min, min. R and min. S). 2 (S)-2-Amino-3-buten-1-ol [α] D 25 = (c=1.01, C 2 Cl 2 ); R f =0.25 (Me); IR (thin film) 3354, 2869, 1644, 1591, 1458, 1425, 1361, 1143, 1052, 996, 922, 859, 668 cm -1 ; 1 MR (300 Mz, CDCl 3 ) δ 5.83 (ddd, J=17.4, 10.5, 5.7 z, 1), 5.24 (dd, J=17.1, 0.9 z, 1), 5.16 (dd, J=10.5, 0.9 z, 1), 3.62 (dd, J=10.2, 3.9 z, 1), 3.47 (m, 2), 3.36 (dd, J=10.2, 7.5 z, 1), 1.99 (bs, 2, 2 ) ppm; 13 C MR (75 MZ, CDCl 3 ) δ 139.3, 115.4, 66.2, 55.5 ppm. EA calculated for C : C, 55.15;, 10.41;, Found: C, 55.31;, 10.78;, (S)-4-Vinyl-1,3-oxazolidin-2-one 5 [α] D 25 = (c=1.07, C 2 Cl 2 ); R f =0.50 (20:1 Et 2 :Me); IR (thin film) 3286, 2988, 2914, 1748, 1647, 1538, 1480, 1401, 1349, 1320, 1290, 1236, 1095, 1050, 1024, 992, 935,
3 770, 724 cm -1 ; 1 MR (300 Mz, CDCl 3 ) δ 6.17 (bs, 1, ), 5.83 (ddd, J=17.1, 10.2, 7.2 z, 1), 5.33 (dd, J=17.1, 0.6 z, 1), 5.26 (d, J=10.2 z, 1), 4.55 (dd, J=8.6 z, 1), 4.40 (dd, J=7.5, 0.9 z, 1), 4.07 (dd, J=8.6, 6.5 z, 1) ppm; 13 C MR (75 MZ, CDCl 3 ) δ 159.3, 135.6, 118.8, 69.9, 55.2 ppm; EA calculated for C : C, 53.09;, 6.24;, Found: C, 53.17;, 6.31;, [4S,3(1S)]-3-(1-ydroxymethyl-propenyl)-4-vinyl-oxazolidin-2-one 6 [α] D 25 = (c=1.14, C 2 Cl 2 ); R f =0.50 (20:1 Et 2 :Me); IR (thin film) 3418, 3085, 2985, 2912, 1732, 1644, 1480, 1417, 1336, 1253, 1064, 1030, 992, 936, 764, 706 cm -1 ; 1 MR (300 Mz, CDCl 3 ) δ 5.90 (ddd, J=17.4, 10.2, 7.5 z, 1), 5.74 (ddd, J=17.7, 9.3, 8.7 z, 1), 5.38 (d, J=10.2 z, 1), 5.37 (d, J=16.8 z, 1), 5.29 (d, J=10.2 z, 1), 5.21 (d, J=17.4 z, 1), 4.48 (t, J=8.7 z, 1), 4.32 (dd, J=8.7, 8.1 z, 1), 4.02 (dd, J=8.4, 7.8 z, 1), 3.90 (m, 3), 3.57 (dd, J=8.4, 4.8 z, 1) ppm; 13 C MR (75 MZ, CDCl 3 ) δ 158.5, 134.9, 132.3, 121.7, 118.9, 67.6, 62.8, 59.6, 59.2 ppm; RMS (EI+) m/z calculated for C (M + +): , found Diastereomeric ratio determined by GC of crude product (Varian 3600, 100/ /2; 7.46 min. S,S and 7.69 min. S,R 92.6:7.4 anti:syn). Bn [4S,3(1S)]-3-[1-benzyloxymethyl-allyl]-4-vinyl-oxazolidin-2-one [α] D 25 = -6.2 (c=1.15, C 2 Cl 2 ); R f =0.24 (1:1 PE:Et 2 ); IR (thin film) 2959, 2928, 2885, 1752, 1463, 1335, 1110, 943, 820, 763 cm -1 ; 1 MR (300 Mz, CDCl 3 ) δ 5.82 (ddd, J=17.3, 10.2, 7.5 z, 1), 5.70 (ddd, J=17.6, 8.3, 1.2 z, 1), 5.23 (m. 6), 4.60 (d, J=12.0 z, 1), 4.45 (d, J=12.0 z, 1), (m, 3), (m, 2), 3.62 (dd, J=10.3, 5.4 z, 1) ppm; 13 C MR (125 MZ, CDCl 3 ) δ 156.1, 135.1, 132.9m 123.5, 122.7, 122.4, 120.1, 118.3, 66.5, 65.4, 65.0, 62.1, 59.6 ppm; EA calculated for C : C, 70.31;, 7.01;, Found: C, 70.20;, 6.90;, Bn (3S,6aS)-1,2-Dehydro-3-benzyloxymethyl-5-oxapyrrolizidin-4-one 13 m.p C; [α] D 25 = (c=1.01, C 2 Cl 2 ); R f =0.18 (4:1 PE:Et 2 ); IR (thin film) 2992, 2929, 1746, 1385, 1366, 1222, 1126, 1031, 746 cm -1 ; 1 MR (300 Mz, CDCl 3 ) δ 7.29 (m, 5) 6.00 (d, J=6.1 z, 1), 5.93 (d, J=6.1 z, 1), 4.74 (m, 2), 4.54 (m, 3), 4.19 (dd, J=8.5, 5.1 z, 1), 3.53 (d, J=4.9 z, 2) ppm; 13 C MR (75 MZ, 2
4 CDCl 3 ) δ 158.2, 133.2, 127.4, 125.5, 123.6, 122.9, 122.7, 68.4, 66.4, 64.0, 62.1, 59.7 ppm; EA calculated for C : C, 68.56;, 6.16;, Found: C, 68.71;, 6.02;, Bn (2S,5S)-(5-Benzyloxymethyl-2,5-dihydro-1-pyrrol-2-yl)-methanol [α] D 25 = (c=0.98, C 2 Cl 2 ); R f =0.10 (20:1 C 2 Cl 2 :Me); IR (thin film) 3404, 2930, 2865, 1478, 1392, 1219, 970, 805, 740, 670 cm -1 ; 1 MR (300 Mz, CDCl 3 ) δ 7.32 (m, 5), 5.78 (d, J=6.1 z, 1), 5.74 (d, J=6.1 z, 1), 4.53 (s, 2), 4.21 (bs, 2, -,-), (m, 4), (m, 2) ppm; 13 C MR (75 MZ, CDCl 3 ) δ 133.2, 125.9, 125.8, 123.6, 122.9, 68.4, 68.1, 61.5, 60.1, 59.7 ppm; C : C, 54.19;, 5.85;, Found: C, 53.93;, 6.03;, RMS (EI+) m/z calculated for C (M + +): , found Bn Cbz (2S,5S)-2-Benzyloxymethyl-5-hydroxymethyl-2,5-dihydro-pyrrole-1-carboxylic acid benzyl ester 14 [α] D 25 = (c=1.019, C 2 Cl 2 ); R f =0.56 (20:1 C 2 Cl 2 :Me); IR (thin film) 3428, 2920, 1701, 1454, 1416, 1356, 1318, 1105, 735, 698 cm -1 ; 1 MR (500 Mz, CDCl 3 ) 4:1 mixture of amide rotamers Major: δ (m, 10), 5.88 (dt, J=6.3, 2.1 z, 1), 5.68 (dt, J=6.3, 1.7 z, 1), 5.08 (s, 2), 4.76 (m, 1), 4.66 (m, 1), 4.43 (d, J=12.2 z, 1), 4.38 (d, J=12.3 z, 1), 3.86 (dd, J=12.1, 1.5 z, 1), 3.68 (dd, J=9.4, 2.7 z, 1), 3.65 (dd, J=12.2, 6.3 z, 1), 3.54 (dd, J=9.3, 6.1 z, 1) ppm; Minor: δ (m, 10), 5.98 (d, J=6.5 z, 1), 5.77 (d, J=6.5 z, 1), 5.17 (d, J=12.3 z, 1), 5.11 (d, J=12.3 z, 1), 4.76 (m, 1), 4.66 (m, 1), 4.49 (m, 2), 3.85 (m, 2) ppm; 13 CMR (125 MZ, CDCl 3 ) δ 156.2, 138.4, 136.3, 129.8, 128.9, 128.8, 128.7, 128.6, 128.5, 128.5, 128.4, 128.0, 127.7, 73.6, 73.5, 70.6, 69.7, 69.4, 67.7, 67.3, 67.2, 66.6, 66.0, 65.4, 63.7 ppm; RMS (EI+) m/z calculated for C (M + -C 2 ): , found: Bn Cbz (2S,5S)-5-Benzyloxymethyl-2,5-dihydro-pyrrole-1,2-dicarboxylic acid 1-benzyl ester [α] D 25 = (c=1.094, C 2 Cl 2 ); R f =0.10 (2:1 EtAc:PE); IR (thin film) , 3033, 2925, 1715, 1418, 1357, 1303, 1206, 1109, 1002, 912, 737, 698 cm -1 ; 1 MR (400 Mz, CDCl 3 ) 11:9 mixture of amide rotamers Major: δ 9.90 (bs, 1), (m, 10), 6.04 (m, 1), 5.77 (dt, J=6.4, 2.0 z, 1), (m, 3), 4.88 (m, 1), 4.50 (m, 2), 3.87 (dd, J=9.6, 2.9 z, 1), 3.78 (dd, J=9.6, 5.7 z, 1), 3.65 (dd, J=12.2, 6.3 z, 1), 3.54 (dd, J=9.3, 6.1 z, 1) ppm; Minor: δ 9.90 (bs, 1), (m, 10), 6.04 (m, 1), 5.83 (dt, J=6.3, 1.8 z, 1), (m, 3), 4.78 (m, 1), 4.70 (m, 2), 3.71 (dd, J=9.5, 2.7 z, 1), 3.55 (dd, J=9.6, 6.1 z, 1) ppm; 13 CMR (125 MZ, 3
5 CDCl 3 ) δ 175.4(C), 174.7(C), 154.3(C), 153.7(C), 138.1(C), 137.9(C), 136.0(C), 135.9(C), 132.5(C), 132.5(C), 128.5(C), 128.4(C), 128.3(C), 128.3(C), 128.1(C), 128.0(C), 127.9(C), 127.7(C), 127.7(C), 127.6(C), 127.5(C), 127.4(C), 124.4(C), 124.2(C), 73.3(C 2 ), 73.2(C 2 ), 70.0(C 2 ), 69.0(C 2 ), 67.6(C 2 ), 67.5(C 2 ), 67.2(C), 67.1(C), 65.3(C), 64.4(C) ppm; RMS (EI+) m/z calculated for C (M + +): , found: Bn Cbz Me (2S,5S)-2-Benzyloxymethyl-5-(methoxy-methyl-carbamoyl)-2,5-dihydro-pyrrole-1- carboxylic acid benzyl ester 16 [α] 25 D = (c=7.18, CCl 3 ); R f =0.52 (2:1 EtAc:PE); IR (thin film) 3402, 2969, 2870, 1714, 1694, 1682, 1455, 1392, 1088, 1014, 915, 708 cm -1 ; 1 MR (500 Mz, CDCl 3 ) 11:9 mixture of amide rotamers Major: δ (m, 10), 6.01 (dt, J=6.3, 2.0 z, 1), 5.73 (dt, J=6.3, 2.1 z, 1), 5.37 (m, 1), (m, 2), 4.95 (m, 1), 4.56 (s, 2), 3.90 (dd, J=9.6, 3.1 z, 1), 3.81 (dd, J=9.5, 5.7 z, 1), 3.38 (s, 3), 3.07 (s, 3) ppm; Minor: δ (m, 10), 5.99 (dt, J=6.3, 2.0 z, 1), 5.81 (dt, J=6.3, 2.0 z, 1), 5.51 (m, 1), (m, 2), 4.85 (m, 1), 4.45 (d, J=12.5 z, 2), 4.41 (d, J=12.5 z, 1), 3.81 (s, 3), 3.74 (dd, J=9.5, 2.8 z, 1), 3.59 (dd, J=9.5, 6.0 z, 1), 3.23 (s, 3) ppm; 13 CMR (125 MZ, CDCl 3 ) δ 170.0, 169.7, 154.3, 153.9, 138.2, 138.0, 136.2, 136.1, 131.7, 131.6, 128.4, 128.4, 128.3, 129.2, 128.2, 128.0, 127.9, 127.6, 127.5, 127.4, 124.5, 124.3, 73.3, 73.1, 70.3, 69.4, 67.2, 67.1, 65.7, 65.4, 65.2, 64.4, 61.3, 63.8, 32.3, 32.1 ppm; RMS (EI+) m/z calculated for C (M + +): , found: Bn Cbz (2S,5S)-2-Benzyloxymethyl-5-[5-{(5R,7R)-1,6-dioxaspiro[4.5]dec-7-yl}-pentanoyl]- 2,5-dihydro-pyrrole-1-carboxylic acid benzyl ester [α] D 26 = (c=1.38, CCl 3 ); R f =0.22 (4:1 Et 2 :PE); IR (thin film) 2938, 2868, 1713, 1497, 1455, 1410, 1352, 1105, 1008, 737, 698 cm -1 ; 1 MR (500 Mz, CDCl 3 ) 3:2 mixture of amide rotamers Major: δ (m, 10), 6.04 (dt, J=6.4, 2.0 z, 1), 5.65 (dt, J=6.3, 2.0 z, 1), (m, 3), 4.89 (m, 1), 4.54 (d, J=12.2 z, 1), 4.50 (d, J=12.2 z, 1), (m, 3), 3.73 (dd, J=9.5, 2.9 z, 1), 3.62 (m, 2), 2.25 (ddd, J=17.7, 8.7, 6.4z, 1), 2.10 (ddd, J=17.7, 8.7, 6.4 z, 1), 2.01 (m, 1), (m, 3), (m, 6), (m, 6) ppm; Minor: δ (m, 10), 6.01 (dt, J=6.4, 1.8 z, 1), 5.71 (dt, J=6.4, 1.8 z, 1), 5.00 (m, 3), 4.79 (m, 1), 4.42 (d, J=12.2 z, 1), 4.39 (d, J=12.2 z, 1), (m, 3), 3.61 (m, 3), 2.47 (ddd, J=17.1, 10.5, 6.5 z, 1), 2.33 (ddd, J=17.1, 10.5, 6.5 z, 1), 2.01 (m, 1), (m, 3), (m, 6), (m, 6) ppm; 13 CMR (125 MZ, CDCl 3 ) δ 207.1, 206.5, 154.1, 153.6, 138.2, 138.0, 136.2, 135.9, 132.2, 132.1, 128.5, 4
6 128.4, 128.4, 128.3, 128.2, 128.2, 128.1, 128.1, 127.9, 127.8, 127.7, 127.6, 127.4, 126.5, 125.0, 124.9, 105.7, 74.4, 74.3, 73.3, 73.3, 73.2, 72.0, 70.2, 70.0, 69.9, 69.1, 67.3, 67.1, 66.6, 65.6, 65.4, 64.5, 38.1, 37.8, 36.9, 35.9, 32.8, 30.9, 25.2, 25.1, 23.6, 22.2, 23.1, 20.4 ppm; RMS (EI+) m/z calculated for C (M + +): , found: ; MS (ESI+) [M] + : 548.4; RMS (EI+) m/z calculated for C (M + -C ): , found: , m/z calculated for C (M + -C ): , found: Bn Cbz (2S,5S)-2-Benzyloxymethyl-5-[(1R)-5-{(5R,7R)-1,6-dioxaspiro[4.5]dec-7-yl}-1- hydroxy-pentyl]-2,5-dihydro-pyrrole-1-carboxylic acid benzyl ester 17 [α] D 24 = (c=1.54, CCl 3 ); R f =0.46 (1:1 EtAc:PE); IR (thin film) 3440, 3032, 2936, 1694, 1634, 1455, 1417, 1354, 1328, 1214, 1105, 1009, 958, 916, 826, 736, 698 cm -1 ; 1 MR (500 Mz, CDCl 3 ) 4:1 mixture of amide rotamers Major: δ (m, 10), 5.88 (dt, J=6.4, 2.0 z, 1), 5.70 (dt, J=6.4, 1.8 z, 1), 5.10 (d, J=12.2 z, 1), 5.06 (d, J=12.2 z, 1), 4.87 (m, 1), 4.63 (m, 1), 4.42 (d, J=12.2, z, 1), 4.37 (d, J=12.2 z, 1), 4.12 (d, J=9.5 z, 1), 3.85 (m, 3), 3.69 (m, 2), 3.56 (dd, J=9.3, 6.0 z, 1), 2.03 (m, 1), (m, 8), (m, 9) ppm; Minor: δ (m, 10), 6.02 (d, J=6.5 z, 1), 5.73 (d, J=6.5 z, 1), 5.19 (d, J=12.2 z, 1), 5.12 (d, J=12.2 z, 1), 4.74 (m, 1), 4.63 (m, 1), 4.50 (s, 2), 4.04 (m, 1), 3.85 (m, 3), 3.69 (m, 3), 2.03 (m, 1), (m, 17) ppm; 13 CMR (125 MZ, CDCl 3 ) δ 155.7, 138.1, 136.1, 129.5, 128.5, 128.4, 128.3, 128.2, 128.1, 128.0, 127.6, 127.5, 127.4, 105.8, 73.1, 72.6, 72.1, 70.2, 67.4, 66.6, 65.3, 37.8, 36.3, 32.9, 31.3, 30.9, 29.7, 26.4, 25.7, 23.7, 20.5 ppm; MS (ESI+) [M] + : 550.4, [M+a] ; RMS (EI+) m/z calculated for C (M + -Bn): , found: The absolute stereochemistry of the secondary alcohol was determined by formation of the (R)- and (S)--methylmandelates and analysis of their 1 MR spectra 2, selected data used in the analysis is shown below: δ = δr-δs Ph Ph Ph Me
7 Bn Cbz (2R,3R,4R,5R)-2-Benzyloxymethyl-5-[(1R)-5-{(5R,7R)-1,6-dioxaspiro[4.5]dec-7-yl}- 1-hydroxy-pentyl]-3,4-dihydroxy-pyrrolidine-1-carboxylic acid benzyl ester 18 [α] D 24 = (c=0.70, CCl 3 ); R f =0.39 (2:1 EtAc:PE); IR (thin film) 3374, 2930, 1700, 1455, 1412, 1348, 1213, 1084, 1010, 736, 698 cm -1 ; 1 MR (500 Mz, CDCl 3 ) 1.3:1 mixture of amide rotamers Major: δ (m, 10), 5.13 (m, 1), 5.06 (d, J=12.1 z, 1), 4.98 (d, J=12.2 z, 1), 4.78 (d, J=12.1 z, 1), 4.60 (m, 1), 4.44 (d, J=12.0 z, 1), 4.36 (d, J=11.8, z, 1), 4.13 (bt, J=5.7 z, 1), 4.06 (d, J=12.0 z, 1), 4.00 (m, 1), 3.87 (m, 4), 3.69 (m, 1), 3.51 (d, J=8.6 z, 1), 2.73 (bs, 1), 2.03 (m, 1), 1.85 (m, 2), (m, 13) ppm; Minor: δ (m, 10), 5.17 (d, J=12.4 z, 1), 5.13 (m, 1), 5.11 (d, J=12.4 z, 1), 4.84 (d, J=12.1 z, 1), 4.55 (d, J=12.0 z, 1), 4.49 (d, J=12.0 z, 1), 4.30 (dd, J=9.8, 2.8 z, 1), 4.05 (d, J=11.8 z, 1), 4.00 (m, 2), 3.87 (m, 4), 3.69 (m, 1), 3.57 (d, J=9.8 z, 1), 2.03 (m, 1), 1.85 (m, 2), (m, 13) ppm; 13 CMR (125 MZ, CDCl 3 ) δ 154.5, 154.3, 136.5, 136.4, 136.3, 136.2, 128.7, 128.6, 128.6, 128.6, 128.3, 128.2, 128.0, 127.9, 127.9, 105.9, 80.5, 79.6, 76.5, 75.8, 73.8, 73.6, 73.4, 72.7, 71.2, 70.1, 69.4, 69.0, 68.3, 67.6, 66.9, 66.7, 37.8, 35.9, 35.8, 34.4, 34.2, 32.8, 30.9, 29.7, 25.9, 25.7, 25.5, 25.3, 23.6, 20.4 ppm; RMS (EI+) m/z calculated for C (M + -CBn): , found: (+)-Broussonetine G 1 [α] D 25 = 19.3 (c=1.01, Me); R f =0.80 (5:1:1 CCl 3 :Me:56% aq. 4 ); IR (thin film 3356, 2937, 1683, 1458, 1202 cm -1 ; 1 MR (500 Mz, pyridine-d 5 ) δ 6.95 (bs, 1, -), 6.78 (bs, 1, -), 6.30 (bs, 1, -), 6.01 (bs, 1, -), 4.95 (t, J=6.4 z, 1), 4.72 (t, J=6.4 z, 1), 4.28 (dd, J=10.8, 5.5 z, 1), 4.24 (dd, J=10.8, 3.8 z, 1), 4.11 (ddd, J=6.4, 4.8, 4.8 z, 1), 3.83 (m, 4), 3.64 (dd, J=6.5, 4.8 z, 1), (m, 6), (m, 7), (m, 4), 1.13 (m, 1) ppm; 13 CMR (125 MZ, pyridine-d 5 ) δ 105.9, 80.6, 80.4, 74.1, 70.3, 67.6, 66.8, 65.9, 63.5, 38.2, 36.7, 35.1, 33.4, 31.4, 26.9, 26.3, 24.1, 21.0 ppm. RMS (EI+) m/z calculated for C (M + ): , found:
8 1-ydroxy-dec-6-yn-5-one 3 R f =0.44 (1:1 EtAc:PE); 1 MR (300 Mz, CDCl 3 ) δ 3.65 (m, 2), 2.60 (t, J=7.2 z, 2), 2.35 (t, J=7.2 z, 2), 1.76 (m, 2), (m, 5), 1.05 (t, J=7.2 z, 3) ppm; 13 CMR (125 MZ, CDCl 3 ) δ 172.8, 94.4, 80.9, 62.2, 45.0, 31.8, 21.2, 20.8, 20.1, 13.4 ppm. Tr 1-Trityloxy-dec-6-yn-5-one 8 R f =0.53 (4:1 PE:Et 2 ); IR (thin film) 3059, 3032, 2961, 2872, 2212, 1674, 1491, 1449, 1223, 1161, 1073, 1034, 899, 762, 705, 648, 633 cm -1 ; 1 MR (200 Mz, CDCl 3 ) δ 7.44 (m, 6), 7.34 (m, 9), 3.07 (t, J=6.2 z, 2), 2.51 (t, J=6.5 z, 2), 2.33 (t, J=7.0 z, 2), (m, 6), 1.00 (t, J=7.3 z, 3) ppm; 13 CMR (75 MZ, CDCl 3 ) δ 183.5, 139.6, 123.9, 122.9, 122.0, 89.4, 81.6, 76.2, 58.2, 40.5, 24.5, 16.5, 16.3, 16.1 ppm; EA calculated for C : C, 84.84;, Found: C, 84.88;, Tr (5R)-1-Trityloxy-dec-6-yn-5-ol 9 [α] D 24 = -2.2 (c=2.63, C 2 Cl 2 ); R f =0.17 (4:1 PE:Et 2 ); IR (thin film) 3405, 3087, 2936, 2870, 1958, 1817, 1597, 1491, 1449, 1381, 1221, 1077, 1033, 900, 746, 699, 633 cm -1 ; 1 MR (300 Mz, CDCl 3 ) δ 7.43 (m, 6), (m, 9), 4.33 (bs, 1), 3.07 (t, J=6.4 z, 2), 2.16 (dt, J=2.0, 7.1 z, 2), 1.71 (d, J=3.4 z, 1, -), (m, 8), 0.95 (t, J=7.3 z, 3) ppm; 13 CMR (75 MZ, CDCl 3 ) δ 139.7, 123.9, 122.9, 122.0, 81.6, 80.7, 76.6, 58.6, 57.9, 33.2, 24.9, 17.3, 17.2, 15.9, 8.7 ppm; RMS (EI+) m/z calculated for C (M + -): , found: Enantiomeric excess was determined to be 97% ee by chiral PLC (Chiralpak D, 97:3 heptane:i- Pr, 1.0 ml/min, 9.43 R and S). Tr (5S)-1-Trityloxy-dec-9-yn-5-ol [α] 23 D = -1.4 (c=1.27, C 2 Cl 2 ); R f =0.40 (1:1 PE:Et 2 ); IR (thin film) 3584, 3388, 3058, 2937, 2867, 2116, 1959, 1821, 1597, 1490, 1449, 1266, 1074, 1032, 900, 764, 740, 707, 648, 603 cm -1 ; 1 MR (300 Mz, CDCl 3 ) δ 7.43 (m, 6), (m, 9), 3.59 (bs, 1), 3.07 (t, J=6.4 z, 2), 2.21 (dt, J=3.6, 6.3 z, 2), 1.94 (t, J=2.5 z, 1), s(m, 11) ppm; 13 CMR (75 MZ, CDCl 3 ) δ 139.7, 123.9, 122.9, 122.1, 81.6, 79.6, 66.5, 58.6, 32.6, 31.5, 25.2, 19.8, 17.5, 13.6 ppm; RMS (EI+) m/z calculated for C (M + ): , found: TP Tr (5S)-1-Trityloxy-5-(Tetrahydro-pyran-2-yloxy)-dec-9-yne 7
9 [α] D 28 = -1.7 (c=4.80, C 2 Cl 2 ); R f =0.75 (1:1 PE:Et 2 ); IR (thin film) 3305, 3058, 2940, 2868, 1597, 1491, 1449, 1200, 1154, 1076, 1032, 1002, 902, 774, 707 cm -1 ; 1 MR (500 Mz, CDCl 3 ) Splitting complicated by mixture of TP diastereomers δ 7.44 (m, 6), 7.27 (m, 6), 7.20 (m, 3) 4.62 (m, 1), 3.87 (m, 1), 3.62 (m, 1), 3.44 (m, 1), 3.06 (m, 2), 2.19 (m, 2), 1.90 (m, 1), 1.80 (m, 1), (m, 15) ppm; 13 CMR (75 MZ, CDCl 3 ) Double peaks due to TP diastereomers δ 144.8, 144.7, 129.0, 128.0, 127.9, 127.1, 97.9, 97.7, 86.6, 86.5, 84.9, 84.6, 76.3, 76.2, 68.8, 68.6, 63.6, 63.2, 62.9, 35.1, 34.3, 33.8, 32.7, 31.5, 31.4, 30.5, 30.4, 25.8, 24.8, 24.2, 22.9, 22.7, 22.1, 20.4, 20.2, 18.9, 18.8 ppm; RMS (EI+) m/z calculated for C (M + -Ph): , found: ; calculated for C (M + -C 5 9 [TP]): , found: EA calculated for C : C, 82.22;, Found: C, 81.98;, TP Tr (5S)-1-Trityloxy-5-(tetrahydro-pyran-2-yloxy)-dodec-9-yn-12-ol [α] 26 D = -1.1 (c=1.16, C 2 Cl 2 ); R f =0.27 (1:1 PE:Et 2 ); IR (thin film) 3442, 3059, 2941, 2868, 1490, 1449, 1200, 1133, 1113, 1076, 1032, 1002, 909, 733, 707 cm -1 ; 1 MR (200 Mz, CDCl 3 ) Splitting complicated by mixture of TP diastereomers δ 7.44 (m, 6), (m, 9), 4.63 (m, 1), 3.87 (m, 1), 3.63 (m, 3), 3.49 (m, 1), 3.06 (t, J=6.2 z, 2), 2.39 (m, 2), 2.17 (m, 2), 2.04 (bs, 1), (m, 16) ppm; 13 CMR (50 MZ, CDCl 3 ) Double peaks due to TP diastereomers δ 142.3, 142.2, 126.4, 125.4, 124.5, 95.6, 94.8, 83.9, 79.8, 73.9, 73.5, 67.8, 61.1, 60.9, 60.5, 60.1, 59.0, 32.4, 31.7, 31.1, 30.1, 28.8, 28.7, 27.7, 23.1, 22.5, 21.9, 20.7, 20.0, 19.4, 17.6, 17.3, 16.5, 16.4 ppm; MS (+ESI) m/z calculated for C a 4 (M + +a): , found: EA calculated for C : C, 79.96;, Found: C, 79.86;, (5S)-Dodec-9-yne-1,5,12-triol 10 [α] 29 D = -5.7 (c=0.42, Me); R f =0.20 (10:1 C 2 Cl 2 :Me); IR (thin film) 3333, 2937, 1457, 1434, 1374, 1335, 1048 cm -1 ; 1 MR (500 Mz, CDCl 3 ) δ 3.68 (m, 5), 2.43 (m, 2), 2.21 (m, 3), 1.73 (bs, 2), (m, 10) ppm; 13 CMR (75 MZ, CDCl 3 ) δ 81.7, 76.8, 70.7, 62.1, 60.8, 36.5, 36.0, 32.0, 24.5, 22.7, 21.4, 18.3 ppm; EA calculated for C : C, 67.26;, Found: C, 67.16;, [(5R,7R)-1,6-Dioxaspiro[4.5]dec-7-yl]-butan-1-ol [α] 24 D = 76.2 (c=1.24, Me); R f =0.51 (10:1 C 2 Cl 2 :Me); IR (thin film) 3406, 2937, 1457, 1386, 1310, 1271, 1215, 1161, 1115, 1008, 960, 918 cm -1 ; 1 MR (500 Mz, CDCl 3 ) δ 3.88 (m, 2), 3.71 (m, 1), 3.65 (t, J=7.0 z, 2), 2.04 (m, 1), (m, 3), (m, 12) 1.20 (ddd, J=16.5, 13.0, 3.5 z, 1) ppm; 13 CMR (75 MZ, CDCl 3 ) δ 105.8, 70.1, 66.6, 37.7, 35.7, 32.7, 32.5, 30.8, 23.6, 21.7, 20.3 ppm; RMS 8
10 (EI+) m/z calculated for C (M + -): , found: Diastereomeric excess determined by GC of crude product (P 5973 GC/MS, 100/ /2; 7.46 min. R,R and 7.69 min. S,R 97:3). Br (5R,7S)-7-(4-Bromo-butyl)-1,6-dioxaspiro[4.5]decane 11 [α] 23 D = 64.3 (c=1.250, CCl 3 ); R f =0.81 (4:1 Et 2 :PE); IR (thin film) 2938, 2870, 1457, 1439, 1216, 1081, 1041 cm -1 ; 1 MR (300 Mz, CDCl 3 ) δ 3.88 (m, 2), 3.70 (m, 1), 3.41 (t, J=6.8z, 2), 2.04 (m, 1), (m, 5), (m, 4), (m, 5), 1.21 (ddd, J=16.5, 13.0, 3.5 z, 1) ppm; 13 CMR (125 MZ, CDCl 3 ) δ 105.8, 69.8, 66.8, 37.8, 35.2, 34.0, 32.8, 32.7, 30.9, 24.3, 23.7, 20.4 ppm; RMS (EI+) m/z calculated for C Br 2 (M + +): , found: Supporting Figure 1. RTEP representation of 13. See Cambridge Crystallographic Data Centre (CCDC 21668) for the corresponding data. Refrences: [1] Trost, B.M.; Bunt, R.C.; Lemoine, R.; Calkins, T.L. J. Am. Chem. Soc. 2000, 122, [2] Trost, B.M.; Belletire, J.L.; Godleski, S.; McDougal, P.G.; Balkovec, J.M.; Baldwin, J.J.; Christy, M.E.; Ponticello, G.S.; Varga, S.L.; Springer, J.P. J. rg. Chem., 1985, 51, [3] Wedler, C.; Schick,. J. Prakt. Chem./Chem.-Ztg , 335(5),
Divergent synthesis of various iminocyclitols from D-ribose
 Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 205 Divergent synthesis of various iminocyclitols from D-ribose Ramu Petakamsetty,
Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 205 Divergent synthesis of various iminocyclitols from D-ribose Ramu Petakamsetty,
First Total Synthesis of Antimitotic Compound, (+)-Phomopsidin
 First Total Synthesis of Antimitotic Compound, (+)-Phomopsidin Takahiro Suzuki, a Kenji Usui, a Yoshiharu Miyake, a Michio Namikoshi, b and Masahisa Nakada a, * a Department of Chemistry, School of Science
First Total Synthesis of Antimitotic Compound, (+)-Phomopsidin Takahiro Suzuki, a Kenji Usui, a Yoshiharu Miyake, a Michio Namikoshi, b and Masahisa Nakada a, * a Department of Chemistry, School of Science
Supporting Information
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Synthesis of 3-omosubstituted Pyrroles via Palladium- Catalyzed Intermolecular Oxidative Cyclization
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Synthesis of 3-omosubstituted Pyrroles via Palladium- Catalyzed Intermolecular Oxidative Cyclization
Supporting Information. Copyright Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2006
 Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 A new route to enantiopure b-aryl-substituted b-amino acids and 4-aryl-substituted b-lactams through lipase-catalyzed
Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 A new route to enantiopure b-aryl-substituted b-amino acids and 4-aryl-substituted b-lactams through lipase-catalyzed
Supporting Information. Asymmetric Binary-acid Catalysis with Chiral. Phosphoric Acid and MgF 2 : Catalytic
 Supporting Information Asymmetric Binary-acid Catalysis with Chiral Phosphoric Acid and MgF 2 : Catalytic Enantioselective Friedel-Crafts Reactions of β,γ- Unsaturated-α-Ketoesters Jian Lv, Xin Li, Long
Supporting Information Asymmetric Binary-acid Catalysis with Chiral Phosphoric Acid and MgF 2 : Catalytic Enantioselective Friedel-Crafts Reactions of β,γ- Unsaturated-α-Ketoesters Jian Lv, Xin Li, Long
Phosphorus Oxychloride as an Efficient Coupling Reagent for the Synthesis of Ester, Amide and Peptide under Mild Conditions
 Supplementary Information for Phosphorus xychloride as an Efficient Coupling Reagent for the Synthesis of Ester, Amide and Peptide under Mild Conditions u Chen,* a,b Xunfu Xu, a Liu Liu, a Guo Tang,* a
Supplementary Information for Phosphorus xychloride as an Efficient Coupling Reagent for the Synthesis of Ester, Amide and Peptide under Mild Conditions u Chen,* a,b Xunfu Xu, a Liu Liu, a Guo Tang,* a
Room Temperature Highly Diastereoselective Zn-Mediated. Allylation of Chiral N-tert-Butanesulfinyl Imines: Remarkable Reaction Condition Controlled
 Supporting Information for: Room Temperature Highly Diastereoselective Zn-Mediated Allylation of Chiral N-tert-Butanesulfinyl Imines: Remarkable Reaction Condition Controlled Stereoselectivity Reversal
Supporting Information for: Room Temperature Highly Diastereoselective Zn-Mediated Allylation of Chiral N-tert-Butanesulfinyl Imines: Remarkable Reaction Condition Controlled Stereoselectivity Reversal
Mandelamide-Zinc Catalyzed Alkyne Addition to Heteroaromatic Aldehydes
 1 Mandelamide-Zinc Catalyzed Alkyne Addition to Heteroaromatic Aldehydes Gonzalo Blay, Isabel Fernández, Alícia Marco-Aleixandre, and José R. Pedro Departament de Química Orgànica, Facultat de Química,
1 Mandelamide-Zinc Catalyzed Alkyne Addition to Heteroaromatic Aldehydes Gonzalo Blay, Isabel Fernández, Alícia Marco-Aleixandre, and José R. Pedro Departament de Química Orgànica, Facultat de Química,
Supporting Information One-Pot Approach to Chiral Chromenes via Enantioselective Organocatalytic Domino Oxa-Michael-Aldol Reaction
 Supporting Information ne-pot Approach to Chiral Chromenes via Enantioselective rganocatalytic Domino xa-michael-aldol Reaction Hao Li, Jian Wang, Timiyin E-Nunu, Liansuo Zu, Wei Jiang, Shaohua Wei, *
Supporting Information ne-pot Approach to Chiral Chromenes via Enantioselective rganocatalytic Domino xa-michael-aldol Reaction Hao Li, Jian Wang, Timiyin E-Nunu, Liansuo Zu, Wei Jiang, Shaohua Wei, *
Protease-catalysed Direct Asymmetric Mannich Reaction in Organic Solvent
 Supplementary information for the paper Protease-catalysed Direct Asymmetric Mannich Reaction in Organic Solvent Yang Xue, Ling-Po Li, Yan-Hong He * & Zhi Guan * School of Chemistry and Chemical Engineering,
Supplementary information for the paper Protease-catalysed Direct Asymmetric Mannich Reaction in Organic Solvent Yang Xue, Ling-Po Li, Yan-Hong He * & Zhi Guan * School of Chemistry and Chemical Engineering,
Copper-Catalyzed Oxidative Dehydrogenative N-N Bond. Formation for the Synthesis of N,N -Diarylindazol-3-ones
 Electronic Supplementary Material (ESI) for Organic Chemistry Frontiers. This journal is the Partner Organisations 2016 Supporting information Copper-Catalyzed Oxidative Dehydrogenative - Bond Formation
Electronic Supplementary Material (ESI) for Organic Chemistry Frontiers. This journal is the Partner Organisations 2016 Supporting information Copper-Catalyzed Oxidative Dehydrogenative - Bond Formation
Efficient and Simple Zinc mediated Synthesis of 3 Amidoindoles
 Electronic Supplementary Material (ESI) for rganic and Biomolecular Chemistry SUPPRTIG IFRMATI Efficient and Simple Zinc mediated Synthesis of 3 Amidoindoles Anahit Pews-Davtyan and Matthias Beller* Leibniz-Institut
Electronic Supplementary Material (ESI) for rganic and Biomolecular Chemistry SUPPRTIG IFRMATI Efficient and Simple Zinc mediated Synthesis of 3 Amidoindoles Anahit Pews-Davtyan and Matthias Beller* Leibniz-Institut
Synthesis of novel 1,2,3-triazolyl derivatives of pregnane, androstane and D-homoandrostane. Tandem Click reaction/cu-catalyzed D-homo rearrangement
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 Supporting Information Synthesis of novel 1,2,3-triazolyl derivatives of
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 Supporting Information Synthesis of novel 1,2,3-triazolyl derivatives of
Supporting Information. Experimental section
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Experimental section General. Proton nuclear magnetic resonance ( 1
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Experimental section General. Proton nuclear magnetic resonance ( 1
Supporting Information. Synthesis and biological evaluation of nojirimycin- and
 Supporting Information for Synthesis and biological evaluation of nojirimycin- and pyrrolidine-based trehalase inhibitors Davide Bini 1, Francesca Cardona 2, Matilde Forcella 1, Camilla Parmeggiani 2,3,
Supporting Information for Synthesis and biological evaluation of nojirimycin- and pyrrolidine-based trehalase inhibitors Davide Bini 1, Francesca Cardona 2, Matilde Forcella 1, Camilla Parmeggiani 2,3,
Chiral Phosphoric acid Catalyzed Enantioselective N- Alkylation of Indoles with in situ Generated Cyclic N-Acyl Ketimines
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 Chiral osphoric acid Catalyzed Enantioselective - Alkylation of Indoles with in situ Generated
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 Chiral osphoric acid Catalyzed Enantioselective - Alkylation of Indoles with in situ Generated
Supplementary Figure S1. Single X-ray structure 3a at probability ellipsoids of 20%.
 Supplementary Figure S1. Single X-ray structure 3a at probability ellipsoids of 20%. S1 Supplementary Figure S2. Single X-ray structure 5a at probability ellipsoids of 20%. S2 H 15 Ph Ac Ac I AcH Ph Ac
Supplementary Figure S1. Single X-ray structure 3a at probability ellipsoids of 20%. S1 Supplementary Figure S2. Single X-ray structure 5a at probability ellipsoids of 20%. S2 H 15 Ph Ac Ac I AcH Ph Ac
Supplement: Intramolecular N to N acyl migration in conformationally mobile 1 -acyl-1- systems promoted by debenzylation conditions (HCOONH 4
 Cent. Eur. J. Chem. 9(5) 2011 S164-S175 DI: 10.2478/s11532-011-0082-y Central European Journal of Chemistry Supplement: Intramolecular to acyl migration in conformationally mobile 1 -acyl-1- benzyl-3,4
Cent. Eur. J. Chem. 9(5) 2011 S164-S175 DI: 10.2478/s11532-011-0082-y Central European Journal of Chemistry Supplement: Intramolecular to acyl migration in conformationally mobile 1 -acyl-1- benzyl-3,4
The Free Internet Journal for Organic Chemistry
 The Free Internet Journal for Organic Chemistry Paper Archive for Organic Chemistry Arkivoc 2018, part iii, S1-S6 Synthesis of dihydropyranones and dihydropyrano[2,3- d][1,3]dioxine-diones by cyclization
The Free Internet Journal for Organic Chemistry Paper Archive for Organic Chemistry Arkivoc 2018, part iii, S1-S6 Synthesis of dihydropyranones and dihydropyrano[2,3- d][1,3]dioxine-diones by cyclization
Supporting Information
 Supporting Information for AgOTf-catalyzed one-pot reactions of 2-alkynylbenzaldoximes with α,β-unsaturated carbonyl compounds Qiuping Ding 1, Dan Wang 1, Puying Luo* 2, Meiling Liu 1, Shouzhi Pu* 3 and
Supporting Information for AgOTf-catalyzed one-pot reactions of 2-alkynylbenzaldoximes with α,β-unsaturated carbonyl compounds Qiuping Ding 1, Dan Wang 1, Puying Luo* 2, Meiling Liu 1, Shouzhi Pu* 3 and
Aluminium triflate as a Lewis acid catalyst for the ring opening of epoxides in alcohols
 Aluminium triflate as a Lewis acid catalyst for the ring opening of epoxides in alcohols D. Bradley G. Williams* and Michelle Lawton a Department of Chemistry, University of Johannesburg, P.O. Box 524,
Aluminium triflate as a Lewis acid catalyst for the ring opening of epoxides in alcohols D. Bradley G. Williams* and Michelle Lawton a Department of Chemistry, University of Johannesburg, P.O. Box 524,
Lewis Acid Catalyzed Propargylation of Arenes with O-Propargyl Trichloroacetimidate: Synthesis of 1,3-Diarylpropynes
 Supporting Information for Lewis Acid Catalyzed Propargylation of Arenes with O-Propargyl Trichloroacetimidate: Synthesis of 1,3-Diarylpropynes Changkun Li and Jianbo Wang* Beijing National Laboratory
Supporting Information for Lewis Acid Catalyzed Propargylation of Arenes with O-Propargyl Trichloroacetimidate: Synthesis of 1,3-Diarylpropynes Changkun Li and Jianbo Wang* Beijing National Laboratory
SUPPLEMENTARY INFORMATION
 SUPPLEMETARY IFRMATI DI: 10.1038/CEM.1406 ighly enantioselective trapping of zwitterionic intermediates by imines uang Qiu,a, Ming Li,a, Li-Qin Jiang a, Feng-Ping Lv a, Li Zan a, Chang-Wei Zhai a, Michael.
SUPPLEMETARY IFRMATI DI: 10.1038/CEM.1406 ighly enantioselective trapping of zwitterionic intermediates by imines uang Qiu,a, Ming Li,a, Li-Qin Jiang a, Feng-Ping Lv a, Li Zan a, Chang-Wei Zhai a, Michael.
Supporting Information
 Supporting Information for ovel FR-900493 Analogs that Inhibit utgrowth of Clostridium difficile Spores Katsuhiko Mitachi, a yun Gi Yun, b Sara M. Kurosu, a Shakiba Eslamimehr, a Maddie R. Lemieux, a Lada
Supporting Information for ovel FR-900493 Analogs that Inhibit utgrowth of Clostridium difficile Spores Katsuhiko Mitachi, a yun Gi Yun, b Sara M. Kurosu, a Shakiba Eslamimehr, a Maddie R. Lemieux, a Lada
Transfer of axial chirality through the nickel-catalyzed hydrocyanation of chiral allenes
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 7 Electronic supplementary information for Transfer of axial chirality through
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 7 Electronic supplementary information for Transfer of axial chirality through
Metal-free Oxidative Coupling of Amines with Sodium Sulfinates: A Mild Access to Sulfonamides
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting information for Metal-free Oxidative Coupling of Amines with Sodium Sulfinates:
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting information for Metal-free Oxidative Coupling of Amines with Sodium Sulfinates:
Cu(I)-Catalyzed Asymmetric Multicomponent Cascade Inverse. Electron-Demand aza-diels-alder/nucleophilic Addition/Ring-Opening
 Cu(I)-Catalyzed Asymmetric Multicomponent Cascade Inverse Electron-Demand aza-diels-alder/nucleophilic Addition/Ring-Opening Reaction Involving 2-Methoxyfurans as Efficient Dienophiles Rong Huang, Xin
Cu(I)-Catalyzed Asymmetric Multicomponent Cascade Inverse Electron-Demand aza-diels-alder/nucleophilic Addition/Ring-Opening Reaction Involving 2-Methoxyfurans as Efficient Dienophiles Rong Huang, Xin
Supporting Information for
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2015 Supporting Information for Quinine-Catalyzed Highly Enantioselective Cycloannulation
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2015 Supporting Information for Quinine-Catalyzed Highly Enantioselective Cycloannulation
Supporting Information
 Supporting Information Wiley-VC 007 9 Weinheim, Germany ew ear Infrared Dyes and Fluorophores Based on Diketopyrrolopyrroles Dipl.-Chem. Georg M. Fischer, Dipl.-Chem. Andreas P. Ehlers, Prof. Dr. Andreas
Supporting Information Wiley-VC 007 9 Weinheim, Germany ew ear Infrared Dyes and Fluorophores Based on Diketopyrrolopyrroles Dipl.-Chem. Georg M. Fischer, Dipl.-Chem. Andreas P. Ehlers, Prof. Dr. Andreas
Copper-catalyzed formal O-H insertion reaction of α-diazo-1,3-dicarb- onyl compounds to carboxylic acids with the assistance of isocyanide
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Copper-catalyzed formal O-H insertion reaction of α-diazo-1,3-dicarb- onyl compounds to carboxylic
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Copper-catalyzed formal O-H insertion reaction of α-diazo-1,3-dicarb- onyl compounds to carboxylic
Supporting Information
 Supporting Information Lewis acid catalyzed ring-opening reactions of methylenecyclopropanes with diphenylphosphine oxide in the presence of sulfur or selenium Min Shi,* Min Jiang and Le-Ping Liu State
Supporting Information Lewis acid catalyzed ring-opening reactions of methylenecyclopropanes with diphenylphosphine oxide in the presence of sulfur or selenium Min Shi,* Min Jiang and Le-Ping Liu State
Supporting Information for Iron-catalyzed decarboxylative alkenylation of cycloalkanes with arylvinylic carboxylic acids via a radical process
 Supporting Information for Iron-catalyzed decarboxylative alkenylation of cycloalkanes with arylvinylic carboxylic acids via a radical process Jincan Zhao 1, Hong Fang 1, Jianlin Han* 1,2 and Yi Pan* 1
Supporting Information for Iron-catalyzed decarboxylative alkenylation of cycloalkanes with arylvinylic carboxylic acids via a radical process Jincan Zhao 1, Hong Fang 1, Jianlin Han* 1,2 and Yi Pan* 1
Supporting Information Table of Contents 1. General remarks...s1 2. Synthesis of catalyst L S1
 Diastereo- and Enantioselective Conjugate Addition of 3-Substituted xindoles to itroolefins Catalyzed by a Chiral i(ac) 2 -Diamine Complex under Mild Conditions Yan-Yan Han,, Zhi-Jun Wu, Wen-Bing Chen,,
Diastereo- and Enantioselective Conjugate Addition of 3-Substituted xindoles to itroolefins Catalyzed by a Chiral i(ac) 2 -Diamine Complex under Mild Conditions Yan-Yan Han,, Zhi-Jun Wu, Wen-Bing Chen,,
Supporting Information. Consecutive hydrazino-ugi-azide reactions: synthesis of acylhydrazines bearing 1,5- disubstituted tetrazoles
 Supporting Information for Consecutive hydrazino-ugi-azide reactions: synthesis of acylhydrazines bearing 1,5- disubstituted tetrazoles Angélica de Fátima S. Barreto*, Veronica Alves dos Santos, and Carlos
Supporting Information for Consecutive hydrazino-ugi-azide reactions: synthesis of acylhydrazines bearing 1,5- disubstituted tetrazoles Angélica de Fátima S. Barreto*, Veronica Alves dos Santos, and Carlos
Supporting Information. Copyright Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2006
 Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 1 A Facile Way to Synthesize 2H-Chromenes: Reconsideration of the Reaction Mechanism between Salicylic Aldehyde and
Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 1 A Facile Way to Synthesize 2H-Chromenes: Reconsideration of the Reaction Mechanism between Salicylic Aldehyde and
A Dinuclear Zinc Catalyzed Asymmetric Alkynylation of Unsaturated Aldehydes
 A Dinuclear Zinc Catalyzed Asymmetric Alkynylation of Unsaturated Aldehydes Barry M. Trost,* Andrew H. Weiss, Axel Jacobi von Wangelin Department of Chemistry, Stanford University, Stanford, California
A Dinuclear Zinc Catalyzed Asymmetric Alkynylation of Unsaturated Aldehydes Barry M. Trost,* Andrew H. Weiss, Axel Jacobi von Wangelin Department of Chemistry, Stanford University, Stanford, California
Supporting Information. for. A novel application of 2-silylated 1,3-dithiolanes for the. synthesis of aryl/hetaryl-substituted ethenes and
 Supporting Information for A novel application of 2-silylated 1,3-dithiolanes for the synthesis of aryl/hetaryl-substituted ethenes and dibenzofulvenes Grzegorz Mlostoń *1, Paulina Pipiak 1, Róża Hamera-Fałdyga
Supporting Information for A novel application of 2-silylated 1,3-dithiolanes for the synthesis of aryl/hetaryl-substituted ethenes and dibenzofulvenes Grzegorz Mlostoń *1, Paulina Pipiak 1, Róża Hamera-Fałdyga
Supporting Information. Copyright Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2009
 Supporting Information Copyright Wiley-VC Verlag Gmb & Co. KGaA, 69451 Weinheim, 2009 Supporting Information for Graphite-Supported Gold anoparticles as Efficient Catalyst for Aerobic xidation of Benzylic
Supporting Information Copyright Wiley-VC Verlag Gmb & Co. KGaA, 69451 Weinheim, 2009 Supporting Information for Graphite-Supported Gold anoparticles as Efficient Catalyst for Aerobic xidation of Benzylic
Modular Synthesis of Cyclic cis- and trans-1,2-diamine Derivatives
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 4 Modular Synthesis of Cyclic cis- and trans-,-diamine Derivatives Anna K. Weber,
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 4 Modular Synthesis of Cyclic cis- and trans-,-diamine Derivatives Anna K. Weber,
Supporting Information for
 Supporting Information for An atom-economic route to densely functionalized thiophenes via base-catalyzed rearrangement of 5-propargyl-2H-thiopyran-4(3H)-ones Chunlin Tang a, Jian Qin b, Xingqi Li *a a
Supporting Information for An atom-economic route to densely functionalized thiophenes via base-catalyzed rearrangement of 5-propargyl-2H-thiopyran-4(3H)-ones Chunlin Tang a, Jian Qin b, Xingqi Li *a a
Supporting Information
 Supporting Information Ceric Ammonium Nitrate (CAN) catalyzed efficient one-pot three component aza-diels-alder reactions for a facile synthesis of tetrahydropyranoquinoline derivatives Ravinder Goud Puligoundla
Supporting Information Ceric Ammonium Nitrate (CAN) catalyzed efficient one-pot three component aza-diels-alder reactions for a facile synthesis of tetrahydropyranoquinoline derivatives Ravinder Goud Puligoundla
Fluorinative Ring-opening of Cyclopropanes by Hypervalent Iodine Reagents. An Efficient Method for 1,3- Oxyfluorination and 1,3-Difluorination
 Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2016 Supporting Information Fluorinative Ring-opening of Cyclopropanes by Hypervalent Iodine
Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2016 Supporting Information Fluorinative Ring-opening of Cyclopropanes by Hypervalent Iodine
Highly enantioselective cascade synthesis of spiropyrazolones. Supporting Information. NMR spectra and HPLC traces
 Highly enantioselective cascade synthesis of spiropyrazolones Alex Zea a, Andrea-Nekane R. Alba a, Andrea Mazzanti b, Albert Moyano a and Ramon Rios a,c * Supporting Information NMR spectra and HPLC traces
Highly enantioselective cascade synthesis of spiropyrazolones Alex Zea a, Andrea-Nekane R. Alba a, Andrea Mazzanti b, Albert Moyano a and Ramon Rios a,c * Supporting Information NMR spectra and HPLC traces
Peptidomimetics as Protein Arginine Deiminase 4 (PAD4) Inhibitors
 Peptidomimetics as Protein Arginine Deiminase 4 (PAD4) Inhibitors Andrea Trabocchi a, icolino Pala b, Ilga Krimmelbein c, Gloria Menchi a, Antonio Guarna a, Mario Sechi b, Tobias Dreker c, Andrea Scozzafava
Peptidomimetics as Protein Arginine Deiminase 4 (PAD4) Inhibitors Andrea Trabocchi a, icolino Pala b, Ilga Krimmelbein c, Gloria Menchi a, Antonio Guarna a, Mario Sechi b, Tobias Dreker c, Andrea Scozzafava
Supplementary Figure 1. (X-ray structures of 6p and 7f) O N. Br 6p
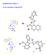 Supplementary Figure 1 (X-ray structures of 6p and 7f) Me Br 6p 6p Supplementary Figures 2-68 (MR Spectra) Supplementary Figure 2. 1 H MR of the 6a Supplementary Figure 3. 13 C MR of the 6a Supplementary
Supplementary Figure 1 (X-ray structures of 6p and 7f) Me Br 6p 6p Supplementary Figures 2-68 (MR Spectra) Supplementary Figure 2. 1 H MR of the 6a Supplementary Figure 3. 13 C MR of the 6a Supplementary
SUPPORTING INFORMATION
 SUPPORTING INFORMATION Heronamides A C, new polyketide macrolactams from an Australian marine-derived Streptomyces sp. A biosynthetic case for synchronized tandem electrocyclization. Ritesh Raju, Andrew
SUPPORTING INFORMATION Heronamides A C, new polyketide macrolactams from an Australian marine-derived Streptomyces sp. A biosynthetic case for synchronized tandem electrocyclization. Ritesh Raju, Andrew
A New Type of Bis(sulfonamide)-Diamine Ligand for a Cu(OTf) 2 -Catalyzed Asymmetric Friedel-Crafts Alkylation Reaction of Indoles with Nitroalkenes
 A ew Type of Bis(sulfonamide)-Diamine Ligand for a Cu(OTf) 2 -Catalyzed Asymmetric Friedel-Crafts Alkylation Reaction of Indoles with itroalkenes Jing Wu, Xincheng Li, Fan Wu, and Boshun Wan* Dalian Institute
A ew Type of Bis(sulfonamide)-Diamine Ligand for a Cu(OTf) 2 -Catalyzed Asymmetric Friedel-Crafts Alkylation Reaction of Indoles with itroalkenes Jing Wu, Xincheng Li, Fan Wu, and Boshun Wan* Dalian Institute
Regioselectivity in the Stille coupling reactions of 3,5- dibromo-2-pyrone.
 Regioselectivity in the Stille coupling reactions of 3,5- dibromo-2-pyrone. Won-Suk Kim, Hyung-Jin Kim and Cheon-Gyu Cho Department of Chemistry, Hanyang University, Seoul 133-791, Korea Experimental Section
Regioselectivity in the Stille coupling reactions of 3,5- dibromo-2-pyrone. Won-Suk Kim, Hyung-Jin Kim and Cheon-Gyu Cho Department of Chemistry, Hanyang University, Seoul 133-791, Korea Experimental Section
Chiral Brønsted Acid Catalyzed Enantioselective Intermolecular Allylic Aminations. Minyang Zhuang and Haifeng Du*
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 Chiral Brønsted Acid Catalyzed Enantioselective Intermolecular Allylic
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 Chiral Brønsted Acid Catalyzed Enantioselective Intermolecular Allylic
Supporting Information for
 Supporting Information for Palladium-Catalyzed C-H Bond Functionalization of C6-Arylpurines Hai-Ming Guo,* Wei-Hao Rao, Hong-Ying iu, Li-Li Jiang, Ge ng, Jia-Jia Jin, Xi-ing Yang, and Gui-Rong Qu* College
Supporting Information for Palladium-Catalyzed C-H Bond Functionalization of C6-Arylpurines Hai-Ming Guo,* Wei-Hao Rao, Hong-Ying iu, Li-Li Jiang, Ge ng, Jia-Jia Jin, Xi-ing Yang, and Gui-Rong Qu* College
Asymmetric Allylic Alkylation of Ketone Enolates: An Asymmetric Claisen Surrogate.
 Asymmetric Allylic Alkylation of Ketone Enolates: An Asymmetric Claisen Surrogate. Erin C. Burger and Jon A. Tunge* Department of Chemistry, University of Kansas, Lawrence, KS 0 Supporting Information
Asymmetric Allylic Alkylation of Ketone Enolates: An Asymmetric Claisen Surrogate. Erin C. Burger and Jon A. Tunge* Department of Chemistry, University of Kansas, Lawrence, KS 0 Supporting Information
Supporting information
 Supporting information Spectroscopic data of compounds 1a-f Benzyl bromide (1a) Yield: 81%. Colorless oil. b.p. 79-82 o C/15 mmhg (Lit. 83.5 84 o C/13 mmhg). 1 1 H NMR (CDCl 3 ): δ 4.45 (2H, s, PhCH 2
Supporting information Spectroscopic data of compounds 1a-f Benzyl bromide (1a) Yield: 81%. Colorless oil. b.p. 79-82 o C/15 mmhg (Lit. 83.5 84 o C/13 mmhg). 1 1 H NMR (CDCl 3 ): δ 4.45 (2H, s, PhCH 2
Supplementary Information for
 Supplementary Information for Organocatalytic Asymmetric Intramolecular [3+2] Cycloaddition: A Straightforward Approach to Access Multiply Substituted Hexahydrochromeno[4,3-b]pyrrolidine Derivatives in
Supplementary Information for Organocatalytic Asymmetric Intramolecular [3+2] Cycloaddition: A Straightforward Approach to Access Multiply Substituted Hexahydrochromeno[4,3-b]pyrrolidine Derivatives in
Rh(III)-Catalyzed C-H Amidation with N-hydroxycarbamates: A. new Entry to N-Carbamate Protected Arylamines
 Rh(III)-Catalyzed C-H Amidation with N-hydroxycarbamates: A new Entry to N-Carbamate Protected Arylamines Bing Zhou,* Juanjuan Du, Yaxi Yang,* Huijin Feng, Yuanchao Li Shanghai Institute of Materia Medica,
Rh(III)-Catalyzed C-H Amidation with N-hydroxycarbamates: A new Entry to N-Carbamate Protected Arylamines Bing Zhou,* Juanjuan Du, Yaxi Yang,* Huijin Feng, Yuanchao Li Shanghai Institute of Materia Medica,
Supplementary information
 Electronic Supplementary Material (ESI) for MedChemComm. This journal is The Royal Society of Chemistry 2015 Supplementary information Synthesis of carboxyimidamide-substituted benzo[c][1,2,5]oxadiazoles
Electronic Supplementary Material (ESI) for MedChemComm. This journal is The Royal Society of Chemistry 2015 Supplementary information Synthesis of carboxyimidamide-substituted benzo[c][1,2,5]oxadiazoles
Supporting Information
 Supporting Information Lewis Acid Mediated [2,3]-Sigmatropic Rearrangement of Allylic α-amino Amides. Jan Blid, Peter Brandt, Peter Somfai*, Department of Chemistry, rganic Chemistry, Royal Institute of
Supporting Information Lewis Acid Mediated [2,3]-Sigmatropic Rearrangement of Allylic α-amino Amides. Jan Blid, Peter Brandt, Peter Somfai*, Department of Chemistry, rganic Chemistry, Royal Institute of
Facile construction of the functionalized 4H-chromene via tandem. benzylation and cyclization. Jinmin Fan and Zhiyong Wang*
 Facile construction of the functionalized 4H-chromene via tandem benzylation and cyclization Jinmin Fan and Zhiyong Wang* Hefei National Laboratory for Physical Science at Microscale, Joint- Lab of Green
Facile construction of the functionalized 4H-chromene via tandem benzylation and cyclization Jinmin Fan and Zhiyong Wang* Hefei National Laboratory for Physical Science at Microscale, Joint- Lab of Green
Supporting Information
 Electronic Supplementary Material (ESI) for rganic Chemistry Frontiers. This journal is the Partner rganisations 2018 Palladium-catalyzed direct approach to α-cf 3 aryl ketones from arylboronic acids Bo
Electronic Supplementary Material (ESI) for rganic Chemistry Frontiers. This journal is the Partner rganisations 2018 Palladium-catalyzed direct approach to α-cf 3 aryl ketones from arylboronic acids Bo
Supporting information for
 Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2014 Supporting information for Mesoporous silica KIT-6 supported superparamagnetic CuFe 2 O
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2014 Supporting information for Mesoporous silica KIT-6 supported superparamagnetic CuFe 2 O
Cu-catalyzed stereoselective conjugate addition of arylboronic acids to alkynoates
 Cu-catalyzed stereoselective conjugate addition of arylboronic acids to alkynoates Yoshihiko Yamamoto,* Naohiro Kirai and Yu arada Department of Applied Chemistry, Graduate School of Science and Engineering,
Cu-catalyzed stereoselective conjugate addition of arylboronic acids to alkynoates Yoshihiko Yamamoto,* Naohiro Kirai and Yu arada Department of Applied Chemistry, Graduate School of Science and Engineering,
Eco-friendly synthesis of diverse and valuable 2-pyridones by catalyst- and solvent-free thermal multicomponent domino reaction
 Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2015 SUPPRTIG IFRMATI Eco-friendly synthesis of diverse and valuable 2-pyridones by catalyst-
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2015 SUPPRTIG IFRMATI Eco-friendly synthesis of diverse and valuable 2-pyridones by catalyst-
Supporting Information. Table of Contents. II. Experimental procedures. II. Copies of 1H and 13C NMR spectra for all compounds
 Electronic upplementary Material (EI) for rganic & Biomolecular Chemistry. This journal is The Royal ociety of Chemistry 2017 Laboratoire de Méthodologie et ynthèse de Produit aturels. Université du Québec
Electronic upplementary Material (EI) for rganic & Biomolecular Chemistry. This journal is The Royal ociety of Chemistry 2017 Laboratoire de Méthodologie et ynthèse de Produit aturels. Université du Québec
9-amino-(9-deoxy)cinchona alkaloids-derived novel chiral phase-transfer catalysts
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 9-amino-(9-deoxy)cinchona alkaloids-derived novel chiral phase-transfer
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 9-amino-(9-deoxy)cinchona alkaloids-derived novel chiral phase-transfer
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/NCHEM.1998 An organic thiyl radical catalyst for enantioselective cyclization Takuya Hashimoto, Yu Kawamata and Keiji Maruoka Department of Chemistry, Graduate School of Science, Kyoto University,
DOI: 10.1038/NCHEM.1998 An organic thiyl radical catalyst for enantioselective cyclization Takuya Hashimoto, Yu Kawamata and Keiji Maruoka Department of Chemistry, Graduate School of Science, Kyoto University,
Supporting Information
 Supporting Information Wiley-VCH 2008 69451 Weinheim, Germany Diphenylprolinol Silyl Ether as a Catalyst in an Enantioselective, Catalytic, Formal Aza [3+3] Cycloaddition Reaction for the Formation of
Supporting Information Wiley-VCH 2008 69451 Weinheim, Germany Diphenylprolinol Silyl Ether as a Catalyst in an Enantioselective, Catalytic, Formal Aza [3+3] Cycloaddition Reaction for the Formation of
Efficient Synthesis of Ureas by Direct Palladium-Catalyzed. Oxidative Carbonylation of Amines
 S1 Supporting Information for Efficient Synthesis of Ureas by Direct Palladium-Catalyzed Oxidative Carbonylation of Amines Bartolo Gabriele,*, Giuseppe Salerno, Raffaella Mancuso, and Mirco Costa Dipartimento
S1 Supporting Information for Efficient Synthesis of Ureas by Direct Palladium-Catalyzed Oxidative Carbonylation of Amines Bartolo Gabriele,*, Giuseppe Salerno, Raffaella Mancuso, and Mirco Costa Dipartimento
Iodine-catalyzed synthesis of sulfur-bridged enaminones and chromones via double C(sp 2 )-H thiolation
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2017 Iodine-catalyzed synthesis of sulfur-bridged enaminones and chromones via
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2017 Iodine-catalyzed synthesis of sulfur-bridged enaminones and chromones via
A facile and general route to 3-((trifluoromethyl)thio)benzofurans and 3-((trifluoromethyl)thio)benzothiophenes
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 A facile and general route to 3-((trifluoromethyl)thio)benzofurans and 3-((trifluoromethyl)thio)benzothiophenes
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 A facile and general route to 3-((trifluoromethyl)thio)benzofurans and 3-((trifluoromethyl)thio)benzothiophenes
Fast copper-, ligand- and solvent-free Sonogashira coupling in a ball mill
 Electronic Supporting Information Fast copper-, ligand- and solvent-free Sonogashira coupling in a ball mill Rico Thorwirth, Achim Stolle * and Bernd ndruschka Institute for Technical Chemistry and Environmental
Electronic Supporting Information Fast copper-, ligand- and solvent-free Sonogashira coupling in a ball mill Rico Thorwirth, Achim Stolle * and Bernd ndruschka Institute for Technical Chemistry and Environmental
Supplementary Data. Engineering, Nanjing University, Nanjing , P. R. China;
 Supplementary Data Synthesis, Chemo-selective Properties of Substituted 9-Aryl-9H-fluorenes from Triarylcarbinols and Enantiomerical Kinetics of Chiral 9-Methoxy-11-(naphthalen-1-yl)-11H-benzo[a]fluorene
Supplementary Data Synthesis, Chemo-selective Properties of Substituted 9-Aryl-9H-fluorenes from Triarylcarbinols and Enantiomerical Kinetics of Chiral 9-Methoxy-11-(naphthalen-1-yl)-11H-benzo[a]fluorene
Supporting Information For: Rhodium-Catalyzed Hydrofunctionalization: Enantioselective Coupling of Indolines and 1,3-Dienes
 Supporting Information For: Rhodium-Catalyzed Hydrofunctionalization: Enantioselective Coupling of Indolines and 1,3-Dienes Xiao-Hui Yang and Vy M. Dong* dongv@uci.edu Department of Chemistry, University
Supporting Information For: Rhodium-Catalyzed Hydrofunctionalization: Enantioselective Coupling of Indolines and 1,3-Dienes Xiao-Hui Yang and Vy M. Dong* dongv@uci.edu Department of Chemistry, University
Enantioselective Organocatalytic Michael Addition of Isorhodanines. to α, β-unsaturated Aldehydes
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2016 Enantioselective Organocatalytic Michael Addition of Isorhodanines to α,
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2016 Enantioselective Organocatalytic Michael Addition of Isorhodanines to α,
Supporting Information
 Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2010 A Highly Effective Bis(sulfonamide) Diamine Ligand: A Unique Chiral Skeleton for the Enantioselective Cu-Catalyzed
Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2010 A Highly Effective Bis(sulfonamide) Diamine Ligand: A Unique Chiral Skeleton for the Enantioselective Cu-Catalyzed
Diastereoselective Access to Trans-2-Substituted Cyclopentylamines
 Supporting Information Diastereoselective Access to Trans-2-Substituted Cyclopentylamines Antoine Joosten, Emilie Lambert, Jean-Luc Vasse, Jan Szymoniak jean-luc.vasse@univ-reims.fr jan.szymoniak@univ-reims.fr
Supporting Information Diastereoselective Access to Trans-2-Substituted Cyclopentylamines Antoine Joosten, Emilie Lambert, Jean-Luc Vasse, Jan Szymoniak jean-luc.vasse@univ-reims.fr jan.szymoniak@univ-reims.fr
Highly enantioselective hydrogenation of α-aryl-β-substituted acrylic acids catalyzed by Ir-SpinPHOX
 Electronic Supplementary Information (ESI) Highly enantioselective hydrogenation of α-aryl-β-substituted acrylic acids catalyzed by Ir-SpinPHOX Yi Zhang, a,b Zhaobin Han, b Fuying Li, a Kuiling Ding, b,*
Electronic Supplementary Information (ESI) Highly enantioselective hydrogenation of α-aryl-β-substituted acrylic acids catalyzed by Ir-SpinPHOX Yi Zhang, a,b Zhaobin Han, b Fuying Li, a Kuiling Ding, b,*
Supporting Information
 Supporting Information Regioselective Reversal in the Cyclization of 2-Diazo-3,5-dioxo-6-ynoates (ynones, ynamide): Construction of -Pyrones and 3(2H)-Furanones Starting from Identical Materials Feng Wang,
Supporting Information Regioselective Reversal in the Cyclization of 2-Diazo-3,5-dioxo-6-ynoates (ynones, ynamide): Construction of -Pyrones and 3(2H)-Furanones Starting from Identical Materials Feng Wang,
Cinchona-Alkaloids based Isoselenazolones: Synthesis and Their Catalytic Application in Asymmetric Bromolactonization of Alkenoic Acid
 Cinchona-Alkaloids based Isoselenazolones: Synthesis and Their Catalytic Application in Asymmetric Bromolactonization of Alkenoic Acid Contents Analytical data for isoselenazolones 1-14 and their substrates,
Cinchona-Alkaloids based Isoselenazolones: Synthesis and Their Catalytic Application in Asymmetric Bromolactonization of Alkenoic Acid Contents Analytical data for isoselenazolones 1-14 and their substrates,
Fischer Indole Synthesis in Low Melting Mixtures
 Fischer Indole Synthesis in Low Melting Mixtures Sangram Gore, a,b Sundarababu Baskaran* a and Burkhard König* b Supporting Information Table of Contents: General Information General Procedure for the
Fischer Indole Synthesis in Low Melting Mixtures Sangram Gore, a,b Sundarababu Baskaran* a and Burkhard König* b Supporting Information Table of Contents: General Information General Procedure for the
SUPPORTING INFORMATION. 1. General... S1. 2. General procedure for the synthesis of compounds 3 and 4 in the absence of AgOAc...
 SUPPORTING INFORMATION Table of contents 1. General.... S1 2. General procedure for the synthesis of compounds 3 and 4 in the absence of AgOAc... S2 3. General procedure for the synthesis of compounds
SUPPORTING INFORMATION Table of contents 1. General.... S1 2. General procedure for the synthesis of compounds 3 and 4 in the absence of AgOAc... S2 3. General procedure for the synthesis of compounds
Novel and Selective Palladium-Catalyzed Annulation of 2-Alkynylphenols to Form 2-Substituted 3-Halobenzo[b]furans. Supporting Information
![Novel and Selective Palladium-Catalyzed Annulation of 2-Alkynylphenols to Form 2-Substituted 3-Halobenzo[b]furans. Supporting Information Novel and Selective Palladium-Catalyzed Annulation of 2-Alkynylphenols to Form 2-Substituted 3-Halobenzo[b]furans. Supporting Information](/thumbs/64/50937700.jpg) Novel and Selective Palladium-Catalyzed Annulation of 2-Alkynylphenols to Form 2-Substituted 3-Halobenzo[b]furans Liang Yun, Shi Tang, Xu-Dong Zhang, Li-Qiu Mao, Ye-Xiang Xie and Jin-Heng Li* Key Laboratory
Novel and Selective Palladium-Catalyzed Annulation of 2-Alkynylphenols to Form 2-Substituted 3-Halobenzo[b]furans Liang Yun, Shi Tang, Xu-Dong Zhang, Li-Qiu Mao, Ye-Xiang Xie and Jin-Heng Li* Key Laboratory
Supporting Information
 Supporting Information Palladium-Catalyzed Asymmetric Benzylic Substitution of Secondary Benzyl Carbonates with Nitrogen and Oxygen Nucleophiles Atifah Najib, Koji Hirano,* and Masahiro Miura* Department
Supporting Information Palladium-Catalyzed Asymmetric Benzylic Substitution of Secondary Benzyl Carbonates with Nitrogen and Oxygen Nucleophiles Atifah Najib, Koji Hirano,* and Masahiro Miura* Department
Available online at
 J. Serb. Chem. Soc. 76 (12) S1 S5 (2011) Supplementary material SUPPLEMENTARY MATERIAL TO Synthesis of quinoline-attached furan-2(3h)-ones having anti-inflammatory and antibacterial properties with reduced
J. Serb. Chem. Soc. 76 (12) S1 S5 (2011) Supplementary material SUPPLEMENTARY MATERIAL TO Synthesis of quinoline-attached furan-2(3h)-ones having anti-inflammatory and antibacterial properties with reduced
D-Glucosamine-derived copper catalyst for Ullmann-type C- N coupling reaction: theoretical and experimental study
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 D-Glucosamine-derived copper catalyst for Ullmann-type C- N coupling reaction: theoretical
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 D-Glucosamine-derived copper catalyst for Ullmann-type C- N coupling reaction: theoretical
Zuxiao Zhang, Xiaojun Tang and William R. Dolbier, Jr.* Department of Chemistry, University of Florida, Gainesville, FL
 Photoredox-Catalyzed Intramolecular Difluoromethylation of -Benzylacrylamides Coupled with a Dearomatizing Spirocyclization: Access to CF2H Containing 2- Azaspiro[4.5]deca-6,9-diene-3,8-diones. Zuxiao
Photoredox-Catalyzed Intramolecular Difluoromethylation of -Benzylacrylamides Coupled with a Dearomatizing Spirocyclization: Access to CF2H Containing 2- Azaspiro[4.5]deca-6,9-diene-3,8-diones. Zuxiao
Ligand-free Cu(II)-mediated aerobic oxidations of aldehyde. hydrazones leading to N,N -diacylhydrazines and 1,3,4-oxadiazoles
 Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2017 Ligand-free Cu(II)-mediated aerobic oxidations of aldehyde hydrazones leading
Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2017 Ligand-free Cu(II)-mediated aerobic oxidations of aldehyde hydrazones leading
Eur. J. Inorg. Chem WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim, 2007 ISSN SUPPORTING INFORMATION
 Eur. J. Inorg. Chem. 2007 WILEY-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2007 ISSN 1434 1948 SUPPORTING INFORMATION Title: Synthesis of Cyclic Carbonates from Atmospheric Pressure Carbon Dioxide Using
Eur. J. Inorg. Chem. 2007 WILEY-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2007 ISSN 1434 1948 SUPPORTING INFORMATION Title: Synthesis of Cyclic Carbonates from Atmospheric Pressure Carbon Dioxide Using
Tributylphosphine-Catalyzed Cycloaddition of Aziridines with Carbon Disulfide and Isothiocyanate
 upporting Information Tributylphosphine-Catalyzed Cycloaddition of Aziridines with Carbon Disulfide and Isothiocyanate Jing-Yu Wu, Zhi-Bin Luo, Li-Xin Dai and Xue-Long Hou* a tate Key Laboratory of Organometallic
upporting Information Tributylphosphine-Catalyzed Cycloaddition of Aziridines with Carbon Disulfide and Isothiocyanate Jing-Yu Wu, Zhi-Bin Luo, Li-Xin Dai and Xue-Long Hou* a tate Key Laboratory of Organometallic
Direct Transformation of Ethylarenes into Primary Aromatic Amides with N-Bromosuccinimide and I 2 -aq NH 3
 Supporting Information Direct Transformation of Ethylarenes into Primary Aromatic Amides with N-Bromosuccinimide and I 2 -aq NH 3 Shohei Shimokawa, Yuhsuke Kawagoe, Katsuhiko Moriyama, Hideo Togo* Graduate
Supporting Information Direct Transformation of Ethylarenes into Primary Aromatic Amides with N-Bromosuccinimide and I 2 -aq NH 3 Shohei Shimokawa, Yuhsuke Kawagoe, Katsuhiko Moriyama, Hideo Togo* Graduate
Electronic Supplementary Information
 Electronic Supplementary Information NbCl 3 -catalyzed [2+2+2] intermolecular cycloaddition of alkynes and alkenes to 1,3-cyclohexadiene derivatives Yasushi Obora,* Keisuke Takeshita and Yasutaka Ishii*
Electronic Supplementary Information NbCl 3 -catalyzed [2+2+2] intermolecular cycloaddition of alkynes and alkenes to 1,3-cyclohexadiene derivatives Yasushi Obora,* Keisuke Takeshita and Yasutaka Ishii*
SUPPLEMENTARY MATERIAL
 10.1071/CH16014_AC The Authors 2016 Australian Journal of Chemistry 2016, 69(9), 1049-1053 SUPPLEMENTARY MATERIAL A Green Approach for the Synthesis of Novel 7,11-Dihydro-6H-chromeno[3,4- e]isoxazolo[5,4-b]pyridin-6-one
10.1071/CH16014_AC The Authors 2016 Australian Journal of Chemistry 2016, 69(9), 1049-1053 SUPPLEMENTARY MATERIAL A Green Approach for the Synthesis of Novel 7,11-Dihydro-6H-chromeno[3,4- e]isoxazolo[5,4-b]pyridin-6-one
Supporting Information
 Supporting Information Enantiospecific Synthesis of the Cubitane Skeleton Elisabeth Schöttner, M. Wiechoczek, Peter G. Jones, and Thomas Lindel * TU Braunschweig, Institutes of rganic, Inorganic and Analytical
Supporting Information Enantiospecific Synthesis of the Cubitane Skeleton Elisabeth Schöttner, M. Wiechoczek, Peter G. Jones, and Thomas Lindel * TU Braunschweig, Institutes of rganic, Inorganic and Analytical
Supporting Information. Copyright Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2007
 Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 6945 Weinheim, 2007 A New Method for Constructing Quaternary Carbon Centres: Tandem Rhodium-Catalysed,4-Addition/Intramolecular Cyclization.
Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 6945 Weinheim, 2007 A New Method for Constructing Quaternary Carbon Centres: Tandem Rhodium-Catalysed,4-Addition/Intramolecular Cyclization.
Supporting Information
 Supporting Information Direct Olefination of Alcohols with Sulfones by Using Heterogeneous Platinum Catalysts S. M. A. Hakim Siddiki, [a] Abeda Sultana Touchy, [b] Kenichi Kon, [b] [a, b] and Ken-ichi
Supporting Information Direct Olefination of Alcohols with Sulfones by Using Heterogeneous Platinum Catalysts S. M. A. Hakim Siddiki, [a] Abeda Sultana Touchy, [b] Kenichi Kon, [b] [a, b] and Ken-ichi
Hiyama Cross-Coupling of Chloro-, Fluoroand Methoxy- pyridyl trimethylsilanes : Room-temperature Novel Access to Functional Bi(het)aryl
 Hiyama Cross-Coupling of Chloro-, Fluoroand Methoxy- pyridyl trimethylsilanes : Room-temperature Novel Access to Functional Bi(het)aryl Philippe Pierrat, Philippe Gros* and Yves Fort Synthèse Organométallique
Hiyama Cross-Coupling of Chloro-, Fluoroand Methoxy- pyridyl trimethylsilanes : Room-temperature Novel Access to Functional Bi(het)aryl Philippe Pierrat, Philippe Gros* and Yves Fort Synthèse Organométallique
Electronic Supplementary Information. Carbon dioxide as a reversible amine-protecting
 Electronic Supplementary Information Carbon dioxide as a reversible amine-protecting agent in selective Michael additions and acylations Annelies Peeters, Rob Ameloot and Dirk E. De Vos * Centre for Surface
Electronic Supplementary Information Carbon dioxide as a reversible amine-protecting agent in selective Michael additions and acylations Annelies Peeters, Rob Ameloot and Dirk E. De Vos * Centre for Surface
Free Radical Initiated Coupling Reaction of Alcohols and. Alkynes: not C-O but C-C Bond Formation. Context. General information 2. Typical procedure 2
 Free Radical Initiated Coupling Reaction of Alcohols and Alkynes: not C-O but C-C Bond Formation Zhongquan Liu,* Liang Sun, Jianguo Wang, Jie Han, Yankai Zhao, Bo Zhou Institute of Organic Chemistry, Gannan
Free Radical Initiated Coupling Reaction of Alcohols and Alkynes: not C-O but C-C Bond Formation Zhongquan Liu,* Liang Sun, Jianguo Wang, Jie Han, Yankai Zhao, Bo Zhou Institute of Organic Chemistry, Gannan
Oxyhalogenation of thiols and disulfides into sulfonyl chlorides/ bromides in water using oxone-kx(x= Cl or Br)
 Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2014 Oxyhalogenation of thiols and disulfides into sulfonyl chlorides/ bromides in water using
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2014 Oxyhalogenation of thiols and disulfides into sulfonyl chlorides/ bromides in water using
phase: synthesis of biaryls, terphenyls and polyaryls
 Supporting Information for Studies on Pd/NiFe 2 O 4 catalyzed ligand-free Suzuki reaction in aqueous phase: synthesis of biaryls, terphenyls and polyaryls Sanjay R. Borhade and Suresh B. Waghmode* Address:
Supporting Information for Studies on Pd/NiFe 2 O 4 catalyzed ligand-free Suzuki reaction in aqueous phase: synthesis of biaryls, terphenyls and polyaryls Sanjay R. Borhade and Suresh B. Waghmode* Address:
An Enantioselective Oxidative C H/C H Cross-Coupling Reaction: Highly Efficient Method to Prepare Planar Chiral Ferrocenes
 Supporting Information for An Enantioselective Oxidative C H/C H Cross-Coupling Reaction: Highly Efficient Method to Prepare Planar Chiral Ferrocenes De-Wei Gao, Qing Gu, and Shu-Li You* State Key Laboratory
Supporting Information for An Enantioselective Oxidative C H/C H Cross-Coupling Reaction: Highly Efficient Method to Prepare Planar Chiral Ferrocenes De-Wei Gao, Qing Gu, and Shu-Li You* State Key Laboratory
Trienamine-Mediated Asymmetric [4+2]-Cycloaddition of α,β-unsaturated Ester Surrogates Applying 4-Nitro-5-Styrylisoxazoles
![Trienamine-Mediated Asymmetric [4+2]-Cycloaddition of α,β-unsaturated Ester Surrogates Applying 4-Nitro-5-Styrylisoxazoles Trienamine-Mediated Asymmetric [4+2]-Cycloaddition of α,β-unsaturated Ester Surrogates Applying 4-Nitro-5-Styrylisoxazoles](/thumbs/53/30969816.jpg) Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 14 Trienamine-Mediated Asymmetric [4+2]-Cycloaddition of α,β-unsaturated Ester Surrogates Applying 4-Nitro-5-Styrylisoxazoles
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 14 Trienamine-Mediated Asymmetric [4+2]-Cycloaddition of α,β-unsaturated Ester Surrogates Applying 4-Nitro-5-Styrylisoxazoles
