Supplementary Information Chemical Partition of the Radiative Decay Rate of Luminescence of Europium Complexes
|
|
|
- Θεοδοσία Βαρουξής
- 6 χρόνια πριν
- Προβολές:
Transcript
1 Supplementary Information Chemical Partition of the Radiative Decay Rate of Luminescence of Europium Complexes Nathalia B. D. Lima [a], José Diogo L. Dutra [a,b], Simone M. C. Gonçalves [a], Ricardo O. Freire [b], and Alfredo M. Simas [a] * [a] Departamento de Química Fundamental, CCEN, UFPE, Recife, PE, Brazil. [b] Pople Computational Chemistry Laboratory, Departamento de Química, CCET, UFS, São Cristóvão, SE, Brazil. * Corresponding author: simas@ufpe.br Tel
2 List of Tables Table S1. Fitted Q, D, and C values for all complexes studied, with electronic densities and electrophilic superdelocalizabilities 1, together with calculated and erimental values for the complexes of the general formula Eu(β-diketonate) 3(TPPO) 2 with the RM1 model (except where otherwise indicated), where β-diketonate stands for the ionic ligands TTA, BTFA, and DBM Table S2. Fitted Q, D, and C values for all complexes studied, with electronic densities and electrophilic superdelocalizabilities 1, together with calculated and erimental values for the complexes of the general formula Eu(β-diketonate) 3(L) 2 with the RM1 model (except where otherwise indicated), where β-diketonate stands for the ionic ligands TTA, BTFA, and DBM; and L stands for the non-ionic ligands TPPO, DBSO, and PTSO Table S3. Fitted Q, D, and C values for all complexes studied, with electronic densities and electrophilic superdelocalizabilities 1, together with calculated and erimental values for the complexes of the general formula Eu(β-diketonate) 3(L,L ) with the RM1 model (except where otherwise indicated), where β-diketonate stands for the ionic ligands TTA, BTFA, and DBM; and L and L stand for the non-ionic ligands TPPO, DBSO, and PTSO Table S4. Radiative decay rates A rad and Arad, as well as the ionic and non-ionic partitions of ionic A rad : A rad and A non ionic rad. A rad corresponds to the transitions from 5 D 0 to 7 F 2, 7 F 4, and 7 F 6, and is therefore always smaller thana rad which, in addition, also includes the transitions to 7 F 0, 7 F 1, 7 F 3, and 7 ionic F 5. The A rad partition comprises the terms for each of the three identical - diketonates, ordered into maximum (Max), medium (Med) and minimum (Min) values. The A non ionic rad partition comprises the terms for each of the two identical non-ionic ligands, ordered into maximum (Max), and minimum (Min) values. Geometries were optimized and the chemical partitions were calculated with the RM1 model (except where otherwise indicated) Table S5. Radiative decay rates A rad and Arad, as well as the ionic and non-ionic partitions ionic of A rad : A rad and A non ionic rad. A rad corresponds to the transitions from 5 D 0 to 7 F 2, 7 F 4, and 7 F 6, and is therefore always smaller than A rad which, in addition, also includes the transitions to 7 F 0, 7 F 1, 7 F 3, and 7 ionic F 5. The A rad partition comprises the terms for each of the three identical - diketonates, ordered into maximum (Max), medium (Med) and minimum (Min) values. The non ionic A rad partition comprises the terms for each of the two identical non-ionic ligands, ordered into maximum (Max), and minimum (Min) values. Geometries were optimized and the chemical partitions were calculated with the RM1 model (except where otherwise indicated) Table S6. Radiative decay rates A rad and Arad, as well as the ionic and non-ionic partitions of ionic A rad : A rad and A non ionic rad. A rad corresponds to the transitions from 5 D 0 to 7 F 2, 7 F 4, and 7 F 6, and is therefore always smaller than A rad which, in addition, also includes the transitions to 7 F 0, 7 F 1, 7 F 3, and 7 ionic F 5. The A rad partition comprises the terms for each of the three identical - diketonates, ordered into maximum (Max), medium (Med) and minimum (Min) values. The non ionic A rad partition is comprised of terms corresponding to each of the non-ionic ligands, as 2
3 indicated. Geometries were optimized and the chemical partitions were calculated with the RM1 model (except where otherwise indicated) Table S7. Spherical coordinates of all atoms belonging to the coordination polyhedron of the complexes of general formula Eu(BTFA) 3(L) 2 and Eu(BTFA) 3(L,L ), optimized with the RM1 model (except where otherwise indicated), where L and L stand for the non-ionic ligands TPPO, DBSO, and PTSO Table S8. Spherical coordinates of all atoms belonging to the coordination polyhedron of the complexes of general formula Eu(TTA) 3(L) 2 and Eu(TTA) 3(L,L ) optimized with the RM1 model, where L and L stand for the non-ionic ligands TPPO, DBSO, and PTSO Table S9. Spherical coordinates of all atoms belonging to the coordination polyhedron of the complexes of general formula Eu(DBM) 3(L) 2 and Eu(DBM) 3(L,L ) optimized with the RM1 model (except where otherwise indicated), where β-diketonate stands for the ionic ligands DBM; and L and L stand for the non-ionic ligands TPPO, DBSO, and PTSO
4 Table S1. Fitted Q, D, and C values for all complexes studied, with electronic densities and electrophilic superdelocalizabilities 1, together with calculated and erimental values for the complexes of the general formula Eu(β-diketonate)3(TPPO)2 with the RM1 model (except where otherwise indicated), where β-diketonate stands for the ionic ligands TTA, BTFA, and DBM. Complex Q D C D/C calc Ω 2 Ω 2 calc Ω 4 calc Ω 6 Eu(TTA) 3(TPPO) 2,Non-Adj Eu(TTA) 3(TPPO) 2,Adj Eu(BTFA) 3(TPPO) 2,Non-Adj Eu(BTFA) 3(TPPO) 2,Adj Eu(DBM) 3(TPPO) (a) 2,Non-Adj Eu(DBM) 3(TPPO) 2,Adj Units are: Q (au -1 ); D (au -1 Å3 ); C (Å 3 ); D/C (au -1 ); (10-20 cm 2 ). (a) Geometry was optimized and the chemical partition was calculated with Sparkle/PM3. Ω 4 4
5 Table S2. Fitted Q, D, and C values for all complexes studied, with electronic densities and electrophilic superdelocalizabilities 1, together with calculated and erimental values for the complexes of the general formula Eu(β-diketonate)3(L)2 with the RM1 model (except where otherwise indicated), where β-diketonate stands for the ionic ligands TTA, BTFA, and DBM; and L stands for the non-ionic ligands TPPO, DBSO, and PTSO. Complex Q D C D/C calc Ω 2 Ω 2 calc Ω 4 calc Ω 6 Eu(TTA) 3(DBSO) 2,Non-Adj Eu(TTA) 3(PTSO) 2,Non-Adj Eu(TTA) 3(TPPO) 2,Non-Adj Eu(BTFA) 3(DBSO) 2,Non-Adj Eu(BTFA) 3(PTSO) 2,Non-Adj Eu(BTFA) 3(TPPO) 2,Non-Adj Eu(DBM) 3(DBSO) 2,Non-Adj Eu(DBM) 3(PTSO) 2,Non-Adj Eu(DBM) 3(TPPO) (a) 2,Non-Adj Units are: Q (au -1 ); D (au -1 Å3 ); C (Å 3 ); D/C (au -1 ); (10-20 cm 2 ). (a) Geometry was optimized and the chemical partition was calculated with Sparkle/PM3. Ω 4 5
6 Table S3. Fitted Q, D, and C values for all complexes studied, with electronic densities and electrophilic superdelocalizabilities 1, together with calculated and erimental values for the complexes of the general formula Eu(β-diketonate)3(L,L ) with the RM1 model (except where otherwise indicated), where β-diketonate stands for the ionic ligands TTA, BTFA, and DBM; and L and L stand for the non-ionic ligands TPPO, DBSO, and PTSO. Complex Q D C D/C calc Ω 2 Ω 2 calc Ω 4 calc Ω 6 Eu(TTA) 3(DBSO,TPPO) Non-Adj Eu(TTA) 3(PTSO,TPPO) Non-Adj Eu(TTA) 3(DBSO,PTSO) Non-Adj Eu(BTFA) 3(DBSO,TPPO) Non-Adj (a) Eu(BTFA) 3(PTSO,TPPO) Non-Adj Eu(BTFA) 3(DBSO,PTSO) Non-Adj Eu(DBM) 3(DBSO,TPPO) Non-Adj Eu(DBM) 3(PTSO,TPPO) Non-Adj Eu(DBM) 3(DBSO,PTSO) Non-Adj Units are: Q (au -1 ); D (au -1 Å3 ); C (Å 3 ); D/C (au -1 ); (10-20 cm 2 ). (a) Geometry was optimized and the chemical partition was calculated with Sparkle/RM1. Ω 4 6
7 Complex Table S4. Radiative decay rates A rad and A rad, as well as the ionic and non-ionic ionic partitions of A rad : A rad and A non ionic rad. A rad corresponds to the transitions from 5 D0 to 7 F2, 7 F4, and 7 F6, and is therefore always smaller than A rad which, in addition, also includes the transitions to 7 F0, 7 F1, 7 F3, and 7 ionic F5. The A rad partition comprises the terms for each of the three identical -diketonates, ordered into maximum (Max), medium (Med) and minimum (Min) values. The A non ionic rad partition comprises the terms for each of the two identical non-ionic ligands, ordered into maximum (Max), and minimum (Min) values. Geometries were optimized and the chemical partitions were calculated with the RM1 model (except where otherwise indicated). A rad (s -1 ) A rad (s -1 ) Eu(TTA) 3(TPPO) 2,Non-Adj Eu(TTA) 3(TPPO) 2,Adj Eu(BTFA) 3(TPPO) 2,Non-Adj Eu(BTFA) 3(TPPO) 2,Adj ionic A rad (s -1 ) A rad (s -1 ) Max Med Min Max Min (42%) (34%) (8%) (12%) (4%) (23%) (19%) (7%) (26%) (24%) (30%) (27%) (23%) (20%) (0%) (33%) (11%) (8%) (45%) (4%) Eu(DBM) 3(TPPO) (a) 2,Non-Adj (23%) (23%) (20%) (22%) (13%) Eu(DBM) 3(TPPO) 2,Adj (31%) (22%) (18%) (25%) (4%) (a) Geometry was optimized and the chemical partition was calculated with Sparkle/PM3. 7
8 Table S5. Radiative decay rates A rad and A rad, as well as the ionic and non-ionic ionic partitions of A rad : A rad and A non ionic rad. A rad corresponds to the transitions from 5 D0 to 7 F2, 7 F4, and 7 F6, and is therefore always smaller than A rad which, in addition, also includes the transitions to 7 F0, 7 F1, 7 F3, and 7 ionic F5. The A rad partition comprises the terms for each of the three identical -diketonates, ordered into maximum (Max), medium (Med) and minimum (Min) values. The A non ionic rad partition comprises the terms for each of the two identical non-ionic ligands, ordered into maximum (Max), and minimum (Min) values. Geometries were optimized and the chemical partitions were calculated with the RM1 model (except where otherwise indicated). Complex Eu(TTA) 3(DBSO) 2,Non-Adj Eu(TTA) 3(PTSO) 2,Non-Adj Eu(TTA) 3(TPPO) 2,Non-Adj Eu(BTFA) 3(DBSO) 2,Non-Adj Eu(BTFA) 3(PTSO) 2,Non-Adj Eu(BTFA) 3(TPPO) 2,Non-Adj Eu(DBM) 3(DBSO) 2,Non-Adj A rad (s -1 ) A rad (s -1 ) ionic A rad (s -1 ) A rad (s -1 ) Max Med Min Max Min (65%) (17%) (8%) (9%) (2%) (71%) (15%) (12%) (1%) (0%) (42%) (34%) (8%) (12%) (4%) (52%) (21%) (9%) (14%) (3%) (36%) (20%) (4%) (38%) (2%) (30%) (27%) (23%) (20%) (0%) (52%) (19%) (17%) (7%) (5%) Eu(DBM) 3(PTSO) 2,Non-Adj (38%) (32%) (23%) (4%) (3%) Eu(DBM) 3(TPPO) (a) 2,Non-Adj (23%) (23%) (20%) (22%) (13%) (a) Geometry was optimized and the chemical partition was calculated with Sparkle/PM3. 8
9 Table S6. Radiative decay rates A rad and A rad, as well as the ionic and non-ionic ionic partitions of A rad : A rad and A non ionic rad. A rad corresponds to the transitions from 5 D0 to 7 F2, 7 F4, and 7 F6, and is therefore always smaller than A rad which, in addition, also includes the transitions to 7 F0, 7 F1, 7 F3, and 7 ionic F5. The A rad partition comprises the terms for each of the three identical -diketonates, ordered into maximum (Max), medium (Med) and minimum (Min) values. The A non ionic rad partition is comprised of terms corresponding to each of the non-ionic ligands, as indicated. Geometries were optimized and the chemical partitions were calculated with the RM1 model (except where otherwise indicated). Complex A rad (s -1 ) A rad (s -1 ) ionic A rad (s -1 ) A non ionic rad (s -1 ) Max Min Med DBSO PTSO TPPO Eu(TTA) 3(DBSO,TPPO) Non-Adj (44%) (30%) (10%) (3%) Eu(TTA) 3(PTSO,TPPO) Non-Adj (21%) (13%) (11%) (52%) Eu(TTA) 3(DBSO,PTSO) Non-Adj (39%) (18%) (6%) (36%) (1%) Eu(BTFA) 3(DBSO,TPPO) Non-Adj (a) (23%) (12%) (12%) (44%) Eu(BTFA) 3(PTSO,TPPO) Non-Adj (44%) (39%) (6%) (5%) Eu(BTFA) 3(DBSO,PTSO) Non-Adj (11%) (9%) (2%) (27%) (51%) Eu(DBM) 3(DBSO,PTSO) Non-Adj (55%) (22%) (16%) (6%) Eu(DBM) 3(PTSO,TPPO) Non-Adj (52%) (20%) (6%) (7%) Eu(DBM) 3(DBSO,PTSO) Non-Adj (49%) (16%) (14%) (7%) (15%) (a) Geometry was optimized and the chemical partition was calculated with Sparkle/RM (10%) 28 (3%) (10%) 54 (5%) (1%) 79 (15%)
10 Table S7. Spherical coordinates of all atoms belonging to the coordination polyhedron of the complexes of general formula Eu(BTFA)3(L)2 and Eu(BTFA)3(L,L ), optimized with the RM1 model (except where otherwise indicated), where L and L stand for the nonionic ligands TPPO, DBSO, and PTSO. Complex R (Å) θ ( ) φ ( ) Eu(BTFA) 3(DBSO) 2,Adj O (DBSO1) O (DBSO2) O (BTFA2) O (BTFA2) O (BTFA1) O (BTFA1) O (BTFA3) O (BTFA3) Eu(BTFA) 3(DBSO) 2,Non-Adj O (BTFA3) O (BTFA1) O (DBSO1) O (DBSO2) O (BTFA2) O (BTFA2) O (BTFA3) O (BTFA1) Eu(BTFA) 3(DBSO,PTSO) Adj O (DBSO) O (PTSO) O (BTFA3) O (BTFA3) O (BTFA2) O (BTFA2) O (BTFA1) O (BTFA1) Eu(BTFA) 3(DBSO,PTSO) Non-Adj O (PTSO) O (DBSO) O (BTFA3) O (BTFA3) O (BTFA1) O (BTFA1) O (BTFA2) O (BTFA2) Eu(BTFA) 3(DBSO,TPPO) Adj O (TPPO)
11 O (BTFA2) O (BTFA2) O (BTFA1) O (BTFA1) O (BTFA3) O (BTFA3) O (DBSO) Eu(BTFA) 3(DBSO,TPPO) (a) Non-Adj O (BTFA3) O (BTFA3) O (BTFA2) O (BTFA2) O (DBSO) O (TPPO) O (BTFA1) O (BTFA1) Eu(BTFA) 3(PTSO) 2,Adj O (PTSO1) O (BTFA1) O (BTFA1) O (BTFA2) O (BTFA2) O (BTFA3) O (BTFA3) O (PTSO2) Eu(BTFA) 3(PTSO) 2,Non-Adj O (BTFA2) O (BTFA2) O (PTSO1) O (BTFA3) O (BTFA3) O (PTSO2) O (BTFA1) O (BTFA1) Eu(BTFA) 3(PTSO,TPPO) Adj O (BTFA1) O (BTFA1) O (BTFA3) O (BTFA3) O (BTFA2) O (BTFA2) O (TPPO) O (PTSO) Eu(BTFA) 3(PTSO,TPPO) Non-Adj 11
12 O (BTFA1) O (BTFA2) O (TPPO) O (BTFA3) O (BTFA3) O (PTSO) O (BTFA1) O (BTFA2) Eu(BTFA) 3(TPPO) 2,Adj O (BTFA1) O (TPPO1) O (TPPO2) O (BTFA2) O (BTFA2) O (BTFA3) O (BTFA3) O (BTFA1) Eu(BTFA) 3(TPPO) 2,Non-Adj O (BTFA1) O (TPPO1) O (BTFA2) O (TPPO2) O (BTFA1) O (BTFA3) O (BTFA2) O (BTFA3) (a) Geometry optimized with Sparkle/RM1. 12
13 Table S8. Spherical coordinates of all atoms belonging to the coordination polyhedron of the complexes of general formula Eu(TTA)3(L)2 and Eu(TTA)3(L,L ) optimized with the RM1 model, where L and L stand for the non-ionic ligands TPPO, DBSO, and PTSO. Complex R (Å) θ ( ) φ ( ) Eu(TTA) 3(DBSO) 2,Adj O (DBSO1) O (TTA1) O (TTA1) O (TTA3) O (TTA2) O (TTA2) O (TTA3) O (DBSO2) Eu(TTA) 3(DBSO) 2,Non-Adj O (TTA1) O (TTA1) O (DBSO1) O (TTA2) O (TTA2) O (DBSO2) O (TTA3) O (TTA3) Eu(TTA) 3(DBSO,PTSO) Adj O (PTSO) O (DBSO) O (TTA2) O (TTA2) O (TTA1) O (TTA1) O (TTA3) O (TTA3) Eu(TTA) 3(DBSO,PTSO) Non-Adj O (DBSO) O (TTA1) O (PTSO) O (TTA3) O (TTA3) O (TTA2) O (TTA2) O (TTA1) Eu(TTA) 3(DBSO,TPPO) Adj O (TTA3) O (TTA3) O (DBSO)
14 O (TPPO) O (TTA2) O (TTA2) O (TTA1) O (TTA1) Eu(TTA) 3(DBSO,TPPO) Non-Adj O (TTA2) O (TTA2) O (DBSO) O (TPPO) O (TTA3) O (TTA3) O (TTA1) O (TTA1) Eu(TTA) 3(PTSO) 2,Adj O (PTSO1) O (TTA3) O (TTA3) O (TTA2) O (TTA2) O (PTSO2) O (TTA1) O (TTA1) Eu(TTA) 3(PTSO) 2,Non-Adj O (PTSO1) O (PTSO2) O (TTA2) O (TTA2) O (TTA1) O (TTA1) O (TTA3) O (TTA3) Eu(TTA) 3(PTSO,TPPO) Adj O (TPPO) O (TTA1) O (TTA1) O (TTA2) O (TTA2) O (PTSO) O (TTA3) O (TTA3) Eu(TTA) 3(PTSO,TPPO) Non-Adj O (PTSO) O (TTA1)
15 O (TPPO) O (TTA2) O (TTA2) O (TTA3) O (TTA3) O (TTA1) Eu(TTA) 3(TPPO) 2,Adj O (TTA2) O (TTA2) O (TTA3) O (TTA3) O (TPPO1) O (TTA1) O (TTA1) O (TPPO2) Eu(TTA) 3(TPPO) 2,Non-Adj O (TTA1) O (TPPO1) O (TTA2) O (TTA2) O (TPPO2) O (TTA3) O (TTA3) O (TTA1)
16 Table S9. Spherical coordinates of all atoms belonging to the coordination polyhedron of the complexes of general formula Eu(DBM)3(L)2 and Eu(DBM)3(L,L ) optimized with the RM1 model (except where otherwise indicated), where β-diketonate stands for the ionic ligands DBM; and L and L stand for the non-ionic ligands TPPO, DBSO, and PTSO. Complex R (Å) θ ( ) φ ( ) Eu(DBM) 3(DBSO) 2,Adj O (DBSO1) O (DBSO2) O (DBM1) O (DBM1) O (DBM2) O (DBM2) O (DBM3) O (DBM3) Eu(DBM) 3(DBSO) 2,Non-Adj O (DBM1) O (DBM1) O (DBM2) O (DBM2) O (DBSO1) O (DBM3) O (DBM3) O (DBSO2) Eu(DBM) 3(DBSO,PTSO) Adj O (DBM1) O (DBM1) O (DBM2) O (DBM2) O (PTSO) O (DBSO) O (DBM3) O (DBM3) Eu(DBM) 3(DBSO,PTSO) Non-Adj O (DBSO) O (DBM1) O (DBM1) O (DBM2) O (DBM2) O (PTSO) O (DBM3) O (DBM3) Eu(DBM) 3(DBSO,TPPO) Adj O (DBSO)
17 O (TPPO) O (DBM1) O (DBM2) O (DBM2) O (DBM3) O (DBM3) O (DBM1) Eu(DBM) 3(DBSO,TPPO) Non-Adj O (DBSO) O (TPPO) O (DBM1) O (DBM1) O (DBM2) O (DBM2) O (DBM3) O (DBM3) Eu(DBM) 3(PTSO) 2,Adj O (DBM1) O (PTSO1) O (PTSO2) O (DBM2) O (DBM2) O (DBM3) O (DBM3) O (DBM1) Eu(DBM) 3(PTSO) 2,Non-Adj O (DBM1) O (DBM1) O (DBM2) O (DBM2) O (PTSO1) O (DBM3) O (DBM3) O (PTSO2) Eu(DBM)3(PTSO,TPPO) Adj O (PTSO) O (TPPO) O (DBM1) O (DBM3) O (DBM3) O (DBM2) O (DBM2) O (DBM1) Eu(DBM) 3(PTSO,TPPO) Non-Adj 17
18 O (DBM1) O (PTSO) O (DBM2) O (DBM2) O (TPPO) O (DBM3) O (DBM1) O (DBM3) Eu(DBM) 3(TPPO) (a) 2,Adj O (DBM1) O (TPPO1) O (DBM2) O (TPPO2) O (DBM2) O (DBM3) O (DBM3) O (DBM1) Eu(DBM) 3(TPPO) 2,Non-Adj O (TPPO1) O (TPPO2) O (DBM1) O (DBM3) O (DBM2) O (DBM1) O (DBM2) O (DBM3) (a) Geometry optimized with Sparkle/PM3. Reference 1. Dutra, J.D.L., Lima, N.B.D., Freire, R.O. & Simas, A.M. Europium Luminescence: Electronic Densities and Superdelocalizabilities for a Unique Adjustment of Theoretical Intensity Parameters. Sci. Rep. 5, (2015). 18
Electronic Supplementary Information (ESI)
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information (ESI) CPh 3 as a functional group in P-heterocyclic
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information (ESI) CPh 3 as a functional group in P-heterocyclic
10-π-electron arenes à la carte: Structure. Sr, Ba; n = 6-8) complexes
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2016 Supporting information 10-π-electron arenes à la carte: Structure and Bonding of
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2016 Supporting information 10-π-electron arenes à la carte: Structure and Bonding of
Butadiene as a Ligand in Open Sandwich Compounds
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2018 Butadiene as a Ligand in Open Sandwich Compounds Qunchao Fan, a Jia Fu, a Huidong
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2018 Butadiene as a Ligand in Open Sandwich Compounds Qunchao Fan, a Jia Fu, a Huidong
Supporting Information. A Combined Crossed Molecular Beams and ab Initio Investigation on the Formation of Vinylsulfidoboron (C 2 H
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2014 Supporting Information for A Combined Crossed Molecular Beams and ab Initio Investigation
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2014 Supporting Information for A Combined Crossed Molecular Beams and ab Initio Investigation
Computational study of the structure, UV-vis absorption spectra and conductivity of biphenylene-based polymers and their boron nitride analogues
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Computational study of the structure, UV-vis absorption spectra and conductivity of biphenylene-based
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Computational study of the structure, UV-vis absorption spectra and conductivity of biphenylene-based
Engineering Tunable Single and Dual Optical. Emission from Ru(II)-Polypyridyl Complexes. Through Excited State Design
 Engineering Tunable Single and Dual Optical Emission from Ru(II)-Polypyridyl Complexes Through Excited State Design Supplementary Information Julia Romanova 1, Yousif Sadik 1, M. R. Ranga Prabhath 1,,
Engineering Tunable Single and Dual Optical Emission from Ru(II)-Polypyridyl Complexes Through Excited State Design Supplementary Information Julia Romanova 1, Yousif Sadik 1, M. R. Ranga Prabhath 1,,
Zebra reaction or the recipe for heterodimeric zinc complexes synthesis
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 05 Supporting Information Zebra reaction or the recipe for heterodimeric zinc complexes synthesis
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 05 Supporting Information Zebra reaction or the recipe for heterodimeric zinc complexes synthesis
Table of Contents 1 Supplementary Data MCD
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2017 Supporting Information for Magnetic circular dichroism and density functional theory
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2017 Supporting Information for Magnetic circular dichroism and density functional theory
Electronic Supplementary Information (ESI)
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information (ESI) Cyclopentadienyl iron dicarbonyl (CpFe(CO) 2 ) derivatives
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information (ESI) Cyclopentadienyl iron dicarbonyl (CpFe(CO) 2 ) derivatives
chlorostibine Iou-Sheng Ke and François P. Gabbai Department of Chemistry, Texas A&M University, College Station, TX
 σ-donor/acceptor confused ligands: The case of a chlorostibine Iou-Sheng Ke and François P. Gabbai Department of Chemistry, Texas A&M University, College Station, TX 77843-3255. *To whom correspondence
σ-donor/acceptor confused ligands: The case of a chlorostibine Iou-Sheng Ke and François P. Gabbai Department of Chemistry, Texas A&M University, College Station, TX 77843-3255. *To whom correspondence
Supporting Information
 Supporting Information rigin of the Regio- and Stereoselectivity of Allylic Substitution of rganocopper Reagents Naohiko Yoshikai, Song-Lin Zhang, and Eiichi Nakamura* Department of Chemistry, The University
Supporting Information rigin of the Regio- and Stereoselectivity of Allylic Substitution of rganocopper Reagents Naohiko Yoshikai, Song-Lin Zhang, and Eiichi Nakamura* Department of Chemistry, The University
difluoroboranyls derived from amides carrying donor group Supporting Information
 The influence of the π-conjugated spacer on photophysical properties of difluoroboranyls derived from amides carrying donor group Supporting Information Anna Maria Grabarz a Adèle D. Laurent b, Beata Jędrzejewska
The influence of the π-conjugated spacer on photophysical properties of difluoroboranyls derived from amides carrying donor group Supporting Information Anna Maria Grabarz a Adèle D. Laurent b, Beata Jędrzejewska
k A = [k, k]( )[a 1, a 2 ] = [ka 1,ka 2 ] 4For the division of two intervals of confidence in R +
[a 1, a 2 ] = [ka 1,ka 2 ] 4For the division of two intervals of confidence in R + k A = [k, k]( )[a 1, a 2 ] = [ka 1,ka 2 ] 4For the division of two intervals of confidence in R +](/thumbs/73/69566903.jpg) Chapter 3. Fuzzy Arithmetic 3- Fuzzy arithmetic: ~Addition(+) and subtraction (-): Let A = [a and B = [b, b in R If x [a and y [b, b than x+y [a +b +b Symbolically,we write A(+)B = [a (+)[b, b = [a +b
Chapter 3. Fuzzy Arithmetic 3- Fuzzy arithmetic: ~Addition(+) and subtraction (-): Let A = [a and B = [b, b in R If x [a and y [b, b than x+y [a +b +b Symbolically,we write A(+)B = [a (+)[b, b = [a +b
An experimental and theoretical study of the gas phase kinetics of atomic chlorine reactions with CH 3 NH 2, (CH 3 ) 2 NH, and (CH 3 ) 3 N
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2015 An experimental and theoretical study of the gas phase kinetics of atomic chlorine
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2015 An experimental and theoretical study of the gas phase kinetics of atomic chlorine
Electronic Supplementary Information (ESI)
 Electronic Supplementary Information (ESI) Lanthanide metal-organic frameworks constructed by asymmetric 2-nitro-biphenyl-4,4 -dicarboxylate ligand: syntheses, structures, luminescence and magnetic investigations
Electronic Supplementary Information (ESI) Lanthanide metal-organic frameworks constructed by asymmetric 2-nitro-biphenyl-4,4 -dicarboxylate ligand: syntheses, structures, luminescence and magnetic investigations
Supporting Information
 Supporting Information Aluminum Complexes of N 2 O 2 3 Formazanate Ligands Supported by Phosphine Oxide Donors Ryan R. Maar, Amir Rabiee Kenaree, Ruizhong Zhang, Yichen Tao, Benjamin D. Katzman, Viktor
Supporting Information Aluminum Complexes of N 2 O 2 3 Formazanate Ligands Supported by Phosphine Oxide Donors Ryan R. Maar, Amir Rabiee Kenaree, Ruizhong Zhang, Yichen Tao, Benjamin D. Katzman, Viktor
Hydrogen Sorption Efficiency of Titanium Decorated Calix[4]pyrroles
![Hydrogen Sorption Efficiency of Titanium Decorated Calix[4]pyrroles Hydrogen Sorption Efficiency of Titanium Decorated Calix[4]pyrroles](/thumbs/84/89186739.jpg) Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 Hydrogen Sorption Efficiency of Titanium Decorated Calix[4]pyrroles Sandeep Kumar,
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 Hydrogen Sorption Efficiency of Titanium Decorated Calix[4]pyrroles Sandeep Kumar,
Carbohydrates in the gas phase: conformational preference of D-ribose and 2-deoxy-D-ribose
 New J. Chem ELECTRONIC SUPPLEMENTARY INFORMATION Carbohydrates in the gas phase: conformational preference of D-ribose and 2-deoxy-D-ribose Luis Miguel Azofra,*, María Mar Quesada-Moreno, Ibon Alkorta,
New J. Chem ELECTRONIC SUPPLEMENTARY INFORMATION Carbohydrates in the gas phase: conformational preference of D-ribose and 2-deoxy-D-ribose Luis Miguel Azofra,*, María Mar Quesada-Moreno, Ibon Alkorta,
Electronic Supplementary Information:
 Electronic Supplementary Information: 2 6 5 7 2 N S 3 9 6 7 5 2 S N 3 9 Scheme S. Atom numbering for thione and thiol tautomers of thioacetamide 3 0.6 0. absorbance 0.2 0.0 0.5 0.0 0.05 0.00 N 2 Ar km/mol
Electronic Supplementary Information: 2 6 5 7 2 N S 3 9 6 7 5 2 S N 3 9 Scheme S. Atom numbering for thione and thiol tautomers of thioacetamide 3 0.6 0. absorbance 0.2 0.0 0.5 0.0 0.05 0.00 N 2 Ar km/mol
Nitric oxide (NO) reactivity studies on mononuclear Iron(II) complexes supported by a tetradentate Schiff base Ligand
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Nitric oxide (NO) reactivity studies on mononuclear Iron(II) complexes supported by a tetradentate
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Nitric oxide (NO) reactivity studies on mononuclear Iron(II) complexes supported by a tetradentate
of the methanol-dimethylamine complex
 Electronic Supplementary Information for: Fundamental and overtone virational spectroscopy, enthalpy of hydrogen ond formation and equilirium constant determination of the methanol-dimethylamine complex
Electronic Supplementary Information for: Fundamental and overtone virational spectroscopy, enthalpy of hydrogen ond formation and equilirium constant determination of the methanol-dimethylamine complex
Electronic Supplementary Information DFT Characterization on the Mechanism of Water Splitting Catalyzed by Single-Ru-substituted Polyoxometalates
 Electronic Supplementary Information DFT Characterization on the Mechanism of Water Splitting Catalyzed by Single-Ru-substituted Polyoxometalates Zhong-Ling Lang, Guo-Chun Yang, Na-Na Ma, Shi-Zheng Wen,
Electronic Supplementary Information DFT Characterization on the Mechanism of Water Splitting Catalyzed by Single-Ru-substituted Polyoxometalates Zhong-Ling Lang, Guo-Chun Yang, Na-Na Ma, Shi-Zheng Wen,
Supporting Information for: electron ligands: Complex formation, oxidation and
 Supporting Information for: The diverse reactions of PhI(OTf) 2 with common 2- electron ligands: Complex formation, oxidation and oxidative coupling Thomas P. Pell, Shannon A. Couchman, Sara Ibrahim, David
Supporting Information for: The diverse reactions of PhI(OTf) 2 with common 2- electron ligands: Complex formation, oxidation and oxidative coupling Thomas P. Pell, Shannon A. Couchman, Sara Ibrahim, David
Supplementary Information
 9.4 6.60. 8.6 8.8 9.29 20.04 29.6 0.07 6. 40.92 4.64 06.0 6.29 6.0-0.00 Supplementary Information Conformational Analysis, Experimental and GIA-DFT C NMR Chemical Shift Calculation on 2 -Hydroxy-,4,-trimethoxy-chalcone
9.4 6.60. 8.6 8.8 9.29 20.04 29.6 0.07 6. 40.92 4.64 06.0 6.29 6.0-0.00 Supplementary Information Conformational Analysis, Experimental and GIA-DFT C NMR Chemical Shift Calculation on 2 -Hydroxy-,4,-trimethoxy-chalcone
Heterobimetallic Pd-Sn Catalysis: Michael Addition. Reaction with C-, N-, O-, S- Nucleophiles and In-situ. Diagnostics
 Supporting Information (SI) Heterobimetallic Pd-Sn Catalysis: Michael Addition Reaction with C-, N-, -, S- Nucleophiles and In-situ Diagnostics Debjit Das, a Sanjay Pratihar a,b and Sujit Roy c * a rganometallics
Supporting Information (SI) Heterobimetallic Pd-Sn Catalysis: Michael Addition Reaction with C-, N-, -, S- Nucleophiles and In-situ Diagnostics Debjit Das, a Sanjay Pratihar a,b and Sujit Roy c * a rganometallics
Supporting Information
 Supporting Information On the Ambiguity of 1,3,2-Benzodiazaboroles as Donor/Acceptor unctionalities in Luminescent Molecules Lothar Weber *[a], Johannes Halama [a], Kenny Hanke [a], Lena Böhling [a], Andreas
Supporting Information On the Ambiguity of 1,3,2-Benzodiazaboroles as Donor/Acceptor unctionalities in Luminescent Molecules Lothar Weber *[a], Johannes Halama [a], Kenny Hanke [a], Lena Böhling [a], Andreas
ELECTRONIC SUPPORTING INFORMATION
 ELECTRONIC SUPPORTING INFORMATION Spectroscopic signatures of the carbon buckyonions C 60 @C 180 and C 60 @C 240 : a dispersion-corrected DFT study. Girolamo Casella, a Alessandro Bagno a and Giacomo Saielli*
ELECTRONIC SUPPORTING INFORMATION Spectroscopic signatures of the carbon buckyonions C 60 @C 180 and C 60 @C 240 : a dispersion-corrected DFT study. Girolamo Casella, a Alessandro Bagno a and Giacomo Saielli*
Electronic Supplementary Information
 Electronic Supplementary Information The preferred all-gauche conformations in 3-fluoro-1,2-propanediol Laize A. F. Andrade, a Josué M. Silla, a Claudimar J. Duarte, b Roberto Rittner, b Matheus P. Freitas*,a
Electronic Supplementary Information The preferred all-gauche conformations in 3-fluoro-1,2-propanediol Laize A. F. Andrade, a Josué M. Silla, a Claudimar J. Duarte, b Roberto Rittner, b Matheus P. Freitas*,a
Supporting Information. Generation Response. Physics & Chemistry of CAS, 40-1 South Beijing Road, Urumqi , China. China , USA
 Supporting Information Pb 3 B 6 O 11 F 2 : A First Noncentrocentric Lead Fluoroborate with Large Second Harmonic Generation Response Hongyi Li, a Hongping Wu, a * Xin Su, a Hongwei Yu, a,b Shilie Pan,
Supporting Information Pb 3 B 6 O 11 F 2 : A First Noncentrocentric Lead Fluoroborate with Large Second Harmonic Generation Response Hongyi Li, a Hongping Wu, a * Xin Su, a Hongwei Yu, a,b Shilie Pan,
Supporting Information
 Supporting Information Wiley-VCH 27 69451 Weinheim, Germany Supplementary Figure 1. Synthetic results as detected by XRD (Cu-Kα). Simulation pattern of MCM-68 Relative Intensity / a.u. YNU-2P Conventional
Supporting Information Wiley-VCH 27 69451 Weinheim, Germany Supplementary Figure 1. Synthetic results as detected by XRD (Cu-Kα). Simulation pattern of MCM-68 Relative Intensity / a.u. YNU-2P Conventional
Heavier chalcogenone complexes of bismuth(iii)trihalides: Potential catalysts for acylative cleavage of cyclic ethers. Supporting Information
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Heavier chalcogenone complexes of bismuth(iii)trihalides: Potential catalysts for acylative
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Heavier chalcogenone complexes of bismuth(iii)trihalides: Potential catalysts for acylative
Supplementary Information
 Electronic upplementary Material (EI) for Photochemical & Photobiological ciences. This journal is The Royal ociety of Chemistry and wner ocieties 214 upplementary Information elective and sensitive fluorescence-shift
Electronic upplementary Material (EI) for Photochemical & Photobiological ciences. This journal is The Royal ociety of Chemistry and wner ocieties 214 upplementary Information elective and sensitive fluorescence-shift
Nickel and Platinum PCP Pincer Complexes Incorporating an Acyclic Diaminoalkyl Central Moiety Connecting Imidazole or Pyrazole Rings
 ickel and Platinum PCP Pincer Complexes Incorporating an Acyclic Diaminoalkyl Central Moiety Connecting Imidazole or Pyrazole Rings Braulio M. Puerta Lombardi, Rudy M. Braun, Chris Gendy, Chia Yun Chang,
ickel and Platinum PCP Pincer Complexes Incorporating an Acyclic Diaminoalkyl Central Moiety Connecting Imidazole or Pyrazole Rings Braulio M. Puerta Lombardi, Rudy M. Braun, Chris Gendy, Chia Yun Chang,
Supplementary Information for
 Supplentary Information for Aggregation induced blue-shifted ission molecular picture from QM/MM study Qunyan Wu, a Tian Zhang, a Qian Peng,* b Dong Wang, a and Zhigang Shuai* a a Key Loratory of Organic
Supplentary Information for Aggregation induced blue-shifted ission molecular picture from QM/MM study Qunyan Wu, a Tian Zhang, a Qian Peng,* b Dong Wang, a and Zhigang Shuai* a a Key Loratory of Organic
Structural Expression of Exo-Anomeric Effect
 Supporting Information for Structural Expression of Exo-Anomeric Effect Elena R. Alonso, Isabel Peña, Carlos Cabezas, and José L. Alonso* Contents Table S1: Transition frequencies of conformer cc-β- 4
Supporting Information for Structural Expression of Exo-Anomeric Effect Elena R. Alonso, Isabel Peña, Carlos Cabezas, and José L. Alonso* Contents Table S1: Transition frequencies of conformer cc-β- 4
ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΤΜΗΜΑ ΟΔΟΝΤΙΑΤΡΙΚΗΣ ΕΡΓΑΣΤΗΡΙΟ ΟΔΟΝΤΙΚΗΣ ΚΑΙ ΑΝΩΤΕΡΑΣ ΠΡΟΣΘΕΤΙΚΗΣ
 ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΤΜΗΜΑ ΟΔΟΝΤΙΑΤΡΙΚΗΣ ΕΡΓΑΣΤΗΡΙΟ ΟΔΟΝΤΙΚΗΣ ΚΑΙ ΑΝΩΤΕΡΑΣ ΠΡΟΣΘΕΤΙΚΗΣ ΣΥΓΚΡΙΤΙΚΗ ΜΕΛΕΤΗ ΤΗΣ ΣΥΓΚΡΑΤΗΤΙΚΗΣ ΙΚΑΝΟΤΗΤΑΣ ΟΡΙΣΜΕΝΩΝ ΠΡΟΚΑΤΑΣΚΕΥΑΣΜΕΝΩΝ ΣΥΝΔΕΣΜΩΝ ΑΚΡΙΒΕΙΑΣ
ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΤΜΗΜΑ ΟΔΟΝΤΙΑΤΡΙΚΗΣ ΕΡΓΑΣΤΗΡΙΟ ΟΔΟΝΤΙΚΗΣ ΚΑΙ ΑΝΩΤΕΡΑΣ ΠΡΟΣΘΕΤΙΚΗΣ ΣΥΓΚΡΙΤΙΚΗ ΜΕΛΕΤΗ ΤΗΣ ΣΥΓΚΡΑΤΗΤΙΚΗΣ ΙΚΑΝΟΤΗΤΑΣ ΟΡΙΣΜΕΝΩΝ ΠΡΟΚΑΤΑΣΚΕΥΑΣΜΕΝΩΝ ΣΥΝΔΕΣΜΩΝ ΑΚΡΙΒΕΙΑΣ
Supporting Information. Palladium Complexes with Bulky Diphosphine. Synthesis of (Bio-) Adipic Acid from Pentenoic. Acid Mixtures.
 Supporting Information Palladium Complexes with Bulky Diphosphine Ligands as Highly Selective Catalysts for the Synthesis of (Bio-) Adipic Acid from Pentenoic Acid Mixtures. Choon Heng Low, James D. Nobbs,*
Supporting Information Palladium Complexes with Bulky Diphosphine Ligands as Highly Selective Catalysts for the Synthesis of (Bio-) Adipic Acid from Pentenoic Acid Mixtures. Choon Heng Low, James D. Nobbs,*
Synthesis of New Heteroscorpionate Iridium(I) and Iridium(III) Complexes
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2015 ELECTRONIC SUPPORTING INFORMATION For Synthesis of New Heteroscorpionate Iridium(I)
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2015 ELECTRONIC SUPPORTING INFORMATION For Synthesis of New Heteroscorpionate Iridium(I)
Ordinal Arithmetic: Addition, Multiplication, Exponentiation and Limit
 Ordinal Arithmetic: Addition, Multiplication, Exponentiation and Limit Ting Zhang Stanford May 11, 2001 Stanford, 5/11/2001 1 Outline Ordinal Classification Ordinal Addition Ordinal Multiplication Ordinal
Ordinal Arithmetic: Addition, Multiplication, Exponentiation and Limit Ting Zhang Stanford May 11, 2001 Stanford, 5/11/2001 1 Outline Ordinal Classification Ordinal Addition Ordinal Multiplication Ordinal
Supplementary Information
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2014 Supplementary Information A contribution to the rational design of Ru(CO) 3 Cl 2 L complexes
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2014 Supplementary Information A contribution to the rational design of Ru(CO) 3 Cl 2 L complexes
Supporting Information for Substituent Effects on the Properties of Borafluorenes
 Supporting Information for Substituent Effects on the Properties of Borafluorenes Mallory F. Smith, S. Joel Cassidy, Ian A. Adams, Monica Vasiliu, Deidra L. Gerlach, David Dixon*, Paul A. Rupar* Department
Supporting Information for Substituent Effects on the Properties of Borafluorenes Mallory F. Smith, S. Joel Cassidy, Ian A. Adams, Monica Vasiliu, Deidra L. Gerlach, David Dixon*, Paul A. Rupar* Department
Supplementary Materials for. Kinetic and Computational Studies on Pd(I) Dimer- Mediated Halogen Exchange of Aryl Iodides
 Supplementary Materials for Kinetic and Computational Studies on Pd(I) Dimer- Mediated Halogen Exchange of Aryl Iodides Indrek Kalvet, a Karl J. Bonney, a and Franziska Schoenebeck a * a Institute of Organic
Supplementary Materials for Kinetic and Computational Studies on Pd(I) Dimer- Mediated Halogen Exchange of Aryl Iodides Indrek Kalvet, a Karl J. Bonney, a and Franziska Schoenebeck a * a Institute of Organic
C H Activation of Cp* Ligand Coordinated to Ruthenium. Center: Synthesis and Reactivity of a Thiolate-Bridged
 Supporting Information C H Activation of Cp* Ligand Coordinated to Ruthenium Center: Synthesis and Reactivity of a Thiolate-Bridged Diruthenium Complex Featuring Fulvene-like Cp* Ligand Xiaoxiao Ji, Dawei
Supporting Information C H Activation of Cp* Ligand Coordinated to Ruthenium Center: Synthesis and Reactivity of a Thiolate-Bridged Diruthenium Complex Featuring Fulvene-like Cp* Ligand Xiaoxiao Ji, Dawei
Electronic Supporting Information
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2018 Electronic Supporting Information robing steric influences on electrophilic phosphonium
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2018 Electronic Supporting Information robing steric influences on electrophilic phosphonium
Every set of first-order formulas is equivalent to an independent set
 Every set of first-order formulas is equivalent to an independent set May 6, 2008 Abstract A set of first-order formulas, whatever the cardinality of the set of symbols, is equivalent to an independent
Every set of first-order formulas is equivalent to an independent set May 6, 2008 Abstract A set of first-order formulas, whatever the cardinality of the set of symbols, is equivalent to an independent
Extremely Strong Halogen Bond. The Case of a Double-Charge-Assisted Halogen Bridge
 Supporting Information Extremely Strong Halogen Bond. The Case of a Double-Charge-Assisted Halogen Bridge Małgorzata Domagała*, Aneta Lutyńska, Marcin Palusiak Theoretical and Structural Chemistry Group,
Supporting Information Extremely Strong Halogen Bond. The Case of a Double-Charge-Assisted Halogen Bridge Małgorzata Domagała*, Aneta Lutyńska, Marcin Palusiak Theoretical and Structural Chemistry Group,
Supporting Information
 Supporting Information Mitochondria-Targeting Polydopamine Nanocomposites as Chemophotothermal Therapeutics for Cancer Zhuo Wang *,, Yuzhi Chen, Hui Zhang, Yawen Li, Yufan Ma, Jia Huang, Xiaolei Liu, Fang
Supporting Information Mitochondria-Targeting Polydopamine Nanocomposites as Chemophotothermal Therapeutics for Cancer Zhuo Wang *,, Yuzhi Chen, Hui Zhang, Yawen Li, Yufan Ma, Jia Huang, Xiaolei Liu, Fang
Uncovering the impact of capsule shaped amine-type ligands on. Am(III)/Eu(III) separation
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 Uncovering the impact of capsule shaped amine-type ligands on Am(III)/Eu(III) separation
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 Uncovering the impact of capsule shaped amine-type ligands on Am(III)/Eu(III) separation
Supporting Information
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2018 Supporting Information Crotonols A and B, Two Rare Tigliane Diterpenoid
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2018 Supporting Information Crotonols A and B, Two Rare Tigliane Diterpenoid
SUPPLEMENTARY INFORMATION
 Sensitivity of [Ru(phen) 2 dppz] 2+ Light Switch Emission to Ionic Strength, Temperature, and DNA Sequence and Conformation Andrew W. McKinley, Per Lincoln and Eimer M. Tuite* SUPPLEMENTARY INFORMATION
Sensitivity of [Ru(phen) 2 dppz] 2+ Light Switch Emission to Ionic Strength, Temperature, and DNA Sequence and Conformation Andrew W. McKinley, Per Lincoln and Eimer M. Tuite* SUPPLEMENTARY INFORMATION
Supplementary Materials: A Preliminary Link between Hydroxylated Metabolites of Polychlorinated Biphenyls and Free Thyroxin in Humans
 S1 of S11 Supplementary Materials: A Preliminary Link between Hydroxylated Metabolites of Polychlorinated Biphenyls and Free Thyroxin in Humans Eveline Dirinck, Alin C. Dirtu, Govindan Malarvannan, Adrian
S1 of S11 Supplementary Materials: A Preliminary Link between Hydroxylated Metabolites of Polychlorinated Biphenyls and Free Thyroxin in Humans Eveline Dirinck, Alin C. Dirtu, Govindan Malarvannan, Adrian
Review Test 3. MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Review Test MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. Find the exact value of the expression. 1) sin - 11π 1 1) + - + - - ) sin 11π 1 ) ( -
Review Test MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. Find the exact value of the expression. 1) sin - 11π 1 1) + - + - - ) sin 11π 1 ) ( -
Supporting Information
 Supporting Information Aqueous Biphasic Systems Composed of a Water Stable Ionic Liquid + Carbohydrates and their Applications Mara G. Freire a,b*, Cláudia L. S. Louros a, Luís Paulo N. Rebelo b and João
Supporting Information Aqueous Biphasic Systems Composed of a Water Stable Ionic Liquid + Carbohydrates and their Applications Mara G. Freire a,b*, Cláudia L. S. Louros a, Luís Paulo N. Rebelo b and João
Enantioselective Organocatalytic Michael Addition of Isorhodanines. to α, β-unsaturated Aldehydes
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2016 Enantioselective Organocatalytic Michael Addition of Isorhodanines to α,
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2016 Enantioselective Organocatalytic Michael Addition of Isorhodanines to α,
CRASH COURSE IN PRECALCULUS
 CRASH COURSE IN PRECALCULUS Shiah-Sen Wang The graphs are prepared by Chien-Lun Lai Based on : Precalculus: Mathematics for Calculus by J. Stuwart, L. Redin & S. Watson, 6th edition, 01, Brooks/Cole Chapter
CRASH COURSE IN PRECALCULUS Shiah-Sen Wang The graphs are prepared by Chien-Lun Lai Based on : Precalculus: Mathematics for Calculus by J. Stuwart, L. Redin & S. Watson, 6th edition, 01, Brooks/Cole Chapter
Supporting Information: Design principles for α-tocopherol analogues
 Supplementary Material for Organic & Biomolecular Chemistry This journal is The Royal Society of Chemistry 2005 Supporting Information: Design principles for α-tocopherol analogues David Shanks,* a Håkan
Supplementary Material for Organic & Biomolecular Chemistry This journal is The Royal Society of Chemistry 2005 Supporting Information: Design principles for α-tocopherol analogues David Shanks,* a Håkan
Problem Set 9 Solutions. θ + 1. θ 2 + cotθ ( ) sinθ e iφ is an eigenfunction of the ˆ L 2 operator. / θ 2. φ 2. sin 2 θ φ 2. ( ) = e iφ. = e iφ cosθ.
 Chemistry 362 Dr Jean M Standard Problem Set 9 Solutions The ˆ L 2 operator is defined as Verify that the angular wavefunction Y θ,φ) Also verify that the eigenvalue is given by 2! 2 & L ˆ 2! 2 2 θ 2 +
Chemistry 362 Dr Jean M Standard Problem Set 9 Solutions The ˆ L 2 operator is defined as Verify that the angular wavefunction Y θ,φ) Also verify that the eigenvalue is given by 2! 2 & L ˆ 2! 2 2 θ 2 +
Cycloaddition of Homochiral Dihydroimidazoles: A 1,3-Dipolar Cycloaddition Route to Optically Active Pyrrolo[1,2-a]imidazoles
![Cycloaddition of Homochiral Dihydroimidazoles: A 1,3-Dipolar Cycloaddition Route to Optically Active Pyrrolo[1,2-a]imidazoles Cycloaddition of Homochiral Dihydroimidazoles: A 1,3-Dipolar Cycloaddition Route to Optically Active Pyrrolo[1,2-a]imidazoles](/thumbs/89/100111450.jpg) X-Ray crystallographic data tables for paper: Supplementary Material (ESI) for Organic & Biomolecular Chemistry Cycloaddition of Homochiral Dihydroimidazoles: A 1,3-Dipolar Cycloaddition Route to Optically
X-Ray crystallographic data tables for paper: Supplementary Material (ESI) for Organic & Biomolecular Chemistry Cycloaddition of Homochiral Dihydroimidazoles: A 1,3-Dipolar Cycloaddition Route to Optically
Photo-Induced Self-Assembly of Pt(II)-Linked Rings and Cages via the Photolabilization of a Pt(II) Pyridine Bond
 Photo-Induced Self-Assembly of Pt(II)-Linked Rings and Cages via the Photolabilization of a Pt(II) Pyridine Bond Ken-ichi Yamashita, Kei-ichi Sato, Masaki Kawano and Makoto Fujita* Contents; Figure S1.
Photo-Induced Self-Assembly of Pt(II)-Linked Rings and Cages via the Photolabilization of a Pt(II) Pyridine Bond Ken-ichi Yamashita, Kei-ichi Sato, Masaki Kawano and Makoto Fujita* Contents; Figure S1.
SUPPLEMENTARY MATERIAL. In Situ Spectroelectrochemical Investigations of Ru II Complexes with Bispyrazolyl Methane Triarylamine Ligands
 10.1071/CH16555_AC CSIRO 2017 Australian Journal of Chemistry 2017, 70(5), 546-555 SUPPLEMENTARY MATERIAL In Situ Spectroelectrochemical Investigations of Ru II Complexes with Bispyrazolyl Methane Triarylamine
10.1071/CH16555_AC CSIRO 2017 Australian Journal of Chemistry 2017, 70(5), 546-555 SUPPLEMENTARY MATERIAL In Situ Spectroelectrochemical Investigations of Ru II Complexes with Bispyrazolyl Methane Triarylamine
Second Order RLC Filters
 ECEN 60 Circuits/Electronics Spring 007-0-07 P. Mathys Second Order RLC Filters RLC Lowpass Filter A passive RLC lowpass filter (LPF) circuit is shown in the following schematic. R L C v O (t) Using phasor
ECEN 60 Circuits/Electronics Spring 007-0-07 P. Mathys Second Order RLC Filters RLC Lowpass Filter A passive RLC lowpass filter (LPF) circuit is shown in the following schematic. R L C v O (t) Using phasor
Quantum dot sensitized solar cells with efficiency over 12% based on tetraethyl orthosilicate additive in polysulfide electrolyte
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supplementary Information (SI) Quantum dot sensitized solar cells with
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supplementary Information (SI) Quantum dot sensitized solar cells with
IV. ANHANG 179. Anhang 178
 Anhang 178 IV. ANHANG 179 1. Röntgenstrukturanalysen (Tabellen) 179 1.1. Diastereomer A (Diplomarbeit) 179 1.2. Diastereomer B (Diplomarbeit) 186 1.3. Aldoladdukt 5A 193 1.4. Aldoladdukt 13A 200 1.5. Aldoladdukt
Anhang 178 IV. ANHANG 179 1. Röntgenstrukturanalysen (Tabellen) 179 1.1. Diastereomer A (Diplomarbeit) 179 1.2. Diastereomer B (Diplomarbeit) 186 1.3. Aldoladdukt 5A 193 1.4. Aldoladdukt 13A 200 1.5. Aldoladdukt
Supporting Information
 Supporting Information Prediction of Novel High-Pressure Structures of Magnesium Niobium Dihydride Chuanzhao Zhang,,, Guoliang Sun, Jingjing Wang, Cheng Lu,*,, Yuanyuan Jin, Xiaoyu Kuang,*, and Andreas
Supporting Information Prediction of Novel High-Pressure Structures of Magnesium Niobium Dihydride Chuanzhao Zhang,,, Guoliang Sun, Jingjing Wang, Cheng Lu,*,, Yuanyuan Jin, Xiaoyu Kuang,*, and Andreas
Pyrrolo[2,3-d:5,4-d']bisthiazoles: Alternate Synthetic Routes and a Comparative Study to Analogous Fused-ring Bithiophenes
![Pyrrolo[2,3-d:5,4-d']bisthiazoles: Alternate Synthetic Routes and a Comparative Study to Analogous Fused-ring Bithiophenes Pyrrolo[2,3-d:5,4-d']bisthiazoles: Alternate Synthetic Routes and a Comparative Study to Analogous Fused-ring Bithiophenes](/thumbs/92/108867714.jpg) SUPPORTING INFORMATION Pyrrolo[2,3-d:5,4-d']bisthiazoles: Alternate Synthetic Routes and a Comparative Study to Analogous Fused-ring Bithiophenes Eric J. Uzelac, Casey B. McCausland, and Seth C. Rasmussen*
SUPPORTING INFORMATION Pyrrolo[2,3-d:5,4-d']bisthiazoles: Alternate Synthetic Routes and a Comparative Study to Analogous Fused-ring Bithiophenes Eric J. Uzelac, Casey B. McCausland, and Seth C. Rasmussen*
Supporting Information
 SUPPORTING INFORMATION FOR: Trialkylstibine complexes of boron, aluminium, gallium and indium trihalides: synthesis, properties and bonding Victoria K. Greenacre, William Levason and Gillian Reid Chemistry,
SUPPORTING INFORMATION FOR: Trialkylstibine complexes of boron, aluminium, gallium and indium trihalides: synthesis, properties and bonding Victoria K. Greenacre, William Levason and Gillian Reid Chemistry,
A facile and general route to 3-((trifluoromethyl)thio)benzofurans and 3-((trifluoromethyl)thio)benzothiophenes
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 A facile and general route to 3-((trifluoromethyl)thio)benzofurans and 3-((trifluoromethyl)thio)benzothiophenes
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 A facile and general route to 3-((trifluoromethyl)thio)benzofurans and 3-((trifluoromethyl)thio)benzothiophenes
January 22, University of Minnesota, Minneapolis, Minnesota , USA
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 S-1 ELECTRONIC SUPPLEMENTARY INFORMATION January 22, 2017 Reaction of SO 2 with
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 S-1 ELECTRONIC SUPPLEMENTARY INFORMATION January 22, 2017 Reaction of SO 2 with
derivation of the Laplacian from rectangular to spherical coordinates
 derivation of the Laplacian from rectangular to spherical coordinates swapnizzle 03-03- :5:43 We begin by recognizing the familiar conversion from rectangular to spherical coordinates (note that φ is used
derivation of the Laplacian from rectangular to spherical coordinates swapnizzle 03-03- :5:43 We begin by recognizing the familiar conversion from rectangular to spherical coordinates (note that φ is used
SCITECH Volume 13, Issue 2 RESEARCH ORGANISATION Published online: March 29, 2018
 Journal of rogressive Research in Mathematics(JRM) ISSN: 2395-028 SCITECH Volume 3, Issue 2 RESEARCH ORGANISATION ublished online: March 29, 208 Journal of rogressive Research in Mathematics www.scitecresearch.com/journals
Journal of rogressive Research in Mathematics(JRM) ISSN: 2395-028 SCITECH Volume 3, Issue 2 RESEARCH ORGANISATION ublished online: March 29, 208 Journal of rogressive Research in Mathematics www.scitecresearch.com/journals
Homework 8 Model Solution Section
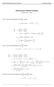 MATH 004 Homework Solution Homework 8 Model Solution Section 14.5 14.6. 14.5. Use the Chain Rule to find dz where z cosx + 4y), x 5t 4, y 1 t. dz dx + dy y sinx + 4y)0t + 4) sinx + 4y) 1t ) 0t + 4t ) sinx
MATH 004 Homework Solution Homework 8 Model Solution Section 14.5 14.6. 14.5. Use the Chain Rule to find dz where z cosx + 4y), x 5t 4, y 1 t. dz dx + dy y sinx + 4y)0t + 4) sinx + 4y) 1t ) 0t + 4t ) sinx
Multifunctinality and Crystal Dynamics of Highly Stable Porous Metal-Organic Framework [Zn 4 O(NTB) 2 ]
![Multifunctinality and Crystal Dynamics of Highly Stable Porous Metal-Organic Framework [Zn 4 O(NTB) 2 ] Multifunctinality and Crystal Dynamics of Highly Stable Porous Metal-Organic Framework [Zn 4 O(NTB) 2 ]](/thumbs/92/109256432.jpg) Supporting Information Multifunctinality and Crystal Dynamics of Highly Stable Porous Metal-Organic Framework [Zn 4 O(NTB) 2 ] Eun Young Lee, Seung Yeon Jang, and Myunghyun Paik Suh* School of Chemistry,
Supporting Information Multifunctinality and Crystal Dynamics of Highly Stable Porous Metal-Organic Framework [Zn 4 O(NTB) 2 ] Eun Young Lee, Seung Yeon Jang, and Myunghyun Paik Suh* School of Chemistry,
Supporting Information To. Microhydration of caesium compounds: Journal of Molecular Modeling
 Supporting Information To Microhydration of caesium compounds: Cs, CsOH, CsI and Cs 2 I 2 complexes with one to three H 2 O molecules of nuclear safety interest Journal of Molecular Modeling Mária Sudolská
Supporting Information To Microhydration of caesium compounds: Cs, CsOH, CsI and Cs 2 I 2 complexes with one to three H 2 O molecules of nuclear safety interest Journal of Molecular Modeling Mária Sudolská
Table S1. Summary of data collections and structure refinements for crystals 1Rb-1h, 1Rb-2h, and 1Rb-4h.
 Supporting Information [NH 3 CH 3 ] [In SbS 9 SH]: A novel methylamine-directed indium thioantimonate with Rb + ion-exchange property Kai-Yao Wang a,b, Mei-Ling Feng a, Jian-Rong Li a and Xiao-Ying Huang
Supporting Information [NH 3 CH 3 ] [In SbS 9 SH]: A novel methylamine-directed indium thioantimonate with Rb + ion-exchange property Kai-Yao Wang a,b, Mei-Ling Feng a, Jian-Rong Li a and Xiao-Ying Huang
Tridiagonal matrices. Gérard MEURANT. October, 2008
 Tridiagonal matrices Gérard MEURANT October, 2008 1 Similarity 2 Cholesy factorizations 3 Eigenvalues 4 Inverse Similarity Let α 1 ω 1 β 1 α 2 ω 2 T =......... β 2 α 1 ω 1 β 1 α and β i ω i, i = 1,...,
Tridiagonal matrices Gérard MEURANT October, 2008 1 Similarity 2 Cholesy factorizations 3 Eigenvalues 4 Inverse Similarity Let α 1 ω 1 β 1 α 2 ω 2 T =......... β 2 α 1 ω 1 β 1 α and β i ω i, i = 1,...,
Supporting Information
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2017 Supporting Information Phosphorescent Pt(II) complexes bearing a monoanionic C^N^N luminophore
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2017 Supporting Information Phosphorescent Pt(II) complexes bearing a monoanionic C^N^N luminophore
Bayesian statistics. DS GA 1002 Probability and Statistics for Data Science.
 Bayesian statistics DS GA 1002 Probability and Statistics for Data Science http://www.cims.nyu.edu/~cfgranda/pages/dsga1002_fall17 Carlos Fernandez-Granda Frequentist vs Bayesian statistics In frequentist
Bayesian statistics DS GA 1002 Probability and Statistics for Data Science http://www.cims.nyu.edu/~cfgranda/pages/dsga1002_fall17 Carlos Fernandez-Granda Frequentist vs Bayesian statistics In frequentist
Electronic structure and spectroscopy of HBr and HBr +
 Electronic structure and spectroscopy of HBr and HBr + Gabriel J. Vázquez 1 H. P. Liebermann 2 H. Lefebvre Brion 3 1 Universidad Nacional Autónoma de México Cuernavaca México 2 Universität Wuppertal Wuppertal
Electronic structure and spectroscopy of HBr and HBr + Gabriel J. Vázquez 1 H. P. Liebermann 2 H. Lefebvre Brion 3 1 Universidad Nacional Autónoma de México Cuernavaca México 2 Universität Wuppertal Wuppertal
w o = R 1 p. (1) R = p =. = 1
 Πανεπιστήµιο Κρήτης - Τµήµα Επιστήµης Υπολογιστών ΗΥ-570: Στατιστική Επεξεργασία Σήµατος 205 ιδάσκων : Α. Μουχτάρης Τριτη Σειρά Ασκήσεων Λύσεις Ασκηση 3. 5.2 (a) From the Wiener-Hopf equation we have:
Πανεπιστήµιο Κρήτης - Τµήµα Επιστήµης Υπολογιστών ΗΥ-570: Στατιστική Επεξεργασία Σήµατος 205 ιδάσκων : Α. Μουχτάρης Τριτη Σειρά Ασκήσεων Λύσεις Ασκηση 3. 5.2 (a) From the Wiener-Hopf equation we have:
EE512: Error Control Coding
 EE512: Error Control Coding Solution for Assignment on Finite Fields February 16, 2007 1. (a) Addition and Multiplication tables for GF (5) and GF (7) are shown in Tables 1 and 2. + 0 1 2 3 4 0 0 1 2 3
EE512: Error Control Coding Solution for Assignment on Finite Fields February 16, 2007 1. (a) Addition and Multiplication tables for GF (5) and GF (7) are shown in Tables 1 and 2. + 0 1 2 3 4 0 0 1 2 3
Monolithic Crystal Filters (M.C.F.)
 Monolithic Crystal Filters (M.C.F.) MCF (MONOLITHIC CRYSTAL FILTER) features high quality quartz resonators such as sharp cutoff characteristics, low loss, good inter-modulation and high stability over
Monolithic Crystal Filters (M.C.F.) MCF (MONOLITHIC CRYSTAL FILTER) features high quality quartz resonators such as sharp cutoff characteristics, low loss, good inter-modulation and high stability over
College of Life Science, Dalian Nationalities University, Dalian , PR China.
 Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2018 Postsynthetic modification
Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2018 Postsynthetic modification
Copper(II) complexes of salicylaldehydes and 2 hydroxyphenones: Synthesis, Structure,
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Copper(II) complexes of salicylaldehydes and 2 hydroxyphenones: Synthesis, Structure, Thermal
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Copper(II) complexes of salicylaldehydes and 2 hydroxyphenones: Synthesis, Structure, Thermal
Selecting Critical Properties of Terpenes and Terpenoids through Group-Contribution Methods and Equations of State
 Supporting Information Selecting Critical Properties of Terpenes and Terpenoids through Group-Contribution Methods and Equations of State Mónia A. R. Martins, 1,2 Pedro J. Carvalho, 1 André M. Palma, 1
Supporting Information Selecting Critical Properties of Terpenes and Terpenoids through Group-Contribution Methods and Equations of State Mónia A. R. Martins, 1,2 Pedro J. Carvalho, 1 André M. Palma, 1
Supporting Information. Asymmetric Binary-acid Catalysis with Chiral. Phosphoric Acid and MgF 2 : Catalytic
 Supporting Information Asymmetric Binary-acid Catalysis with Chiral Phosphoric Acid and MgF 2 : Catalytic Enantioselective Friedel-Crafts Reactions of β,γ- Unsaturated-α-Ketoesters Jian Lv, Xin Li, Long
Supporting Information Asymmetric Binary-acid Catalysis with Chiral Phosphoric Acid and MgF 2 : Catalytic Enantioselective Friedel-Crafts Reactions of β,γ- Unsaturated-α-Ketoesters Jian Lv, Xin Li, Long
ΠΕΡΙΕΧΟΜΕΝΑ. Κεφάλαιο 1: Κεφάλαιο 2: Κεφάλαιο 3:
 4 Πρόλογος Η παρούσα διπλωµατική εργασία µε τίτλο «ιερεύνηση χωρικής κατανοµής µετεωρολογικών µεταβλητών. Εφαρµογή στον ελληνικό χώρο», ανατέθηκε από το ιεπιστηµονικό ιατµηµατικό Πρόγραµµα Μεταπτυχιακών
4 Πρόλογος Η παρούσα διπλωµατική εργασία µε τίτλο «ιερεύνηση χωρικής κατανοµής µετεωρολογικών µεταβλητών. Εφαρµογή στον ελληνικό χώρο», ανατέθηκε από το ιεπιστηµονικό ιατµηµατικό Πρόγραµµα Μεταπτυχιακών
DuPont Suva 95 Refrigerant
 Technical Information T-95 SI DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Technical Information T-95 SI DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Problem 3.1 Vector A starts at point (1, 1, 3) and ends at point (2, 1,0). Find a unit vector in the direction of A. Solution: A = 1+9 = 3.
 Problem 3.1 Vector A starts at point (1, 1, 3) and ends at point (, 1,0). Find a unit vector in the direction of A. Solution: A = ˆx( 1)+ŷ( 1 ( 1))+ẑ(0 ( 3)) = ˆx+ẑ3, A = 1+9 = 3.16, â = A A = ˆx+ẑ3 3.16
Problem 3.1 Vector A starts at point (1, 1, 3) and ends at point (, 1,0). Find a unit vector in the direction of A. Solution: A = ˆx( 1)+ŷ( 1 ( 1))+ẑ(0 ( 3)) = ˆx+ẑ3, A = 1+9 = 3.16, â = A A = ˆx+ẑ3 3.16
SMD Power Inductor-VLH
 SMD Power Inductor-VH Dimensions Unit: mm Type A B C E F H I J 252010 2.5±0.2 2.0±0.2 1.0max. 0.4±0.2 1.0min. 2.1 0.90 0.8 252012 2.5±0.2 2.0±0.2 1.2max. 0.4±0.2 1.0min. 2.1 0.90 0.8 321618C 3.2±0.3 1.6±0.2
SMD Power Inductor-VH Dimensions Unit: mm Type A B C E F H I J 252010 2.5±0.2 2.0±0.2 1.0max. 0.4±0.2 1.0min. 2.1 0.90 0.8 252012 2.5±0.2 2.0±0.2 1.2max. 0.4±0.2 1.0min. 2.1 0.90 0.8 321618C 3.2±0.3 1.6±0.2
DuPont Suva 95 Refrigerant
 Technical Information T-95 ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Technical Information T-95 ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Written Examination. Antennas and Propagation (AA ) April 26, 2017.
 Written Examination Antennas and Propagation (AA. 6-7) April 6, 7. Problem ( points) Let us consider a wire antenna as in Fig. characterized by a z-oriented linear filamentary current I(z) = I cos(kz)ẑ
Written Examination Antennas and Propagation (AA. 6-7) April 6, 7. Problem ( points) Let us consider a wire antenna as in Fig. characterized by a z-oriented linear filamentary current I(z) = I cos(kz)ẑ
Key Laboratory of Functional Materials and Devices for Special Environments, Xinjiang Technical
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry C. This journal is The Royal Society of Chemistry 2016 KPb 2 (PO 3 ) 5 : A Novel Nonlinear Optical Lead Polyphosphate with Short
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry C. This journal is The Royal Society of Chemistry 2016 KPb 2 (PO 3 ) 5 : A Novel Nonlinear Optical Lead Polyphosphate with Short
DATA SHEET Surface mount NTC thermistors. BCcomponents
 DATA SHEET 2322 615 1... Surface mount N thermistors Supersedes data of 17th May 1999 File under BCcomponents, BC02 2001 Mar 27 FEATURES High sensitivity High accuracy over a wide temperature range Taped
DATA SHEET 2322 615 1... Surface mount N thermistors Supersedes data of 17th May 1999 File under BCcomponents, BC02 2001 Mar 27 FEATURES High sensitivity High accuracy over a wide temperature range Taped
Supporting Information. Crown Ether Complexes of Actinyls: A Computational Assessment of
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2017 Supporting Information Crown Ether Complexes of Actinyls: A Computational Assessment
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2017 Supporting Information Crown Ether Complexes of Actinyls: A Computational Assessment
Supporting information. An unusual bifunctional Tb-MOF for highly sensing of Ba 2+ ions and remarkable selectivities of CO 2 /N 2 and CO 2 /CH 4
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Supporting information An unusual bifunctional Tb-MOF for highly sensing
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Supporting information An unusual bifunctional Tb-MOF for highly sensing
Comparison of carbon-sulfur and carbon-amine bond in therapeutic drug: -S-aromatic heterocyclic podophyllum derivatives display antitumor activity
 Comparison of carbon-sulfur and carbon-amine bond in therapeutic drug: -S-aromatic heterocyclic podophyllum derivatives display antitumor activity Jian-Long Li 1,a, Wei Zhao 1,a, Chen Zhou 1,a, Ya-Xuan
Comparison of carbon-sulfur and carbon-amine bond in therapeutic drug: -S-aromatic heterocyclic podophyllum derivatives display antitumor activity Jian-Long Li 1,a, Wei Zhao 1,a, Chen Zhou 1,a, Ya-Xuan
Matrices and Determinants
 Matrices and Determinants SUBJECTIVE PROBLEMS: Q 1. For what value of k do the following system of equations possess a non-trivial (i.e., not all zero) solution over the set of rationals Q? x + ky + 3z
Matrices and Determinants SUBJECTIVE PROBLEMS: Q 1. For what value of k do the following system of equations possess a non-trivial (i.e., not all zero) solution over the set of rationals Q? x + ky + 3z
ΕΠΑΝΑΛΗΨΗ ΨΕΥΔΟΛΕΞΕΩΝ ΑΠΟ ΠΑΙΔΙΑ ΜΕ ΕΙΔΙΚΗ ΓΛΩΣΣΙΚΗ ΔΙΑΤΑΡΑΧΗ ΚΑΙ ΠΑΙΔΙΑ ΤΥΠΙΚΗΣ ΑΝΑΠΤΥΞΗΣ
 Σχολή Επιστημών Υγείας Πτυχιακή εργασία ΕΠΑΝΑΛΗΨΗ ΨΕΥΔΟΛΕΞΕΩΝ ΑΠΟ ΠΑΙΔΙΑ ΜΕ ΕΙΔΙΚΗ ΓΛΩΣΣΙΚΗ ΔΙΑΤΑΡΑΧΗ ΚΑΙ ΠΑΙΔΙΑ ΤΥΠΙΚΗΣ ΑΝΑΠΤΥΞΗΣ Άντρια Πολυκάρπου Λεμεσός, Μάιος 2017 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ
Σχολή Επιστημών Υγείας Πτυχιακή εργασία ΕΠΑΝΑΛΗΨΗ ΨΕΥΔΟΛΕΞΕΩΝ ΑΠΟ ΠΑΙΔΙΑ ΜΕ ΕΙΔΙΚΗ ΓΛΩΣΣΙΚΗ ΔΙΑΤΑΡΑΧΗ ΚΑΙ ΠΑΙΔΙΑ ΤΥΠΙΚΗΣ ΑΝΑΠΤΥΞΗΣ Άντρια Πολυκάρπου Λεμεσός, Μάιος 2017 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ
Table S1 Selected bond lengths [Å] and angles [ ] for complexes 1 8. Complex 1. Complex 2. Complex 3. Complex 4. Complex 5.
![Table S1 Selected bond lengths [Å] and angles [ ] for complexes 1 8. Complex 1. Complex 2. Complex 3. Complex 4. Complex 5. Table S1 Selected bond lengths [Å] and angles [ ] for complexes 1 8. Complex 1. Complex 2. Complex 3. Complex 4. Complex 5.](/thumbs/65/53614116.jpg) Table S1 Selected bond lengths [Å] and angles [ ] for complexes 1 8. Complex 1 Zn(1) O(1) 1.969(2) Zn(1) O(5) 1.989(3) Zn(1) O(6) 1.930(3) Zn(1) O(4) a 1.954(4) Zn(1) O(3) a 2.519(25) O(6) Zn(1) O(4) a
Table S1 Selected bond lengths [Å] and angles [ ] for complexes 1 8. Complex 1 Zn(1) O(1) 1.969(2) Zn(1) O(5) 1.989(3) Zn(1) O(6) 1.930(3) Zn(1) O(4) a 1.954(4) Zn(1) O(3) a 2.519(25) O(6) Zn(1) O(4) a
SOLUTIONS TO MATH38181 EXTREME VALUES AND FINANCIAL RISK EXAM
 SOLUTIONS TO MATH38181 EXTREME VALUES AND FINANCIAL RISK EXAM Solutions to Question 1 a) The cumulative distribution function of T conditional on N n is Pr T t N n) Pr max X 1,..., X N ) t N n) Pr max
SOLUTIONS TO MATH38181 EXTREME VALUES AND FINANCIAL RISK EXAM Solutions to Question 1 a) The cumulative distribution function of T conditional on N n is Pr T t N n) Pr max X 1,..., X N ) t N n) Pr max
