Pof moist air and uses these properties to analyze conditions and
|
|
|
- Ἀπολλωνία Καραμήτσος
- 7 χρόνια πριν
- Προβολές:
Transcript
1 CHAPTER 6 PSYCHROMETRICS Composition of Dry and Moist Air United States Standard Atmosphere Thermodynamic Properties of Moist Air Thermodynamic Properties of Water at Saturation Humidity Parameters Humidity Parameters Involving Saturation Perfect Gas Relationships for Dry and Moist Air Thermodynamic Wet-Bulb Temperature and Dew-Point Temperature Numerical Calculation of Moist Air Properties Exact Relations for Computing W s and φ Moist Air Property Tables for Standard Pressure Psychrometric Charts Typical Air-Conditioning Processes Transport Properties of Moist Air Air, Water, and Steam Properties Symbols SYCHROMETRICS deals with thermodynamic properties Pof moist air and uses these properties to analyze conditions and processes involving moist air. Hyland and Wexler (1983a, 1983b) developed formulas for thermodynamic properties of moist air and water. Perfect gas relations can be used in most air-conditioning problems instead of these formulas. Threlkeld (1970) showed that errors are less than 0.7% in calculating humidity ratio, enthalpy, and specific volume of saturated air at standard atmospheric pressure for a temperature range of 50 to 50 C. Furthermore, these errors decrease with decreasing pressure. This chapter discusses perfect gas relations and describes their use in common air-conditioning problems. The formulas developed by Hyland and Wexler (1983a) may be used where greater precision is required. COMPOSITION OF DRY AND MOIST AIR Atmospheric air contains many gaseous components as well as water vapor and miscellaneous contaminants (e.g., smoke, pollen, and gaseous pollutants not normally present in free air far from pollution sources). Dry air exists when all water vapor and contaminants have been removed from atmospheric air. The composition of dry air is relatively constant, but small variations in the amounts of individual components occur with time, geographic location, and altitude. Harrison (1965) lists the approximate percentage composition of dry air by volume as: nitrogen, ; oxygen, ; argon, 0.934; carbon dioxide, ; neon, ; helium, ; methane, ; sulfur dioxide, 0 to ; hydrogen, ; and minor components such as krypton, xenon, and ozone, The relative molecular mass of all components, for dry air is , based on the carbon-12 scale (Harrison 1965). The gas constant for dry air, based on the carbon-12 scale, is: R a = / = J/(kg K) (1) Moist air is a binary (or two-component) mixture of dry air and water vapor. The amount of water vapor in moist air varies from zero (dry air) to a maximum that depends on temperature and pressure. The latter condition refers to saturation, a state of neutral equilibrium between moist air and the condensed water phase (liquid or solid). Unless otherwise stated, saturation refers to a flat interface surface between the moist air and the condensed phase. The relative molecular mass of water is on the carbon-12 scale. The gas constant for water vapor is: The preparation of this chapter is assigned to TC 1.1, Thermodynamics and Psychrometrics. R a = / = J/(kg K) (2) UNITED STATES STANDARD ATMOSPHERE The temperature and barometric pressure of atmospheric air vary considerably with altitude as well as with local geographic and weather conditions. The standard atmosphere gives a standard of reference for estimating properties at various altitudes. At sea level, standard temperature is 15 C; standard barometric pressure is kpa. The temperature is assumed to decrease linearly with increasing altitude throughout the troposphere (lower atmosphere), and to be constant in the lower reaches of the stratosphere. The lower atmosphere is assumed to consist of dry air that behaves as a perfect gas. Gravity is also assumed constant at the standard value, m/s 2. Table 1 summarizes property data for altitudes to m. The values in Table 1 may be calculated from the equation Table 1 p = ( Z) (3) Standard Atmospheric Data for Altitudes to m Altitude, m Temperature, C Pressure, kpa Data adapted from NASA (1976).
2 ASHRAE Fundamentals Handbook (SI) The equation for temperature as a function of altitude is given as: t = Z where Z = altitude, m p = barometric pressure, kpa t = temperature, K Equations (3) and (4) are accurate from 5000 m to m. For higher altitudes, comprehensive tables of barometric pressure and other physical properties of the standard atmosphere can be found in NASA (1976 U.S. Standard atmosphere). THERMODYNAMIC PROPERTIES OF MOIST AIR Table 2, developed from formulas by Hyland and Wexler (1983a, 1983b), shows values of thermodynamic properties based on the thermodynamic temperature scale. This ideal scale differs slightly from practical temperature scales used for physical measurements. For example, the standard boiling point for water (at kpa) occurs at C on this scale rather than at the traditional value of 100 C. Most measurements are currently based on the International Practical Temperature Scale of 1990 (IPTS-90). The following paragraphs briefly describe each column of Table 2. t = Celsius temperature, based on thermodynamic temperature scale and expressed relative to absolute temperature T in kelvin (K) by the relation: T = t W s = humidity ratio at saturation, condition at which gaseous phase (moist air) exists in equilibrium with condensed phase (liquid or solid) at given temperature and pressure (standard atmospheric pressure). At given values of temperature and pressure, humidity ratio W can have any value from zero to W s. v a = specific volume of dry air, m 3 /kg v as = v s v a, difference between volume of moist air at saturation, per kilogram of dry air, and specific volume of dry air itself, m 3 /kg of dry air, at same pressure and temperature. v s = volume of moist air at saturation per kilogram of dry air, m 3 /kg of dry air. h a = specific enthalpy of dry air, kj/kg of dry air. Specific enthalpy of dry air has been assigned a value of zero at 0 C and standard atmospheric pressure in Table 2. h as = h s h a, difference between enthalpy of moist air at saturation, per kilogram of dry air, and specific enthalpy of dry air itself, kj/kg of dry air, at same pressure and temperature. h s = enthalpy of moist air at saturation of dry air, kj/kg of dry air. h w = specific enthalpy of condensed water (liquid or solid) in equilibrium with saturated air at specified temperature and pressure, kj/kg of water. Specific enthalpy of liquid water is assigned a value of zero at its triple point (0.01 C) and saturation pressure. Note that h w is greater than the steam-table enthalpy of saturated pure condensed phase by the amount of enthalpy increase governed by the pressure increase from saturation pressure to kpa, plus influences from presence of air. s a = specific entropy of dry air, kj/(kg K). In Table 2, specific entropy of dry air has been assigned a value of zero at 0 C and standard atmospheric pressure. s as = s s s a, difference between entropy of moist air at saturation, per kilogram of dry air, and specific entropy of dry air itself, kj/(kg K), at same pressure and temperature. s s = entropy of moist air at saturation per kilogram of dry air, kj/(kg K). s w = specific entropy per kilogram of condensed water (liquid or solid) in equilibrium with saturated air, kj/(kg K); s w differs from entropy of pure water at saturation pressure, similar to h w. p s = vapor pressure of water in saturated moist air, kpa. Pressure p s differs negligibly from saturation vapor pressure of pure water p ws at least for conditions shown. Consequently, values of p s can (4) be used at same pressure and temperature in equations where p ws appears. Pressure p s is defined as p s = x ws p, where x ws is mole fraction of water vapor in moist air saturated with water at temperature t and pressure p, and where p is total barometric pressure of moist air. THERMODYNAMIC PROPERTIES OF WATER AT SATURATION Table 3 shows thermodynamic properties of water at saturation for temperatures from 60 to 200 C, calculated by the formulations described by Hyland and Wexler (1983b). Symbols in the table follow standard steam table nomenclature. These properties are based on the thermodynamic temperature scale. The enthalpy and entropy of saturated liquid water are both assigned the value zero at the triple point, 0.01 C. Between the triple-point and critical-point temperatures of water, two states liquid and vapor may coexist in equilibrium. These states are called saturated liquid and saturated vapor. In determining a number of moist air properties, principally the saturation humidity ratio, the water vapor saturation pressure is required. Values may be obtained from Table 3 or calculated from the following formulas (Hyland and Wexler 1983b). The saturation pressure over ice for the temperature range of 100 to 0 C is given by: where C 1 = E+03 C 2 = E+00 C 3 = E 03 C 4 = E 07 C 5 = E 09 C 6 = E 13 C 7 = E+00 The saturation pressure over liquid water for the temperature range of 0 to 200 C is given by: where ln( p ws ) = C 1 T + C 2 + C 3 T + C 4 T 2 + C 5 T 3 C 8 = E+03 C 9 = E+00 C 10 = E 02 C 11 = E 05 C 12 = E 08 C 13 = E+00 + C 6 T 4 + C 7 lnt ln( p ws ) = C 8 T + C 9 + C 10 T + C 11 T 2 In both Equations (5) and (6), + C 12 T 3 + C 13 lnt ln = natural logarithm p ws = saturation pressure, Pa T = absolute temperature, K = C The coefficients of Equations (5) and (6) have been derived from the Hyland-Wexler equations. Due to rounding errors in the derivations and in some computers calculating precision, the results obtained from Equations (5) and (6) may not agree precisely with Table 3 values. (5) (6)
3 Psychrometrics 6.3 Temp. t, C Humidity Ratio, kg w /kg da W s Table 2 Thermodynamic Properties of Moist Air (Standard Atmospheric Pressure, kpa) Volume, m 3 /kg (dry air) Enthalpy, kj/kg (dry air) Entropy, kj/(kg K) (dry air) v a v as v s h a h as h s s a s as s s Condensed Water Vapor Enthalpy Entropy, Pressure,, kj/kg kj/(kg K) kpa h w s w p s * *Extrapolated to represent metastable equilibrium with undercooled liquid. Temp., C
4 ASHRAE Fundamentals Handbook (SI) Temp. t, C Table 2 Humidity Ratio, kg w /kg da W s Thermodynamic Properties of Moist Air (Standard Atmospheric Pressure, kpa) (Continued) Volume, m 3 /kg (dry air) Enthalpy, kj/kg (dry air) Entropy, kj/(kg K) (dry air) v a v as v s h a h as h s s a s as s s Condensed Water Vapor Enthalpy Entropy, Pressure,, kj/kg kj/(kg K) kpa h w s w p s Temp., C
5 Psychrometrics 6.5 Table 3 Thermodynamic Properties of Water at Saturation Temp. t, C Absolute Pressure kpa p v f /v f Specific Volume, m 3 /kg Enthalpy, kj/kg Entropy, kj/(kg K) v fg /v fg v g h f /h f h fg /h fg h g s f /s f s fg /s fg s g Temp., C
6 ASHRAE Fundamentals Handbook (SI) Table 3 Thermodynamic Properties of Water at Saturation (Continued) Temp. t, C Absolute Pressure kpa p v f /v f Specific Volume, m 3 /kg Enthalpy, kj/kg Entropy, kj/(kg K) v fg /v fg v g h f /h f h fg /h fg h g s f /s f s fg /s fg s g Temp., C
7 Psychrometrics 6.7 Temp. t, C Absolute Pressure kpa p v f /v f Table 3 Thermodynamic Properties of Water at Saturation (Continued) Specific Volume, m 3 /kg Enthalpy, kj/kg Entropy, kj/(kg K) v fg /v fg v g h f /h f h fg /h fg h g s f /s f s fg /s fg s g Temp., C
8 ASHRAE Fundamentals Handbook (SI) HUMIDITY PARAMETERS Humidity ratio (alternatively, the moisture content or mixing ratio) W of a given moist air sample is defined as the ratio of the mass of water vapor to the mass of dry air contained in the sample: W = M w M a The humidity ratio W is equal to the mole fraction ratio x w /x a multiplied by the ratio of molecular masses; namely, / = , i.e.: W = x w x a Specific humidity q is the ratio of the mass of water vapor to the total mass of the moist air sample: q = M w ( M w + M a ) (7) (8) (9a) of the given sample of moist air. It is defined as the solution t d (p, W) of the equation: W s ( pt, d ) = W (15) Thermodynamic wet-bulb temperature t* is the temperature at which water (liquid or solid), by evaporating into moist air at a given dry-bulb temperature t and humidity ratio W, can bring air to saturation adiabatically at the same temperature t* while the pressure p is maintained constant. This parameter is considered separately in a later section. PERFECT GAS RELATIONSHIPS FOR DRY AND MOIST AIR When moist air is considered a mixture of independent perfect gases, dry air, and water vapor, each is assumed to obey the perfect gas equation of state as follows: In terms of the humidity ratio: Dry air p a V = n a RT (16) q = W ( 1 + W ) (9b) Water vapor p w V = n w RT (17) Absolute humidity (alternatively, water vapor density) d v is the ratio of the mass of water vapor to the total volume of the sample: d v = M w V (10) The density ρ of a moist air mixture is the ratio of the total mass to the total volume: ρ = ( M a + M w ) V = ( 1 v) ( 1 + W ) (11) where v is the moist air specific volume, m 3 /kg (dry air), as defined by Equation (27). HUMIDITY PARAMETERS INVOLVING SATURATION The following definitions of humidity parameters involve the concept of moist air saturation: Saturation humidity ratio W s (t, p) is the humidity ratio of moist air saturated with respect to water (or ice) at the same temperature t and pressure p. Degree of saturation µ is the ratio of the air humidity ratio W to the humidity ratio W s of saturated air at the same temperature and pressure: µ = W W s (12) Relative humidity φ is the ratio of the mole fraction of water vapor x w in a given moist air sample to the mole fraction x ws in an air sample, saturated at the same temperature and pressure: tp, where p a = partial pressure of dry air p w = partial pressure of water vapor V = total mixture volume n a = number of moles of dry air n w = number of moles of water vapor R = universal gas constant J/(kg mol K) T = absolute temperature, K or The mixture also obeys the perfect gas equation: (18) (19) where p = p a + p w is the total mixture pressure and n = n a + n w is the total number of moles in the mixture. From Equations (16) through (19), the mole fractions of dry air and water vapor are, respectively: and pv = nrt ( p a + p w )V = n a + n w ( )RT x a = p a ( p a + p w ) = p a p x w = p w ( p a + p w ) = p w p (20) (21) From Equations (8), (20), and (21), the humidity ratio W is given by: W p w = p p w (22) Combining Equations (8), (12), and (13): µ φ = x w x ws tp, φ = ( 1 φ)w s (13) (14) The degree of saturation µ is, by definition, Equation (12): where µ = W W s tp, Dew-point temperature t d is the temperature of moist air saturated at the same pressure p, with the same humidity ratio W as that W s p ws = p p ws (23)
9 Psychrometrics 6.9 The term p ws represents the saturation pressure of water vapor in the absence of air at the given temperature t. This pressure p ws is a function only of temperature and differs slightly from the vapor pressure of water in saturated moist air. The relative humidity φ is, by definition, Equation (13): Substituting Equation (21) for x w and x ws : Substituting Equation (21) for x ws into Equation (14): (24) (25) Both φ and µ are zero for dry air and unity for saturated moist air. At intermediate states their values differ, substantially so at higher temperatures. The specific volume v of a moist air mixture is expressed in terms of a unit mass of dry air, i.e.: (26) where V is the total volume of the mixture, M a is the total mass of dry air, and n a is the number of moles of dry air. By Equations (16) and (26), with the relation p = p a + p w : Using Equation (22): (27) (28) In Equations (27) and (28), v is specific volume, T is absolute temperature, p is total pressure, p w is the partial pressure of water vapor, and W is the humidity ratio. In specific units, Equation (28) may be expressed as where v v φ x w φ = p w p ws x ws φ = v = specific volume, m 3 /kg t = dry-bulb temperature, C W = humidity ratio, kg (water)/kg (dry air) p = total pressure, kpa The enthalpy of a mixture of perfect gases equals the sum of the individual partial enthalpies of the components. Therefore, the enthalpy of moist air can be written: (29) where h a is the specific enthalpy for dry air and h g is the specific enthalpy for saturated water vapor at the temperature of the mixture. Approximately: t, p tp, µ = ( 1 µ )( p ws p) = V M a = V ( n a ) RT = = ( p p w ) R a T p p w RT( W) R v a T( W) = = p p v = ( t ) ( W) p h = h a + Wh g h a = 1.006t ( kj/kg) (30) h g = t ( kj/kg) (31) where t is the dry-bulb temperature, C. The moist air enthalpy then becomes: h = 1.006t+ W( t) ( kj/kg) (32) THERMODYNAMIC WET-BULB TEMPERATURE AND DEW-POINT TEMPERATURE For any state of moist air, a temperature t* exists at which liquid (or solid) water evaporates into the air to bring it to saturation at exactly this same temperature and pressure (Harrison 1965). During the adiabatic saturation process, the saturated air is expelled at a temperature equal to that of the injected water (Figures 8 and 9). In the constant pressure process, the humidity ratio is increased from a given initial value W to the value W s *, corresponding to saturation at the temperature t*; the enthalpy is increased from a given initial value h to the value h s *, corresponding to saturation at the temperature t*; the mass of water added per unit mass of dry air is (W s * W), which adds energy to the moist air of amount (W s * W)h w *, where h w * denotes the specific enthalpy of the water added at the temperature t*. Therefore, if the process is strictly adiabatic, conservation of enthalpy at constant pressure requires that: h+ ( W* s W) h* w = h* s (33) The properties W s *, h w *, and h s * are functions only of the temperature t* for a fixed value of pressure. The value of t*, which satisfies Equation (33) for given values of h, W, and p, is the thermodynamic wet-bulb temperature. The psychrometer consists of two thermometers; one thermometer s bulb is covered by a wick that has been thoroughly wetted with water. When the wet bulb is placed in an airstream, water evaporates from the wick, eventually reaching an equilibrium temperature called the wet-bulb temperature. This process is not one of adiabatic saturation, which defines the thermodynamic wet-bulb temperature, but is one of simultaneous heat and mass transfer from the wet bulb. The fundamental mechanism of this process is described by the Lewis relation (Chapter 5). Fortunately, only small corrections must be applied to wet-bulb thermometer readings to obtain the thermodynamic wet-bulb temperature. As defined, thermodynamic wet-bulb temperature is a unique property of a given moist air sample independent of measurement techniques. Equation (33) is exact since it defines the thermodynamic wetbulb temperature t*. Substituting the approximate perfect gas relation [Equation (32)] for h, the corresponding expression for h s *, and the approximate relation h w * = 4.186t* ( kj kg) into Equation (33), and solving for the humidity ratio: W = ( t* )W* s ( t t* ) t 4.186t* (34) (35) where t and t* are in C. The dew-point temperature t d of moist air with humidity ratio W and pressure p was defined earlier as the solution t d (p, w) of W s (p, t d ). For perfect gases, this reduces to: p ws ( t d ) = p w = ( pw) ( W) (36) where p w is the water vapor partial pressure for the moist air sample and p ws (t d ) is the saturation vapor pressure at temperature t d. The
10 ASHRAE Fundamentals Handbook (SI) saturation vapor pressure is derived from Table 3 or from Equations (5) or (6). Alternatively, the dew-point temperature can be calculated directly by one of the following equations (Peppers 1988): For the dew-point temperature range of 0 to 93 C: t d = C 14 + C 15 α + C 16 α 2 + C 17 α 3 + C 18 ( p w ) and for temperatures below 0 C: t d = α α 2 where t d = dew-point temperature, C α = ln (p w ) p w = water vapor partial pressure, kpa C 14 =6.54 C 15 = C 16 = C 17 = C 18 = NUMERICAL CALCULATION OF MOIST AIR PROPERTIES (37) (38) The following are outlines, citing equations and tables already presented, for calculating moist air properties using perfect gas relations. These relations are sufficiently accurate for most engineering calculations in air-conditioning practice, and are readily adapted to either hand or computer calculating methods. For more details, refer to Tables 15 through 18 in Chapter 1 of Olivieri (1996). Graphical procedures are discussed in the section on psychrometric charts. Situation 1. Given: Dry-bulb temperature t, Wet-bulb temperature t*, Pressure p To Obtain Use Comments p ws (t*) Table 3 or Eq. (5) or (6) Sat. press. for temp. t* W s * Eq. (23) Using p ws (t*) W Eq. (35) p ws (t) Table 3 or Eq. (5) or (6) Sat. press. for temp. t W s Eq. (23) Using p ws (t) µ Eq. (12) Using W s φ Eq. (25) Using p ws (t) v Eq. (28) h Eq. (32) p w Eq. (36) t d Table 3 with Eq. (36), (37), or (38) Situation 2. Given: Dry-bulb temperature t, Dew-point temperature t d, Pressure p To Obtain Use Comments p w = p ws (t d ) Table 3 or Eq. (5) or (6) Sat. press. for temp. t d W Eq. (22) p ws (t) Table 3 or Eq. (5) or (6) Sat. press. for temp. t d W s Eq. (23) Using p ws (t) µ Eq. (12) Using W s φ Eq. (25) Using p ws (t) v Eq. (28) h Eq. (32) t* Eq. (23) and (35) with Table 3 or with Eq. (5) or (6) Requires trial-and-error or numerical solution method Situation 3. Given: Dry-bulb temperature t, Relative humidity φ, Pressure p To Obtain Use Comments p ws (t) Table 3 or Eq. (5) or (6) Sat. press. for temp. t p w Eq. (24) W Eq. (22) W s Eq. (23) Using p ws (t) µ Eq. (12) Using W s v Eq. (28) h Eq. (32) t d Table 3 with Eq. (36), (37), or (38) t* Eq. (23) and (35) with Table 3 or with Eq. (5) or (6) Requires trial-and-error or numerical solution method EXACT RELATIONS FOR COMPUTING W s AND φ Corrections that account for (1) the effect of dissolved gases on properties of condensed phase; (2) the effect of pressure on properties of condensed phase; and (3) the effect of intermolecular force on properties of moisture itself, can be applied to Equations (23) or (25): (23a) (23b) Table 4 lists f s values for a number of pressure and temperature combinations. Hyland and Wexler (1983a) give additional values. Table 4 W s f s p ws = p f s p ws φ = µ ( 1 µ )( f s p ws p) Values of f s and Estimated Maximum Uncertainties (EMU) 0.1 MPa 0.5 MPa 1 MPa EMU EMU EMU T, K f E+04 f E+04 f E MOIST AIR PROPERTY TABLES FOR STANDARD PRESSURE Table 2 shows values of thermodynamic properties for standard atmospheric pressure at temperatures from 60 to 90 C. The properties of intermediate moist air states can be calculated using the degree of saturation µ: Volume v = v a + µv as Enthalpy h = h a + µh as Entropy s = s a + µ s as (39) (40) (41) These equations are accurate to about 70 C. At higher temperatures, the errors can be significant. Hyland and Wexler (1983a) include charts that can be used to estimate errors for v, h, and s for standard barometric pressure. PSYCHROMETRIC CHARTS A psychrometric chart graphically represents the thermodynamic properties of moist air.
11 Psychrometrics 6.11 Fig. 1 ASHRAE Psychrometric Chart No. 1
Technical Information T-9100 SI. Suva. refrigerants. Thermodynamic Properties of. Suva Refrigerant [R-410A (50/50)]
![Technical Information T-9100 SI. Suva. refrigerants. Thermodynamic Properties of. Suva Refrigerant [R-410A (50/50)] Technical Information T-9100 SI. Suva. refrigerants. Thermodynamic Properties of. Suva Refrigerant [R-410A (50/50)]](/thumbs/89/98928029.jpg) d Suva refrigerants Technical Information T-9100SI Thermodynamic Properties of Suva 9100 Refrigerant [R-410A (50/50)] Thermodynamic Properties of Suva 9100 Refrigerant SI Units New tables of the thermodynamic
d Suva refrigerants Technical Information T-9100SI Thermodynamic Properties of Suva 9100 Refrigerant [R-410A (50/50)] Thermodynamic Properties of Suva 9100 Refrigerant SI Units New tables of the thermodynamic
DuPont Suva 95 Refrigerant
 Technical Information T-95 ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Technical Information T-95 ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
DuPont Suva 95 Refrigerant
 Technical Information T-95 SI DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Technical Information T-95 SI DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
DuPont Suva. DuPont. Thermodynamic Properties of. Refrigerant (R-410A) Technical Information. refrigerants T-410A ENG
 Technical Information T-410A ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 410A Refrigerant (R-410A) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Technical Information T-410A ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 410A Refrigerant (R-410A) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Mean bond enthalpy Standard enthalpy of formation Bond N H N N N N H O O O
 Q1. (a) Explain the meaning of the terms mean bond enthalpy and standard enthalpy of formation. Mean bond enthalpy... Standard enthalpy of formation... (5) (b) Some mean bond enthalpies are given below.
Q1. (a) Explain the meaning of the terms mean bond enthalpy and standard enthalpy of formation. Mean bond enthalpy... Standard enthalpy of formation... (5) (b) Some mean bond enthalpies are given below.
Figure 1 T / K Explain, in terms of molecules, why the first part of the graph in Figure 1 is a line that slopes up from the origin.
 Q1.(a) Figure 1 shows how the entropy of a molecular substance X varies with temperature. Figure 1 T / K (i) Explain, in terms of molecules, why the entropy is zero when the temperature is zero Kelvin.
Q1.(a) Figure 1 shows how the entropy of a molecular substance X varies with temperature. Figure 1 T / K (i) Explain, in terms of molecules, why the entropy is zero when the temperature is zero Kelvin.
HOMEWORK 4 = G. In order to plot the stress versus the stretch we define a normalized stretch:
 HOMEWORK 4 Problem a For the fast loading case, we want to derive the relationship between P zz and λ z. We know that the nominal stress is expressed as: P zz = ψ λ z where λ z = λ λ z. Therefore, applying
HOMEWORK 4 Problem a For the fast loading case, we want to derive the relationship between P zz and λ z. We know that the nominal stress is expressed as: P zz = ψ λ z where λ z = λ λ z. Therefore, applying
CHAPTER 25 SOLVING EQUATIONS BY ITERATIVE METHODS
 CHAPTER 5 SOLVING EQUATIONS BY ITERATIVE METHODS EXERCISE 104 Page 8 1. Find the positive root of the equation x + 3x 5 = 0, correct to 3 significant figures, using the method of bisection. Let f(x) =
CHAPTER 5 SOLVING EQUATIONS BY ITERATIVE METHODS EXERCISE 104 Page 8 1. Find the positive root of the equation x + 3x 5 = 0, correct to 3 significant figures, using the method of bisection. Let f(x) =
Homework 3 Solutions
 Homework 3 Solutions Igor Yanovsky (Math 151A TA) Problem 1: Compute the absolute error and relative error in approximations of p by p. (Use calculator!) a) p π, p 22/7; b) p π, p 3.141. Solution: For
Homework 3 Solutions Igor Yanovsky (Math 151A TA) Problem 1: Compute the absolute error and relative error in approximations of p by p. (Use calculator!) a) p π, p 22/7; b) p π, p 3.141. Solution: For
Απόκριση σε Μοναδιαία Ωστική Δύναμη (Unit Impulse) Απόκριση σε Δυνάμεις Αυθαίρετα Μεταβαλλόμενες με το Χρόνο. Απόστολος Σ.
 Απόκριση σε Δυνάμεις Αυθαίρετα Μεταβαλλόμενες με το Χρόνο The time integral of a force is referred to as impulse, is determined by and is obtained from: Newton s 2 nd Law of motion states that the action
Απόκριση σε Δυνάμεις Αυθαίρετα Μεταβαλλόμενες με το Χρόνο The time integral of a force is referred to as impulse, is determined by and is obtained from: Newton s 2 nd Law of motion states that the action
By R.L. Snyder (Revised March 24, 2005)
 Humidity Conversion By R.L. Snyder (Revised March 24, 2005) This Web page provides the equations used to make humidity conversions and tables o saturation vapor pressure. For a pd ile o this document,
Humidity Conversion By R.L. Snyder (Revised March 24, 2005) This Web page provides the equations used to make humidity conversions and tables o saturation vapor pressure. For a pd ile o this document,
CH 3 CH 2 COOCH 2 CH 2 CH 3 + H 2 O
 Q1.An experiment was carried out to determine the equilibrium constant, K c, for the following reaction. CH 3 CH 2 COOH + CH 3 CH 2 CH 2 OH CH 3 CH 2 COOCH 2 CH 2 CH 3 + H 2 O A student added measured
Q1.An experiment was carried out to determine the equilibrium constant, K c, for the following reaction. CH 3 CH 2 COOH + CH 3 CH 2 CH 2 OH CH 3 CH 2 COOCH 2 CH 2 CH 3 + H 2 O A student added measured
4.6 Autoregressive Moving Average Model ARMA(1,1)
 84 CHAPTER 4. STATIONARY TS MODELS 4.6 Autoregressive Moving Average Model ARMA(,) This section is an introduction to a wide class of models ARMA(p,q) which we will consider in more detail later in this
84 CHAPTER 4. STATIONARY TS MODELS 4.6 Autoregressive Moving Average Model ARMA(,) This section is an introduction to a wide class of models ARMA(p,q) which we will consider in more detail later in this
Phys460.nb Solution for the t-dependent Schrodinger s equation How did we find the solution? (not required)
 Phys460.nb 81 ψ n (t) is still the (same) eigenstate of H But for tdependent H. The answer is NO. 5.5.5. Solution for the tdependent Schrodinger s equation If we assume that at time t 0, the electron starts
Phys460.nb 81 ψ n (t) is still the (same) eigenstate of H But for tdependent H. The answer is NO. 5.5.5. Solution for the tdependent Schrodinger s equation If we assume that at time t 0, the electron starts
2 Composition. Invertible Mappings
 Arkansas Tech University MATH 4033: Elementary Modern Algebra Dr. Marcel B. Finan Composition. Invertible Mappings In this section we discuss two procedures for creating new mappings from old ones, namely,
Arkansas Tech University MATH 4033: Elementary Modern Algebra Dr. Marcel B. Finan Composition. Invertible Mappings In this section we discuss two procedures for creating new mappings from old ones, namely,
PROPERTIES OF ORDINARY WATER SUBSTANCE AT SATURATION FROF TME CRITICAL POINT DOWN TO 66 C EQUATIONS & TABLES
 PROPERTIES OF ORDINARY WATER SUBSTANCE AT SATURATION FROF TME CRITICAL POINT DOWN TO 66 C EQUATIONS & TABLES M. CONDE ENGINEERING, 2002 M. CONDE ENGINEERING, Zurich 2002 Properties of ordinary water substance
PROPERTIES OF ORDINARY WATER SUBSTANCE AT SATURATION FROF TME CRITICAL POINT DOWN TO 66 C EQUATIONS & TABLES M. CONDE ENGINEERING, 2002 M. CONDE ENGINEERING, Zurich 2002 Properties of ordinary water substance
Second Order RLC Filters
 ECEN 60 Circuits/Electronics Spring 007-0-07 P. Mathys Second Order RLC Filters RLC Lowpass Filter A passive RLC lowpass filter (LPF) circuit is shown in the following schematic. R L C v O (t) Using phasor
ECEN 60 Circuits/Electronics Spring 007-0-07 P. Mathys Second Order RLC Filters RLC Lowpass Filter A passive RLC lowpass filter (LPF) circuit is shown in the following schematic. R L C v O (t) Using phasor
3.4 SUM AND DIFFERENCE FORMULAS. NOTE: cos(α+β) cos α + cos β cos(α-β) cos α -cos β
 3.4 SUM AND DIFFERENCE FORMULAS Page Theorem cos(αβ cos α cos β -sin α cos(α-β cos α cos β sin α NOTE: cos(αβ cos α cos β cos(α-β cos α -cos β Proof of cos(α-β cos α cos β sin α Let s use a unit circle
3.4 SUM AND DIFFERENCE FORMULAS Page Theorem cos(αβ cos α cos β -sin α cos(α-β cos α cos β sin α NOTE: cos(αβ cos α cos β cos(α-β cos α -cos β Proof of cos(α-β cos α cos β sin α Let s use a unit circle
Section 8.3 Trigonometric Equations
 99 Section 8. Trigonometric Equations Objective 1: Solve Equations Involving One Trigonometric Function. In this section and the next, we will exple how to solving equations involving trigonometric functions.
99 Section 8. Trigonometric Equations Objective 1: Solve Equations Involving One Trigonometric Function. In this section and the next, we will exple how to solving equations involving trigonometric functions.
Matrices and Determinants
 Matrices and Determinants SUBJECTIVE PROBLEMS: Q 1. For what value of k do the following system of equations possess a non-trivial (i.e., not all zero) solution over the set of rationals Q? x + ky + 3z
Matrices and Determinants SUBJECTIVE PROBLEMS: Q 1. For what value of k do the following system of equations possess a non-trivial (i.e., not all zero) solution over the set of rationals Q? x + ky + 3z
6.3 Forecasting ARMA processes
 122 CHAPTER 6. ARMA MODELS 6.3 Forecasting ARMA processes The purpose of forecasting is to predict future values of a TS based on the data collected to the present. In this section we will discuss a linear
122 CHAPTER 6. ARMA MODELS 6.3 Forecasting ARMA processes The purpose of forecasting is to predict future values of a TS based on the data collected to the present. In this section we will discuss a linear
Enthalpy data for the reacting species are given in the table below. The activation energy decreases when the temperature is increased.
 Q1.This question is about the reaction given below. O(g) + H 2O(g) O 2(g) + H 2(g) Enthalpy data for the reacting species are given in the table below. Substance O(g) H 2O(g) O 2(g) H 2(g) ΔH / kj mol
Q1.This question is about the reaction given below. O(g) + H 2O(g) O 2(g) + H 2(g) Enthalpy data for the reacting species are given in the table below. Substance O(g) H 2O(g) O 2(g) H 2(g) ΔH / kj mol
Other Test Constructions: Likelihood Ratio & Bayes Tests
 Other Test Constructions: Likelihood Ratio & Bayes Tests Side-Note: So far we have seen a few approaches for creating tests such as Neyman-Pearson Lemma ( most powerful tests of H 0 : θ = θ 0 vs H 1 :
Other Test Constructions: Likelihood Ratio & Bayes Tests Side-Note: So far we have seen a few approaches for creating tests such as Neyman-Pearson Lemma ( most powerful tests of H 0 : θ = θ 0 vs H 1 :
titanium laser beam volume to be heated joint titanium Fig. 5.1
 1 Lasers are often used to form precision-welded joints in titanium. To form one such joint it is first necessary to increase the temperature of the titanium to its melting point. Fig. 5.1 shows the joint
1 Lasers are often used to form precision-welded joints in titanium. To form one such joint it is first necessary to increase the temperature of the titanium to its melting point. Fig. 5.1 shows the joint
Chapter 6: Systems of Linear Differential. be continuous functions on the interval
 Chapter 6: Systems of Linear Differential Equations Let a (t), a 2 (t),..., a nn (t), b (t), b 2 (t),..., b n (t) be continuous functions on the interval I. The system of n first-order differential equations
Chapter 6: Systems of Linear Differential Equations Let a (t), a 2 (t),..., a nn (t), b (t), b 2 (t),..., b n (t) be continuous functions on the interval I. The system of n first-order differential equations
Strain gauge and rosettes
 Strain gauge and rosettes Introduction A strain gauge is a device which is used to measure strain (deformation) on an object subjected to forces. Strain can be measured using various types of devices classified
Strain gauge and rosettes Introduction A strain gauge is a device which is used to measure strain (deformation) on an object subjected to forces. Strain can be measured using various types of devices classified
Approximation of distance between locations on earth given by latitude and longitude
 Approximation of distance between locations on earth given by latitude and longitude Jan Behrens 2012-12-31 In this paper we shall provide a method to approximate distances between two points on earth
Approximation of distance between locations on earth given by latitude and longitude Jan Behrens 2012-12-31 In this paper we shall provide a method to approximate distances between two points on earth
Areas and Lengths in Polar Coordinates
 Kiryl Tsishchanka Areas and Lengths in Polar Coordinates In this section we develop the formula for the area of a region whose boundary is given by a polar equation. We need to use the formula for the
Kiryl Tsishchanka Areas and Lengths in Polar Coordinates In this section we develop the formula for the area of a region whose boundary is given by a polar equation. We need to use the formula for the
EE512: Error Control Coding
 EE512: Error Control Coding Solution for Assignment on Finite Fields February 16, 2007 1. (a) Addition and Multiplication tables for GF (5) and GF (7) are shown in Tables 1 and 2. + 0 1 2 3 4 0 0 1 2 3
EE512: Error Control Coding Solution for Assignment on Finite Fields February 16, 2007 1. (a) Addition and Multiplication tables for GF (5) and GF (7) are shown in Tables 1 and 2. + 0 1 2 3 4 0 0 1 2 3
The Simply Typed Lambda Calculus
 Type Inference Instead of writing type annotations, can we use an algorithm to infer what the type annotations should be? That depends on the type system. For simple type systems the answer is yes, and
Type Inference Instead of writing type annotations, can we use an algorithm to infer what the type annotations should be? That depends on the type system. For simple type systems the answer is yes, and
Ύγρανση και Αφύγρανση. Ψυχρομετρία. 21-Nov-16
 Ύγρανση και Αφύγρανση Ψυχρομετρία η μελέτη των ιδιοτήτων του υγρού αέρα δηλαδή του μίγματος αέρα-ατμού-νερού. Ένα βασικό πρόβλημα: Δεδομένων των P (bar) Πίεση T ( o C) Θερμοκρασία Υ (kg/kg ξβ) Υγρασία
Ύγρανση και Αφύγρανση Ψυχρομετρία η μελέτη των ιδιοτήτων του υγρού αέρα δηλαδή του μίγματος αέρα-ατμού-νερού. Ένα βασικό πρόβλημα: Δεδομένων των P (bar) Πίεση T ( o C) Θερμοκρασία Υ (kg/kg ξβ) Υγρασία
Problem Set 9 Solutions. θ + 1. θ 2 + cotθ ( ) sinθ e iφ is an eigenfunction of the ˆ L 2 operator. / θ 2. φ 2. sin 2 θ φ 2. ( ) = e iφ. = e iφ cosθ.
 Chemistry 362 Dr Jean M Standard Problem Set 9 Solutions The ˆ L 2 operator is defined as Verify that the angular wavefunction Y θ,φ) Also verify that the eigenvalue is given by 2! 2 & L ˆ 2! 2 2 θ 2 +
Chemistry 362 Dr Jean M Standard Problem Set 9 Solutions The ˆ L 2 operator is defined as Verify that the angular wavefunction Y θ,φ) Also verify that the eigenvalue is given by 2! 2 & L ˆ 2! 2 2 θ 2 +
[1] P Q. Fig. 3.1
![[1] P Q. Fig. 3.1 [1] P Q. Fig. 3.1](/thumbs/79/80362156.jpg) 1 (a) Define resistance....... [1] (b) The smallest conductor within a computer processing chip can be represented as a rectangular block that is one atom high, four atoms wide and twenty atoms long. One
1 (a) Define resistance....... [1] (b) The smallest conductor within a computer processing chip can be represented as a rectangular block that is one atom high, four atoms wide and twenty atoms long. One
APPENDIX A. Summary of the English Engineering (EE) System of Units
 Appendixes A. Summary of the English Engineering (EE) System of Units B. Summary of the International System (SI) of Units C. Friction-Factor Chart D. Oblique-Shock Charts (γ = 1.4) (Two-Dimensional) E.
Appendixes A. Summary of the English Engineering (EE) System of Units B. Summary of the International System (SI) of Units C. Friction-Factor Chart D. Oblique-Shock Charts (γ = 1.4) (Two-Dimensional) E.
Correction Table for an Alcoholometer Calibrated at 20 o C
 An alcoholometer is a device that measures the concentration of ethanol in a water-ethanol mixture (often in units of %abv percent alcohol by volume). The depth to which an alcoholometer sinks in a water-ethanol
An alcoholometer is a device that measures the concentration of ethanol in a water-ethanol mixture (often in units of %abv percent alcohol by volume). The depth to which an alcoholometer sinks in a water-ethanol
Math 6 SL Probability Distributions Practice Test Mark Scheme
 Math 6 SL Probability Distributions Practice Test Mark Scheme. (a) Note: Award A for vertical line to right of mean, A for shading to right of their vertical line. AA N (b) evidence of recognizing symmetry
Math 6 SL Probability Distributions Practice Test Mark Scheme. (a) Note: Award A for vertical line to right of mean, A for shading to right of their vertical line. AA N (b) evidence of recognizing symmetry
Statistical Inference I Locally most powerful tests
 Statistical Inference I Locally most powerful tests Shirsendu Mukherjee Department of Statistics, Asutosh College, Kolkata, India. shirsendu st@yahoo.co.in So far we have treated the testing of one-sided
Statistical Inference I Locally most powerful tests Shirsendu Mukherjee Department of Statistics, Asutosh College, Kolkata, India. shirsendu st@yahoo.co.in So far we have treated the testing of one-sided
Assalamu `alaikum wr. wb.
 LUMP SUM Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. LUMP SUM Lump sum lump sum lump sum. lump sum fixed price lump sum lump
LUMP SUM Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. LUMP SUM Lump sum lump sum lump sum. lump sum fixed price lump sum lump
Ordinal Arithmetic: Addition, Multiplication, Exponentiation and Limit
 Ordinal Arithmetic: Addition, Multiplication, Exponentiation and Limit Ting Zhang Stanford May 11, 2001 Stanford, 5/11/2001 1 Outline Ordinal Classification Ordinal Addition Ordinal Multiplication Ordinal
Ordinal Arithmetic: Addition, Multiplication, Exponentiation and Limit Ting Zhang Stanford May 11, 2001 Stanford, 5/11/2001 1 Outline Ordinal Classification Ordinal Addition Ordinal Multiplication Ordinal
STEAM TABLES. Mollier Diagram
 STEAM TABLES and Mollier Diagram (S.I. Units) dharm \M-therm\C-steam.pm5 CONTENTS Table No. Page No. 1. Saturated Water and Steam (Temperature) Tables I (ii) 2. Saturated Water and Steam (Pressure) Tables
STEAM TABLES and Mollier Diagram (S.I. Units) dharm \M-therm\C-steam.pm5 CONTENTS Table No. Page No. 1. Saturated Water and Steam (Temperature) Tables I (ii) 2. Saturated Water and Steam (Pressure) Tables
derivation of the Laplacian from rectangular to spherical coordinates
 derivation of the Laplacian from rectangular to spherical coordinates swapnizzle 03-03- :5:43 We begin by recognizing the familiar conversion from rectangular to spherical coordinates (note that φ is used
derivation of the Laplacian from rectangular to spherical coordinates swapnizzle 03-03- :5:43 We begin by recognizing the familiar conversion from rectangular to spherical coordinates (note that φ is used
Instruction Execution Times
 1 C Execution Times InThisAppendix... Introduction DL330 Execution Times DL330P Execution Times DL340 Execution Times C-2 Execution Times Introduction Data Registers This appendix contains several tables
1 C Execution Times InThisAppendix... Introduction DL330 Execution Times DL330P Execution Times DL340 Execution Times C-2 Execution Times Introduction Data Registers This appendix contains several tables
k A = [k, k]( )[a 1, a 2 ] = [ka 1,ka 2 ] 4For the division of two intervals of confidence in R +
[a 1, a 2 ] = [ka 1,ka 2 ] 4For the division of two intervals of confidence in R + k A = [k, k]( )[a 1, a 2 ] = [ka 1,ka 2 ] 4For the division of two intervals of confidence in R +](/thumbs/73/69566903.jpg) Chapter 3. Fuzzy Arithmetic 3- Fuzzy arithmetic: ~Addition(+) and subtraction (-): Let A = [a and B = [b, b in R If x [a and y [b, b than x+y [a +b +b Symbolically,we write A(+)B = [a (+)[b, b = [a +b
Chapter 3. Fuzzy Arithmetic 3- Fuzzy arithmetic: ~Addition(+) and subtraction (-): Let A = [a and B = [b, b in R If x [a and y [b, b than x+y [a +b +b Symbolically,we write A(+)B = [a (+)[b, b = [a +b
Homework 8 Model Solution Section
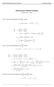 MATH 004 Homework Solution Homework 8 Model Solution Section 14.5 14.6. 14.5. Use the Chain Rule to find dz where z cosx + 4y), x 5t 4, y 1 t. dz dx + dy y sinx + 4y)0t + 4) sinx + 4y) 1t ) 0t + 4t ) sinx
MATH 004 Homework Solution Homework 8 Model Solution Section 14.5 14.6. 14.5. Use the Chain Rule to find dz where z cosx + 4y), x 5t 4, y 1 t. dz dx + dy y sinx + 4y)0t + 4) sinx + 4y) 1t ) 0t + 4t ) sinx
Areas and Lengths in Polar Coordinates
 Kiryl Tsishchanka Areas and Lengths in Polar Coordinates In this section we develop the formula for the area of a region whose boundary is given by a polar equation. We need to use the formula for the
Kiryl Tsishchanka Areas and Lengths in Polar Coordinates In this section we develop the formula for the area of a region whose boundary is given by a polar equation. We need to use the formula for the
ANSWERSHEET (TOPIC = DIFFERENTIAL CALCULUS) COLLECTION #2. h 0 h h 0 h h 0 ( ) g k = g 0 + g 1 + g g 2009 =?
 Teko Classes IITJEE/AIEEE Maths by SUHAAG SIR, Bhopal, Ph (0755) 3 00 000 www.tekoclasses.com ANSWERSHEET (TOPIC DIFFERENTIAL CALCULUS) COLLECTION # Question Type A.Single Correct Type Q. (A) Sol least
Teko Classes IITJEE/AIEEE Maths by SUHAAG SIR, Bhopal, Ph (0755) 3 00 000 www.tekoclasses.com ANSWERSHEET (TOPIC DIFFERENTIAL CALCULUS) COLLECTION # Question Type A.Single Correct Type Q. (A) Sol least
DESIGN OF MACHINERY SOLUTION MANUAL h in h 4 0.
 DESIGN OF MACHINERY SOLUTION MANUAL -7-1! PROBLEM -7 Statement: Design a double-dwell cam to move a follower from to 25 6, dwell for 12, fall 25 and dwell for the remader The total cycle must take 4 sec
DESIGN OF MACHINERY SOLUTION MANUAL -7-1! PROBLEM -7 Statement: Design a double-dwell cam to move a follower from to 25 6, dwell for 12, fall 25 and dwell for the remader The total cycle must take 4 sec
Econ 2110: Fall 2008 Suggested Solutions to Problem Set 8 questions or comments to Dan Fetter 1
 Eon : Fall 8 Suggested Solutions to Problem Set 8 Email questions or omments to Dan Fetter Problem. Let X be a salar with density f(x, θ) (θx + θ) [ x ] with θ. (a) Find the most powerful level α test
Eon : Fall 8 Suggested Solutions to Problem Set 8 Email questions or omments to Dan Fetter Problem. Let X be a salar with density f(x, θ) (θx + θ) [ x ] with θ. (a) Find the most powerful level α test
Second Order Partial Differential Equations
 Chapter 7 Second Order Partial Differential Equations 7.1 Introduction A second order linear PDE in two independent variables (x, y Ω can be written as A(x, y u x + B(x, y u xy + C(x, y u u u + D(x, y
Chapter 7 Second Order Partial Differential Equations 7.1 Introduction A second order linear PDE in two independent variables (x, y Ω can be written as A(x, y u x + B(x, y u xy + C(x, y u u u + D(x, y
ST5224: Advanced Statistical Theory II
 ST5224: Advanced Statistical Theory II 2014/2015: Semester II Tutorial 7 1. Let X be a sample from a population P and consider testing hypotheses H 0 : P = P 0 versus H 1 : P = P 1, where P j is a known
ST5224: Advanced Statistical Theory II 2014/2015: Semester II Tutorial 7 1. Let X be a sample from a population P and consider testing hypotheses H 0 : P = P 0 versus H 1 : P = P 1, where P j is a known
CE 530 Molecular Simulation
 C 53 olecular Siulation Lecture Histogra Reweighting ethods David. Kofke Departent of Cheical ngineering SUNY uffalo kofke@eng.buffalo.edu Histogra Reweighting ethod to cobine results taken at different
C 53 olecular Siulation Lecture Histogra Reweighting ethods David. Kofke Departent of Cheical ngineering SUNY uffalo kofke@eng.buffalo.edu Histogra Reweighting ethod to cobine results taken at different
Solutions to Exercise Sheet 5
 Solutions to Eercise Sheet 5 jacques@ucsd.edu. Let X and Y be random variables with joint pdf f(, y) = 3y( + y) where and y. Determine each of the following probabilities. Solutions. a. P (X ). b. P (X
Solutions to Eercise Sheet 5 jacques@ucsd.edu. Let X and Y be random variables with joint pdf f(, y) = 3y( + y) where and y. Determine each of the following probabilities. Solutions. a. P (X ). b. P (X
2. Chemical Thermodynamics and Energetics - I
 . Chemical Thermodynamics and Energetics - I 1. Given : Initial Volume ( = 5L dm 3 Final Volume (V = 10L dm 3 ext = 304 cm of Hg Work done W = ext V ext = 304 cm of Hg = 304 atm [... 76cm of Hg = 1 atm]
. Chemical Thermodynamics and Energetics - I 1. Given : Initial Volume ( = 5L dm 3 Final Volume (V = 10L dm 3 ext = 304 cm of Hg Work done W = ext V ext = 304 cm of Hg = 304 atm [... 76cm of Hg = 1 atm]
5.4 The Poisson Distribution.
 The worst thing you can do about a situation is nothing. Sr. O Shea Jackson 5.4 The Poisson Distribution. Description of the Poisson Distribution Discrete probability distribution. The random variable
The worst thing you can do about a situation is nothing. Sr. O Shea Jackson 5.4 The Poisson Distribution. Description of the Poisson Distribution Discrete probability distribution. The random variable
Concrete Mathematics Exercises from 30 September 2016
 Concrete Mathematics Exercises from 30 September 2016 Silvio Capobianco Exercise 1.7 Let H(n) = J(n + 1) J(n). Equation (1.8) tells us that H(2n) = 2, and H(2n+1) = J(2n+2) J(2n+1) = (2J(n+1) 1) (2J(n)+1)
Concrete Mathematics Exercises from 30 September 2016 Silvio Capobianco Exercise 1.7 Let H(n) = J(n + 1) J(n). Equation (1.8) tells us that H(2n) = 2, and H(2n+1) = J(2n+2) J(2n+1) = (2J(n+1) 1) (2J(n)+1)
Μονοβάθμια Συστήματα: Εξίσωση Κίνησης, Διατύπωση του Προβλήματος και Μέθοδοι Επίλυσης. Απόστολος Σ. Παπαγεωργίου
 Μονοβάθμια Συστήματα: Εξίσωση Κίνησης, Διατύπωση του Προβλήματος και Μέθοδοι Επίλυσης VISCOUSLY DAMPED 1-DOF SYSTEM Μονοβάθμια Συστήματα με Ιξώδη Απόσβεση Equation of Motion (Εξίσωση Κίνησης): Complete
Μονοβάθμια Συστήματα: Εξίσωση Κίνησης, Διατύπωση του Προβλήματος και Μέθοδοι Επίλυσης VISCOUSLY DAMPED 1-DOF SYSTEM Μονοβάθμια Συστήματα με Ιξώδη Απόσβεση Equation of Motion (Εξίσωση Κίνησης): Complete
Numerical Analysis FMN011
 Numerical Analysis FMN011 Carmen Arévalo Lund University carmen@maths.lth.se Lecture 12 Periodic data A function g has period P if g(x + P ) = g(x) Model: Trigonometric polynomial of order M T M (x) =
Numerical Analysis FMN011 Carmen Arévalo Lund University carmen@maths.lth.se Lecture 12 Periodic data A function g has period P if g(x + P ) = g(x) Model: Trigonometric polynomial of order M T M (x) =
Nuclear Physics 5. Name: Date: 8 (1)
 Name: Date: Nuclear Physics 5. A sample of radioactive carbon-4 decays into a stable isotope of nitrogen. As the carbon-4 decays, the rate at which the amount of nitrogen is produced A. decreases linearly
Name: Date: Nuclear Physics 5. A sample of radioactive carbon-4 decays into a stable isotope of nitrogen. As the carbon-4 decays, the rate at which the amount of nitrogen is produced A. decreases linearly
PARTIAL NOTES for 6.1 Trigonometric Identities
 PARTIAL NOTES for 6.1 Trigonometric Identities tanθ = sinθ cosθ cotθ = cosθ sinθ BASIC IDENTITIES cscθ = 1 sinθ secθ = 1 cosθ cotθ = 1 tanθ PYTHAGOREAN IDENTITIES sin θ + cos θ =1 tan θ +1= sec θ 1 + cot
PARTIAL NOTES for 6.1 Trigonometric Identities tanθ = sinθ cosθ cotθ = cosθ sinθ BASIC IDENTITIES cscθ = 1 sinθ secθ = 1 cosθ cotθ = 1 tanθ PYTHAGOREAN IDENTITIES sin θ + cos θ =1 tan θ +1= sec θ 1 + cot
10.7 Performance of Second-Order System (Unit Step Response)
 Lecture Notes on Control Systems/D. Ghose/0 57 0.7 Performance of Second-Order System (Unit Step Response) Consider the second order system a ÿ + a ẏ + a 0 y = b 0 r So, Y (s) R(s) = b 0 a s + a s + a
Lecture Notes on Control Systems/D. Ghose/0 57 0.7 Performance of Second-Order System (Unit Step Response) Consider the second order system a ÿ + a ẏ + a 0 y = b 0 r So, Y (s) R(s) = b 0 a s + a s + a
Reminders: linear functions
 Reminders: linear functions Let U and V be vector spaces over the same field F. Definition A function f : U V is linear if for every u 1, u 2 U, f (u 1 + u 2 ) = f (u 1 ) + f (u 2 ), and for every u U
Reminders: linear functions Let U and V be vector spaces over the same field F. Definition A function f : U V is linear if for every u 1, u 2 U, f (u 1 + u 2 ) = f (u 1 ) + f (u 2 ), and for every u U
Example Sheet 3 Solutions
 Example Sheet 3 Solutions. i Regular Sturm-Liouville. ii Singular Sturm-Liouville mixed boundary conditions. iii Not Sturm-Liouville ODE is not in Sturm-Liouville form. iv Regular Sturm-Liouville note
Example Sheet 3 Solutions. i Regular Sturm-Liouville. ii Singular Sturm-Liouville mixed boundary conditions. iii Not Sturm-Liouville ODE is not in Sturm-Liouville form. iv Regular Sturm-Liouville note
Inverse trigonometric functions & General Solution of Trigonometric Equations. ------------------ ----------------------------- -----------------
 Inverse trigonometric functions & General Solution of Trigonometric Equations. 1. Sin ( ) = a) b) c) d) Ans b. Solution : Method 1. Ans a: 17 > 1 a) is rejected. w.k.t Sin ( sin ) = d is rejected. If sin
Inverse trigonometric functions & General Solution of Trigonometric Equations. 1. Sin ( ) = a) b) c) d) Ans b. Solution : Method 1. Ans a: 17 > 1 a) is rejected. w.k.t Sin ( sin ) = d is rejected. If sin
Appendix to On the stability of a compressible axisymmetric rotating flow in a pipe. By Z. Rusak & J. H. Lee
 Appendi to On the stability of a compressible aisymmetric rotating flow in a pipe By Z. Rusak & J. H. Lee Journal of Fluid Mechanics, vol. 5 4, pp. 5 4 This material has not been copy-edited or typeset
Appendi to On the stability of a compressible aisymmetric rotating flow in a pipe By Z. Rusak & J. H. Lee Journal of Fluid Mechanics, vol. 5 4, pp. 5 4 This material has not been copy-edited or typeset
Chapter 6: Systems of Linear Differential. be continuous functions on the interval
 Chapter 6: Systems of Linear Differential Equations Let a (t), a 2 (t),..., a nn (t), b (t), b 2 (t),..., b n (t) be continuous functions on the interval I. The system of n first-order differential equations
Chapter 6: Systems of Linear Differential Equations Let a (t), a 2 (t),..., a nn (t), b (t), b 2 (t),..., b n (t) be continuous functions on the interval I. The system of n first-order differential equations
Practice Exam 2. Conceptual Questions. 1. State a Basic identity and then verify it. (a) Identity: Solution: One identity is csc(θ) = 1
 Conceptual Questions. State a Basic identity and then verify it. a) Identity: Solution: One identity is cscθ) = sinθ) Practice Exam b) Verification: Solution: Given the point of intersection x, y) of the
Conceptual Questions. State a Basic identity and then verify it. a) Identity: Solution: One identity is cscθ) = sinθ) Practice Exam b) Verification: Solution: Given the point of intersection x, y) of the
Potential Dividers. 46 minutes. 46 marks. Page 1 of 11
 Potential Dividers 46 minutes 46 marks Page 1 of 11 Q1. In the circuit shown in the figure below, the battery, of negligible internal resistance, has an emf of 30 V. The pd across the lamp is 6.0 V and
Potential Dividers 46 minutes 46 marks Page 1 of 11 Q1. In the circuit shown in the figure below, the battery, of negligible internal resistance, has an emf of 30 V. The pd across the lamp is 6.0 V and
2. THEORY OF EQUATIONS. PREVIOUS EAMCET Bits.
 EAMCET-. THEORY OF EQUATIONS PREVIOUS EAMCET Bits. Each of the roots of the equation x 6x + 6x 5= are increased by k so that the new transformed equation does not contain term. Then k =... - 4. - Sol.
EAMCET-. THEORY OF EQUATIONS PREVIOUS EAMCET Bits. Each of the roots of the equation x 6x + 6x 5= are increased by k so that the new transformed equation does not contain term. Then k =... - 4. - Sol.
Partial Differential Equations in Biology The boundary element method. March 26, 2013
 The boundary element method March 26, 203 Introduction and notation The problem: u = f in D R d u = ϕ in Γ D u n = g on Γ N, where D = Γ D Γ N, Γ D Γ N = (possibly, Γ D = [Neumann problem] or Γ N = [Dirichlet
The boundary element method March 26, 203 Introduction and notation The problem: u = f in D R d u = ϕ in Γ D u n = g on Γ N, where D = Γ D Γ N, Γ D Γ N = (possibly, Γ D = [Neumann problem] or Γ N = [Dirichlet
Jesse Maassen and Mark Lundstrom Purdue University November 25, 2013
 Notes on Average Scattering imes and Hall Factors Jesse Maassen and Mar Lundstrom Purdue University November 5, 13 I. Introduction 1 II. Solution of the BE 1 III. Exercises: Woring out average scattering
Notes on Average Scattering imes and Hall Factors Jesse Maassen and Mar Lundstrom Purdue University November 5, 13 I. Introduction 1 II. Solution of the BE 1 III. Exercises: Woring out average scattering
ω ω ω ω ω ω+2 ω ω+2 + ω ω ω ω+2 + ω ω+1 ω ω+2 2 ω ω ω ω ω ω ω ω+1 ω ω2 ω ω2 + ω ω ω2 + ω ω ω ω2 + ω ω+1 ω ω2 + ω ω+1 + ω ω ω ω2 + ω
 0 1 2 3 4 5 6 ω ω + 1 ω + 2 ω + 3 ω + 4 ω2 ω2 + 1 ω2 + 2 ω2 + 3 ω3 ω3 + 1 ω3 + 2 ω4 ω4 + 1 ω5 ω 2 ω 2 + 1 ω 2 + 2 ω 2 + ω ω 2 + ω + 1 ω 2 + ω2 ω 2 2 ω 2 2 + 1 ω 2 2 + ω ω 2 3 ω 3 ω 3 + 1 ω 3 + ω ω 3 +
0 1 2 3 4 5 6 ω ω + 1 ω + 2 ω + 3 ω + 4 ω2 ω2 + 1 ω2 + 2 ω2 + 3 ω3 ω3 + 1 ω3 + 2 ω4 ω4 + 1 ω5 ω 2 ω 2 + 1 ω 2 + 2 ω 2 + ω ω 2 + ω + 1 ω 2 + ω2 ω 2 2 ω 2 2 + 1 ω 2 2 + ω ω 2 3 ω 3 ω 3 + 1 ω 3 + ω ω 3 +
Εργαστήριο Ανάπτυξης Εφαρμογών Βάσεων Δεδομένων. Εξάμηνο 7 ο
 Εργαστήριο Ανάπτυξης Εφαρμογών Βάσεων Δεδομένων Εξάμηνο 7 ο Procedures and Functions Stored procedures and functions are named blocks of code that enable you to group and organize a series of SQL and PL/SQL
Εργαστήριο Ανάπτυξης Εφαρμογών Βάσεων Δεδομένων Εξάμηνο 7 ο Procedures and Functions Stored procedures and functions are named blocks of code that enable you to group and organize a series of SQL and PL/SQL
6.1. Dirac Equation. Hamiltonian. Dirac Eq.
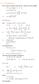 6.1. Dirac Equation Ref: M.Kaku, Quantum Field Theory, Oxford Univ Press (1993) η μν = η μν = diag(1, -1, -1, -1) p 0 = p 0 p = p i = -p i p μ p μ = p 0 p 0 + p i p i = E c 2 - p 2 = (m c) 2 H = c p 2
6.1. Dirac Equation Ref: M.Kaku, Quantum Field Theory, Oxford Univ Press (1993) η μν = η μν = diag(1, -1, -1, -1) p 0 = p 0 p = p i = -p i p μ p μ = p 0 p 0 + p i p i = E c 2 - p 2 = (m c) 2 H = c p 2
Lecture 2: Dirac notation and a review of linear algebra Read Sakurai chapter 1, Baym chatper 3
 Lecture 2: Dirac notation and a review of linear algebra Read Sakurai chapter 1, Baym chatper 3 1 State vector space and the dual space Space of wavefunctions The space of wavefunctions is the set of all
Lecture 2: Dirac notation and a review of linear algebra Read Sakurai chapter 1, Baym chatper 3 1 State vector space and the dual space Space of wavefunctions The space of wavefunctions is the set of all
Calculating the propagation delay of coaxial cable
 Your source for quality GNSS Networking Solutions and Design Services! Page 1 of 5 Calculating the propagation delay of coaxial cable The delay of a cable or velocity factor is determined by the dielectric
Your source for quality GNSS Networking Solutions and Design Services! Page 1 of 5 Calculating the propagation delay of coaxial cable The delay of a cable or velocity factor is determined by the dielectric
Thermodynamic Tables to accompany Modern Engineering Thermodynamics
 Thermodynamic Tables to accompany Modern Engineering Thermodynamics This page intentionally left blank Thermodynamic Tables to accompany Modern Engineering Thermodynamics Robert T. Balmer AMSTERDAM BOSTON
Thermodynamic Tables to accompany Modern Engineering Thermodynamics This page intentionally left blank Thermodynamic Tables to accompany Modern Engineering Thermodynamics Robert T. Balmer AMSTERDAM BOSTON
the total number of electrons passing through the lamp.
 1. A 12 V 36 W lamp is lit to normal brightness using a 12 V car battery of negligible internal resistance. The lamp is switched on for one hour (3600 s). For the time of 1 hour, calculate (i) the energy
1. A 12 V 36 W lamp is lit to normal brightness using a 12 V car battery of negligible internal resistance. The lamp is switched on for one hour (3600 s). For the time of 1 hour, calculate (i) the energy
Space-Time Symmetries
 Chapter Space-Time Symmetries In classical fiel theory any continuous symmetry of the action generates a conserve current by Noether's proceure. If the Lagrangian is not invariant but only shifts by a
Chapter Space-Time Symmetries In classical fiel theory any continuous symmetry of the action generates a conserve current by Noether's proceure. If the Lagrangian is not invariant but only shifts by a
Math221: HW# 1 solutions
 Math: HW# solutions Andy Royston October, 5 7.5.7, 3 rd Ed. We have a n = b n = a = fxdx = xdx =, x cos nxdx = x sin nx n sin nxdx n = cos nx n = n n, x sin nxdx = x cos nx n + cos nxdx n cos n = + sin
Math: HW# solutions Andy Royston October, 5 7.5.7, 3 rd Ed. We have a n = b n = a = fxdx = xdx =, x cos nxdx = x sin nx n sin nxdx n = cos nx n = n n, x sin nxdx = x cos nx n + cos nxdx n cos n = + sin
DERIVATION OF MILES EQUATION FOR AN APPLIED FORCE Revision C
 DERIVATION OF MILES EQUATION FOR AN APPLIED FORCE Revision C By Tom Irvine Email: tomirvine@aol.com August 6, 8 Introduction The obective is to derive a Miles equation which gives the overall response
DERIVATION OF MILES EQUATION FOR AN APPLIED FORCE Revision C By Tom Irvine Email: tomirvine@aol.com August 6, 8 Introduction The obective is to derive a Miles equation which gives the overall response
Srednicki Chapter 55
 Srednicki Chapter 55 QFT Problems & Solutions A. George August 3, 03 Srednicki 55.. Use equations 55.3-55.0 and A i, A j ] = Π i, Π j ] = 0 (at equal times) to verify equations 55.-55.3. This is our third
Srednicki Chapter 55 QFT Problems & Solutions A. George August 3, 03 Srednicki 55.. Use equations 55.3-55.0 and A i, A j ] = Π i, Π j ] = 0 (at equal times) to verify equations 55.-55.3. This is our third
5. Choice under Uncertainty
 5. Choice under Uncertainty Daisuke Oyama Microeconomics I May 23, 2018 Formulations von Neumann-Morgenstern (1944/1947) X: Set of prizes Π: Set of probability distributions on X : Preference relation
5. Choice under Uncertainty Daisuke Oyama Microeconomics I May 23, 2018 Formulations von Neumann-Morgenstern (1944/1947) X: Set of prizes Π: Set of probability distributions on X : Preference relation
ΕΙΣΑΓΩΓΗ ΣΤΗ ΣΤΑΤΙΣΤΙΚΗ ΑΝΑΛΥΣΗ
 ΕΙΣΑΓΩΓΗ ΣΤΗ ΣΤΑΤΙΣΤΙΚΗ ΑΝΑΛΥΣΗ ΕΛΕΝΑ ΦΛΟΚΑ Επίκουρος Καθηγήτρια Τµήµα Φυσικής, Τοµέας Φυσικής Περιβάλλοντος- Μετεωρολογίας ΓΕΝΙΚΟΙ ΟΡΙΣΜΟΙ Πληθυσµός Σύνολο ατόµων ή αντικειµένων στα οποία αναφέρονται
ΕΙΣΑΓΩΓΗ ΣΤΗ ΣΤΑΤΙΣΤΙΚΗ ΑΝΑΛΥΣΗ ΕΛΕΝΑ ΦΛΟΚΑ Επίκουρος Καθηγήτρια Τµήµα Φυσικής, Τοµέας Φυσικής Περιβάλλοντος- Μετεωρολογίας ΓΕΝΙΚΟΙ ΟΡΙΣΜΟΙ Πληθυσµός Σύνολο ατόµων ή αντικειµένων στα οποία αναφέρονται
Fourier Series. MATH 211, Calculus II. J. Robert Buchanan. Spring Department of Mathematics
 Fourier Series MATH 211, Calculus II J. Robert Buchanan Department of Mathematics Spring 2018 Introduction Not all functions can be represented by Taylor series. f (k) (c) A Taylor series f (x) = (x c)
Fourier Series MATH 211, Calculus II J. Robert Buchanan Department of Mathematics Spring 2018 Introduction Not all functions can be represented by Taylor series. f (k) (c) A Taylor series f (x) = (x c)
SCHOOL OF MATHEMATICAL SCIENCES G11LMA Linear Mathematics Examination Solutions
 SCHOOL OF MATHEMATICAL SCIENCES GLMA Linear Mathematics 00- Examination Solutions. (a) i. ( + 5i)( i) = (6 + 5) + (5 )i = + i. Real part is, imaginary part is. (b) ii. + 5i i ( + 5i)( + i) = ( i)( + i)
SCHOOL OF MATHEMATICAL SCIENCES GLMA Linear Mathematics 00- Examination Solutions. (a) i. ( + 5i)( i) = (6 + 5) + (5 )i = + i. Real part is, imaginary part is. (b) ii. + 5i i ( + 5i)( + i) = ( i)( + i)
If we restrict the domain of y = sin x to [ π, π ], the restrict function. y = sin x, π 2 x π 2
![If we restrict the domain of y = sin x to [ π, π ], the restrict function. y = sin x, π 2 x π 2 If we restrict the domain of y = sin x to [ π, π ], the restrict function. y = sin x, π 2 x π 2](/thumbs/53/31933086.jpg) Chapter 3. Analytic Trigonometry 3.1 The inverse sine, cosine, and tangent functions 1. Review: Inverse function (1) f 1 (f(x)) = x for every x in the domain of f and f(f 1 (x)) = x for every x in the
Chapter 3. Analytic Trigonometry 3.1 The inverse sine, cosine, and tangent functions 1. Review: Inverse function (1) f 1 (f(x)) = x for every x in the domain of f and f(f 1 (x)) = x for every x in the
Section 7.6 Double and Half Angle Formulas
 09 Section 7. Double and Half Angle Fmulas To derive the double-angles fmulas, we will use the sum of two angles fmulas that we developed in the last section. We will let α θ and β θ: cos(θ) cos(θ + θ)
09 Section 7. Double and Half Angle Fmulas To derive the double-angles fmulas, we will use the sum of two angles fmulas that we developed in the last section. We will let α θ and β θ: cos(θ) cos(θ + θ)
Every set of first-order formulas is equivalent to an independent set
 Every set of first-order formulas is equivalent to an independent set May 6, 2008 Abstract A set of first-order formulas, whatever the cardinality of the set of symbols, is equivalent to an independent
Every set of first-order formulas is equivalent to an independent set May 6, 2008 Abstract A set of first-order formulas, whatever the cardinality of the set of symbols, is equivalent to an independent
ΗΜΥ 220: ΣΗΜΑΤΑ ΚΑΙ ΣΥΣΤΗΜΑΤΑ Ι Ακαδημαϊκό έτος Εαρινό Εξάμηνο Κατ οίκον εργασία αρ. 2
 ΤΜΗΜΑ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΜΗΧΑΝΙΚΩΝ ΥΠΟΛΟΓΙΣΤΩΝ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΗΜΥ 220: ΣΗΜΑΤΑ ΚΑΙ ΣΥΣΤΗΜΑΤΑ Ι Ακαδημαϊκό έτος 2007-08 -- Εαρινό Εξάμηνο Κατ οίκον εργασία αρ. 2 Ημερομηνία Παραδόσεως: Παρασκευή
ΤΜΗΜΑ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΜΗΧΑΝΙΚΩΝ ΥΠΟΛΟΓΙΣΤΩΝ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΗΜΥ 220: ΣΗΜΑΤΑ ΚΑΙ ΣΥΣΤΗΜΑΤΑ Ι Ακαδημαϊκό έτος 2007-08 -- Εαρινό Εξάμηνο Κατ οίκον εργασία αρ. 2 Ημερομηνία Παραδόσεως: Παρασκευή
1 String with massive end-points
 1 String with massive end-points Πρόβλημα 5.11:Θεωρείστε μια χορδή μήκους, τάσης T, με δύο σημειακά σωματίδια στα άκρα της, το ένα μάζας m, και το άλλο μάζας m. α) Μελετώντας την κίνηση των άκρων βρείτε
1 String with massive end-points Πρόβλημα 5.11:Θεωρείστε μια χορδή μήκους, τάσης T, με δύο σημειακά σωματίδια στα άκρα της, το ένα μάζας m, και το άλλο μάζας m. α) Μελετώντας την κίνηση των άκρων βρείτε
CHAPTER 101 FOURIER SERIES FOR PERIODIC FUNCTIONS OF PERIOD
 CHAPTER FOURIER SERIES FOR PERIODIC FUNCTIONS OF PERIOD EXERCISE 36 Page 66. Determine the Fourier series for the periodic function: f(x), when x +, when x which is periodic outside this rge of period.
CHAPTER FOURIER SERIES FOR PERIODIC FUNCTIONS OF PERIOD EXERCISE 36 Page 66. Determine the Fourier series for the periodic function: f(x), when x +, when x which is periodic outside this rge of period.
CHAPTER 48 APPLICATIONS OF MATRICES AND DETERMINANTS
 CHAPTER 48 APPLICATIONS OF MATRICES AND DETERMINANTS EXERCISE 01 Page 545 1. Use matrices to solve: 3x + 4y x + 5y + 7 3x + 4y x + 5y 7 Hence, 3 4 x 0 5 y 7 The inverse of 3 4 5 is: 1 5 4 1 5 4 15 8 3
CHAPTER 48 APPLICATIONS OF MATRICES AND DETERMINANTS EXERCISE 01 Page 545 1. Use matrices to solve: 3x + 4y x + 5y + 7 3x + 4y x + 5y 7 Hence, 3 4 x 0 5 y 7 The inverse of 3 4 5 is: 1 5 4 1 5 4 15 8 3
ΚΥΠΡΙΑΚΗ ΕΤΑΙΡΕΙΑ ΠΛΗΡΟΦΟΡΙΚΗΣ CYPRUS COMPUTER SOCIETY ΠΑΓΚΥΠΡΙΟΣ ΜΑΘΗΤΙΚΟΣ ΔΙΑΓΩΝΙΣΜΟΣ ΠΛΗΡΟΦΟΡΙΚΗΣ 19/5/2007
 Οδηγίες: Να απαντηθούν όλες οι ερωτήσεις. Αν κάπου κάνετε κάποιες υποθέσεις να αναφερθούν στη σχετική ερώτηση. Όλα τα αρχεία που αναφέρονται στα προβλήματα βρίσκονται στον ίδιο φάκελο με το εκτελέσιμο
Οδηγίες: Να απαντηθούν όλες οι ερωτήσεις. Αν κάπου κάνετε κάποιες υποθέσεις να αναφερθούν στη σχετική ερώτηση. Όλα τα αρχεία που αναφέρονται στα προβλήματα βρίσκονται στον ίδιο φάκελο με το εκτελέσιμο
If we restrict the domain of y = sin x to [ π 2, π 2
 Chapter 3. Analytic Trigonometry 3.1 The inverse sine, cosine, and tangent functions 1. Review: Inverse function (1) f 1 (f(x)) = x for every x in the domain of f and f(f 1 (x)) = x for every x in the
Chapter 3. Analytic Trigonometry 3.1 The inverse sine, cosine, and tangent functions 1. Review: Inverse function (1) f 1 (f(x)) = x for every x in the domain of f and f(f 1 (x)) = x for every x in the
Macromechanics of a Laminate. Textbook: Mechanics of Composite Materials Author: Autar Kaw
 Macromechanics of a Laminate Tetboo: Mechanics of Composite Materials Author: Autar Kaw Figure 4.1 Fiber Direction θ z CHAPTER OJECTIVES Understand the code for laminate stacing sequence Develop relationships
Macromechanics of a Laminate Tetboo: Mechanics of Composite Materials Author: Autar Kaw Figure 4.1 Fiber Direction θ z CHAPTER OJECTIVES Understand the code for laminate stacing sequence Develop relationships
Quadratic Expressions
 Quadratic Expressions. The standard form of a quadratic equation is ax + bx + c = 0 where a, b, c R and a 0. The roots of ax + bx + c = 0 are b ± b a 4ac. 3. For the equation ax +bx+c = 0, sum of the roots
Quadratic Expressions. The standard form of a quadratic equation is ax + bx + c = 0 where a, b, c R and a 0. The roots of ax + bx + c = 0 are b ± b a 4ac. 3. For the equation ax +bx+c = 0, sum of the roots
( ) 2 and compare to M.
 Problems and Solutions for Section 4.2 4.9 through 4.33) 4.9 Calculate the square root of the matrix 3!0 M!0 8 Hint: Let M / 2 a!b ; calculate M / 2!b c ) 2 and compare to M. Solution: Given: 3!0 M!0 8
Problems and Solutions for Section 4.2 4.9 through 4.33) 4.9 Calculate the square root of the matrix 3!0 M!0 8 Hint: Let M / 2 a!b ; calculate M / 2!b c ) 2 and compare to M. Solution: Given: 3!0 M!0 8
Main source: "Discrete-time systems and computer control" by Α. ΣΚΟΔΡΑΣ ΨΗΦΙΑΚΟΣ ΕΛΕΓΧΟΣ ΔΙΑΛΕΞΗ 4 ΔΙΑΦΑΝΕΙΑ 1
 Main source: "Discrete-time systems and computer control" by Α. ΣΚΟΔΡΑΣ ΨΗΦΙΑΚΟΣ ΕΛΕΓΧΟΣ ΔΙΑΛΕΞΗ 4 ΔΙΑΦΑΝΕΙΑ 1 A Brief History of Sampling Research 1915 - Edmund Taylor Whittaker (1873-1956) devised a
Main source: "Discrete-time systems and computer control" by Α. ΣΚΟΔΡΑΣ ΨΗΦΙΑΚΟΣ ΕΛΕΓΧΟΣ ΔΙΑΛΕΞΗ 4 ΔΙΑΦΑΝΕΙΑ 1 A Brief History of Sampling Research 1915 - Edmund Taylor Whittaker (1873-1956) devised a
D Alembert s Solution to the Wave Equation
 D Alembert s Solution to the Wave Equation MATH 467 Partial Differential Equations J. Robert Buchanan Department of Mathematics Fall 2018 Objectives In this lesson we will learn: a change of variable technique
D Alembert s Solution to the Wave Equation MATH 467 Partial Differential Equations J. Robert Buchanan Department of Mathematics Fall 2018 Objectives In this lesson we will learn: a change of variable technique
(1) Describe the process by which mercury atoms become excited in a fluorescent tube (3)
 Q1. (a) A fluorescent tube is filled with mercury vapour at low pressure. In order to emit electromagnetic radiation the mercury atoms must first be excited. (i) What is meant by an excited atom? (1) (ii)
Q1. (a) A fluorescent tube is filled with mercury vapour at low pressure. In order to emit electromagnetic radiation the mercury atoms must first be excited. (i) What is meant by an excited atom? (1) (ii)
