Cyclic Cystine-Bridged Peptides from the Marine. Sponge Clathria basilana Induce Apoptosis in. Tumor Cells and Depolarize the Bacterial
|
|
|
- Φιλύρη Λαμπρόπουλος
- 6 χρόνια πριν
- Προβολές:
Transcript
1 Supporting information Cyclic Cystine-Bridged Peptides from the Marine Sponge Clathria basilana Induce Apoptosis in Tumor Cells and Depolarize the Bacterial Cytoplasmic Membrane Amin Mokhlesi,, Fabian Stuhldreier, Katharina W. Wex, Anne Berscheid, Rudolf Hartmann, Nidja Rehberg, Parichat Sureechatchaiyan, Chaidir Chaidir, O Matthias U. Kassack, Rainer Kalscheuer, Heike Brötz-Oesterhelt, Sebastian Wesselborg, Björn Stork, Georgios Daletos,*, and Peter Proksch*, Institute of Pharmaceutical Biology and Biotechnology, Heinrich Heine University, Universitätsstraße 1, D Düsseldorf, Germany. Department of Marine Biology, Faculty of Marine Sciences, Tarbiat Modares University, Noor, Iran. Institute of Molecular Medicine I, Medical Faculty, Heinrich Heine University, Universitätsstraße 1, D Düsseldorf, Germany. Department of Microbial Bioactive Compounds, Interfaculty Institute of Microbiology and Infection Medicine, University of Tübingen, Auf der Morgenstelle 28/E8, Tübingen, Germany. Institute of Complex Systems: Strukturbiochemie, Forschungszentrum Jülich, Wilhelm- Johnenstrasse, Jülich, Germany. 1
2 Institute of Pharmaceutical and Medicinal Chemistry, Heinrich Heine University, Universitätsstraße 1, D Düsseldorf, Germany. O Center for Pharmaceutical and Medical Technology, Agency for the Assessment and Application Technology, Jakarta, Indonesia. * Corresponding authors. Tel: proksch@uni-duesseldorf.de, georgios.daletos@uni-duesseldorf.de 2
3 Contents S1. Microcionamide C (1)... 6 S H NMR (DMSO-d 6, 600 MHz) spectrum of S1-2. HMBC NMR (DMSO-d 6, 600 MHz) spectrum of S1-3. ROESY NMR (DMSO-d 6, 600 MHz) spectrum of S1-4. HSQC NMR (DMSO-d 6, 600 MHz) spectrum of S1-5. TOCSY NMR (DMSO-d 6, 600 MHz) spectrum of S1-6. HRESIMS spectrum of S1-7. HRESIMS/MS spectrum of S2. Microcionamide D (2) S2-1. Table of 1 H (600 MHz), 13 C (150 MHz), HMBC, and ROESY NMR Data (DMSO-d 6, δ in ppm) of Microcionamide D (2) S H NMR (DMSO-d 6, 600 MHz) spectrum of S2-3. HMBC NMR (DMSO-d 6, 600 MHz) spectrum of S2-4. ROESY NMR (DMSO-d 6, 600 MHz) spectrum of S2-5. HSQC NMR (DMSO-d 6, 600 MHz) spectrum of S2-6. TOCSY NMR (DMSO-d 6, 600 MHz) spectrum of S2-7. COSY NMR (DMSO-d 6, 600 MHz) spectrum of S2-8. HRESIMS spectrum of S2-9. HRESIMS/MS spectrum of S3. Microcionamide A (3) S H NMR (MeOH-d 4, 600 MHz) spectrum of S4. Gombamide B (4) S H NMR (DMSO-d 6, 600 MHz) spectrum of S C NMR (DMSO-d 6, 600 MHz) spectrum of S4-3. HMBC NMR (DMSO-d 6, 600 MHz) spectrum of S4-4. ROESY NMR (DMSO-d 6, 600 MHz) spectrum of S4-5. HSQC NMR (DMSO-d 6, 600 MHz) spectrum of S4-6. TOCSY NMR (DMSO-d 6, 600 MHz) spectrum of
4 S4-7. COSY NMR (DMSO-d 6, 600 MHz) spectrum of S4-8. HRESIMS spectrum of S4-9. HRESIMS/MS spectrum of S5. Gombamide C (5) S5-1. Table of 1 H (600 MHz), 13 C (150 MHz), and HMBC NMR Data (DMSO-d 6, δ in ppm) of Gombamide C (5) S H NMR (DMSO-d 6, 600 MHz) spectrum of S5-3. HMBC NMR (DMSO-d 6, 600 MHz) spectrum of S5-4. HSQC NMR (DMSO-d 6, 600 MHz) spectrum of S5-5. TOCSY NMR (DMSO-d 6, 600 MHz) spectrum of S5-6. HRESIMS spectrum of S5-7. HRESIMS/MS spectrum of S6. Gombamide D (6) S H NMR (DMSO-d 6, 600 MHz) spectrum of S C NMR (DMSO-d 6, 150 MHz) spectrum of S6-3. HMBC NMR (DMSO-d 6, 600 MHz) spectrum of S6-4. ROESY NMR (DMSO-d 6, 600 MHz) spectrum of S6-5. HSQC NMR (DMSO-d 6, 600 MHz) spectrum of S6-6. COSY NMR (DMSO-d 6, 600 MHz) spectrum of S6-7. HRESIMS spectrum of S6-8. HRESIMS/MS spectrum of S7. (E)-2-amino-3-methyl-N-styrylbutanamide (7) S H NMR (MeOH-d4, 600 MHz) spectrum of S C NMR (MeOH-d4, 600 MHz) spectrum of S7-3. HMBC NMR (MeOH-d4, 600 MHz) spectrum of S7-4. COSY NMR (MeOH-d4, 600 MHz) spectrum of S7-5. HRESIMS spectrum of S8. Marfey s analysis S8-1. Table of HPLC (C 18 ) retention times of acid hydrolysates of 1 7 using Marfey s method
5 S8-2. Table of HPLC (C 4 ) retention times for L-Ile and L-allo-Ile of 1 5 using Marfey s method S HPLC chromatogram of derivatized L- and D-allo-Ile standards S HPLC chromatogram of derivatized D-allo-Ile, L-allo-Ile, and L-Ile standards S9. Structures of known compounds S10. Bioactivity results S10-1. Cytotoxicity effects of 1 3 on human lymphoma and leukemia cell lines S10-2. Table of antibacterial activity of new 1 7 and known (8 17) compounds reported as minimal inhibitory concentration (MIC) a
6 S1. Microcionamide C (1) S H NMR (DMSO-d 6, 600 MHz) spectrum of 1 6
7 S1-2. HMBC NMR (DMSO-d 6, 600 MHz) spectrum of 1 7
8 S1-3. ROESY NMR (DMSO-d 6, 600 MHz) spectrum of 1 8
9 S1-4. HSQC NMR (DMSO-d 6, 600 MHz) spectrum of 1 9
10 S1-5. TOCSY NMR (DMSO-d 6, 600 MHz) spectrum of 1 10
11 S1-6. HRESIMS spectrum of 1 11
12 S1-7. HRESIMS/MS spectrum of 1 12
13 S2. Microcionamide D (2) S2-1. Table of 1 H (600 MHz), 13 C (150 MHz), HMBC, and ROESY NMR Data (DMSO-d 6, δ in ppm) of Microcionamide D (2) Unit Position δ c, a type δ H (J in Hz) HMBC ROESY b Z-PEA NH 9.46, d (10.5) Cys 1 -CO Cys 1 -α, Cys 1 -NH α 121.1, CH 6.72, d (10.0) Cys 1 -CO, Z-PEA-β, Z-PEA-1 β 110.8, CH 5.74, d (10.0) Z-PEA-α, Z-PEA-1, Z-PEA- 2/6 aromatic 1: 134.7, C 2: 127.9, CH 7.35 c Z-PEA-β, Z-PEA-1, Z-PEA- 6, Z-PEA-4 3: 128.3, CH 7.35 c Z-PEA-1, Z-PEA-5, Z-PEA-4 4: 126.4, CH 7.24, m Z-PEA-2/6 5: 128.3, CH 7.35 c Z-PEA-1, Z-PEA-3, Z-PEA-4 6: 127.9, CH 7.35 c Z-PEA-β, Z-PEA-1, Z-PEA- 2, Z-PEA-4 Cys 1 NH 8.24, br s Z-PEA- NH, Ile 2 -α CO 168.0, C α 51.7, CH 4.62 c Cys 1 -CO, Ile 2 -CO, Cys 1 -β Z-PEA-NH β 40.9, CH , br t (11.0); 3.28 c Cys 1 -α, Cys 1 -CO Ile 2 NH 7.77, br s CO 170.9, C α 57.2, CH 4.01, br t (8.5) Ile 2 -CO, Ile 3 -CO, Ile 2 -β, Ile 2 - γ, Ile 2 -γ Cys 1 -NH β 34.3, CH 1.77, m Ile 2 -α γ 24.3, CH , m; 1.48, m Ile 2 -α γ 15.1, CH c Ile 2 -α 13
14 δ 11.0, CH c,d Ile 3 NH n.d. e CO α n.d. e 4.08 c β 36.4, CH 1.79, m γ 24.4, CH , m; 1.51, m γ 15.2, CH c δ 11.0, CH c,d Ile 4 NH 7.39 c CO n.d. e α 57.0, CH 4.31, br s Ile 5 -β β 35.9, CH 2.08, br s γ 23.5, CH , m; 1.33, m γ 15.6, CH c δ 11.1, CH c,d Ile 5 NH 8.59, d (9.3) Cys 6 -CO Cys 6 -α CO 170.4, C α 58.0, CH 4.17, dd (9.7, 7.5) Ile 5 -CO, Cys 6 -CO, Ile 5 -β, Ile 5 -γ, Ile 5 -γ Cys 6 -α β 36.4, CH 1.80, m Ile 5 -α Ile 4 -α, Cys 6 -NH, Cys 6 -α γ 24.3, CH , m; 1.41, m Ile 5 -α γ 15.2, CH c Ile 5 -α δ 10.9, CH c,d Cys 6 NH 8.82, br d (7.0) Ile 7 -CO Ile 7 -α, Ile 7 - β, Ile 7 -γ, Ile 5 -β CO 168.6, C 14
15 α 52.5, CH 4.61 c Ile 7 -CO Ile 5 -NH, Ile 7 -α, Ile 5 - α β 40.2, CH , m; 3.35, m Ile 7 NH , br s CO 167.8, C α 56.1, CH 3.66, br t (5.0) Ile 7 -CO, Ile 7 -β, Ile 7 -γ, Ile 7 -γ Cys 6 -α, Cys 6 -NH β 36.2, CH 1.79, m Ile 7 -α Cys 6 -NH γ 23.4, CH , m; 1.46, m Ile 7 -α Cys 6 -NH γ 14.2, CH c Ile 7 -α δ 10.8, CH c.d a Data extracted from HSQC and HMBC spectra. b Sequential NOEs. c Signal overlap prevents determination of couplings. d Assignments within the same column may be interchanged. e n.d. : not detected 15
16 S H NMR (DMSO-d 6, 600 MHz) spectrum of 2 16
17 S2-3. HMBC NMR (DMSO-d 6, 600 MHz) spectrum of 2 17
18 S2-4. ROESY NMR (DMSO-d 6, 600 MHz) spectrum of 2 18
19 S2-5. HSQC NMR (DMSO-d 6, 600 MHz) spectrum of 2 19
20 S2-6. TOCSY NMR (DMSO-d 6, 600 MHz) spectrum of 2 20
21 S2-7. COSY NMR (DMSO-d 6, 600 MHz) spectrum of 2 21
22 S2-8. HRESIMS spectrum of 2 22
23 S2-9. HRESIMS/MS spectrum of 2 23
24 S3. Microcionamide A (3) S H NMR (MeOH-d 4, 600 MHz) spectrum of 3 24
25 S4. Gombamide B (4) S H NMR (DMSO-d 6, 600 MHz) spectrum of 4 25
26 S C NMR (DMSO-d 6, 600 MHz) spectrum of 4 26
27 S4-3. HMBC NMR (DMSO-d 6, 600 MHz) spectrum of 4 27
28 S4-4. ROESY NMR (DMSO-d 6, 600 MHz) spectrum of 4 28
29 S4-5. HSQC NMR (DMSO-d 6, 600 MHz) spectrum of 4 29
30 S4-6. TOCSY NMR (DMSO-d 6, 600 MHz) spectrum of 4 30
31 S4-7. COSY NMR (DMSO-d 6, 600 MHz) spectrum of 4 31
32 S4-8. HRESIMS spectrum of 4 32
33 S4-9. HRESIMS/MS spectrum of 4 33
34 S5. Gombamide C (5) S5-1. Table of 1 H (600 MHz), 13 C (150 MHz), and HMBC NMR Data (DMSO-d 6, δ in ppm) of Gombamide C (5) Unit Position δ c, a type δ H (J in Hz) HMBC Cys 1 NH 7.89, br d (8.6) Phe-CO CO 168.4, C α 51.5, CH 4.76 b β 41.7, CH , dd (14.4, 12.4); 3.14, dd (14.4, 3.4) Z-PEA NH 9.72, br d (10.1) Cys 1 -CO α 121.3, CH 6.71, dd (10.1, 9.7) Cys 1 -CO, Z-PEA-β β 111.6, CH 5.79, d (9.7) Z-PEA-α, Z-PEA-2/6, Z-PEA-1 aromatic 1: 135.2, C 2: 128.1, CH 7.38 b 3: 128.4, CH 7.38 b 4: 126.6, CH 7.38 b 5: 128.4, CH 7.38 b 6: 128.1, CH 7.38 b Phe NH 9.00, br d (8.8) CO 170.5, C α 53.4, CH 4.75 b Phe-CO, Phe-β, Pro 3 -CO β 34.1, CH , m Phe-α, Phe-1, Phe-2/6 aromatic 1: 138.0, C 2: 129.1, CH 7.40, br d (7.5) Phe-β, Phe-6, Phe-4 3: 128.0, CH 7.28, br t (7.5) Phe-1, Phe-5 4: 126.1, CH 7.19, br t (7.5) Phe-2/6 5: 128.0, CH 7.28, br t (7.5) Phe-1, Phe-3 6: 129.1, CH 7.40, br d (7.5) Phe-β, Phe-6, Phe-4 34
35 Pro 3 CO 171.2, C α 60.7, CH 4.34, d (8.0) Pro 3 -CO, Pro 4 -CO, Pro 3 -β, Pro 3 -γ, Pro 3 -δ β 31.1, CH , m; 1.87, m γ 20.0, CH , m; 0.58, m δ 45.9, CH , m Pro 4 CO 169.2, C α 59.0, CH 4.80, dd (8.8, 3.0) Pro 4 -γ β 30.1, CH , m; 2.22, m γ 21.6, CH , m δ 46.7, CH , m; 3.47, m Cys 5 NH 8.00, br d (10.0) Leu-CO CO 167.9, C α 48.1, CH 4.55, td (10.0, 5.1) Leu-CO, Cys 5 -CO, Cys 5 -β β 37.9, CH , dd (13.0, 10.0); 3.20, dd (13.0, 5.1) Cys 5 -α, Cys 5 -CO Leu NH 8.04, br d (9.9) CO 170.3, C α 50.3, CH 4.47, ddd (9.9, 8.6, 6.1) Leu-CO, Leu-β, Ile-CO β 41.0, CH , m Leu-α, Leu-δ, Leu-δ, Leu-CO γ 23.8, CH 1.62, m δ 21.2, CH , d (6.5) Leu-β, Leu-δ δ' 23.2, CH , d (6.6) Leu-β, Leu-δ Ile NH 8.06, br d (8.7) CO 170.5, C α 56.8, CH 4.15, t (8.5) Glp-CO, Ile-CO, Ile-β, Ile-γ, Ile-γ β 35.4, CH 1.72, m Ile-α, Ile-δ γ 24.0, CH , m; 1.42, m Ile-α, Ile-δ 35
36 γ 15.2, CH , d (6.8) Ile-α δ 10.2, CH , t (7.4) Ile-β, Ile-γ Glp NH 7.80, s Glp-CO, Glp-α, Glp-β, Glp-γ CO CO 172.2, C 177.4, C α 55.0, CH 4.09, dd (8.7, 3.6) Glp-CO, Glp-β, Glp-γ β 25.2, CH , m; 2.22, m Glp-CO, Glp-α γ 28.8, CH , m; 2.09, m Glp-α a Data extracted from HSQC and HMBC spectra. b Signal overlap prevents determination of couplings. 36
37 S H NMR (DMSO-d 6, 600 MHz) spectrum of 5 37
38 S5-3. HMBC NMR (DMSO-d 6, 600 MHz) spectrum of 5 38
39 S5-4. HSQC NMR (DMSO-d 6, 600 MHz) spectrum of 5 39
40 S5-5. TOCSY NMR (DMSO-d 6, 600 MHz) spectrum of 5 40
41 S5-6. HRESIMS spectrum of 5 41
42 S5-7. HRESIMS/MS spectrum of 5 42
43 S6. Gombamide D (6) S H NMR (DMSO-d 6, 600 MHz) spectrum of 6 43
44 S C NMR (DMSO-d 6, 150 MHz) spectrum of 6 44
45 S6-3. HMBC NMR (DMSO-d 6, 600 MHz) spectrum of 6 45
46 S6-4. ROESY NMR (DMSO-d 6, 600 MHz) spectrum of 6 46
47 S6-5. HSQC NMR (DMSO-d 6, 600 MHz) spectrum of 6 47
48 S6-6. COSY NMR (DMSO-d 6, 600 MHz) spectrum of 6 48
49 S6-7. HRESIMS spectrum of 6 49
50 S6-8. HRESIMS/MS spectrum of 6 50
51 S7. (E)-2-amino-3-methyl-N-styrylbutanamide (7) S H NMR (MeOH-d4, 600 MHz) spectrum of 7 51
52 S C NMR (MeOH-d4, 600 MHz) spectrum of 7 52
53 S7-3. HMBC NMR (MeOH-d4, 600 MHz) spectrum of 7 53
54 S7-4. COSY NMR (MeOH-d4, 600 MHz) spectrum of 7 54
55 S7-5. HRESIMS spectrum of 7 55
56 S8. Marfey s analysis S8-1. Table of HPLC (C 18 ) retention times of acid hydrolysates of 1 7 using Marfey s method Amino acid Standard L-Cys D-Cys 21.2 L-Ile D-Ile 24.2 L-Leu D-Leu 24.3 L-Val 14.5 D-Val L-Pro D-Pro 11.4 L-Glu D-Glu 14.2 L-Phe D-Phe
57 S8-2. Table of HPLC (C 4 ) retention times for L-Ile and L-allo-Ile of 1 5 using Marfey s method Amino acid Standard L-Ile L-allo-Ile Marfey 160min 25C #4 Marfey 160min DLallo UV_VIS_1 mau WVL:235 nm L-allo-Ile D-allo-Ile min 20,0 30,0 40,0 50,0 60,0 70,0 80,0 S HPLC chromatogram of derivatized L- and D-allo-Ile standards Marfey 160min 25C #3 Marfey 160min DLallo+LIle 2 UV_VIS_1 mau WVL:235 nm L-allo-Ile/L-Ile D-allo-Ile 0-60 min 20,0 30,0 40,0 50,0 60,0 70,0 80,0 S HPLC chromatogram of derivatized D-allo-Ile, L-allo-Ile, and L-Ile standards 57
58 S9. Structures of known compounds S9-1. Structures of known compounds, including 1H-indole-3-carbaldehyde (8), 1H-indole-3-carboxylic acid (9), 6-bromo-1H-indole-3-carbaldehyde (10), 6-bromo-1H-indole-3-carboxylic acid (11), methyl 6- bromo-1h-indole-3-carboxylate (12), ethyl 6-bromo-1H-indole-3-carboxylate (13), 7-bromo-4(1H)- quinolinone (14), (3-(2-(4-hydroxyphenyl)-2-oxoethyl)-5,6-dihydropyridin-2(1H)-one) (15), 2- deoxythymidine (16), and 4-hydroxybenzoic acid (17). 58
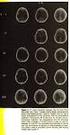
59 S10. Bioactivity results S10-1. Cytotoxicity effects of 1 3 on human lymphoma and leukemia cell lines, measured by MTT assay. (A) Ramos (Burkitt s lymphoma), (B) Jurkat J16 (acute T cell leukemia), (C) Nomo-1 (acute myeloid leukemia) and (D) HL-60 (acute promyelocytic leukemia) cells were seeded at a density of cells/ml and incubated with increasing concentrations of 1, 2 and 3, respectively. Cells treated with DMSO (0.1% v/v) for 24 h were used as the negative control. After an incubation period of 24 h cell viability was monitored using the MTT assay as described in methods. Relative viability in DMSOtreated control cells was set to 100%. Data points shown are the mean of triplicates, error bars = SD. Viability and IC 50 values (IC 50 = half maximal inhibitory concentration) were calculated using Prism 6 (GraphPad Software). 59
60 S10-2. Table of antibacterial activity of new 1 7 and known (8 17) compounds reported as minimal inhibitory concentration (MIC) a Compound Staphylococcus aureus ATCC Enterococcus faecium BM Escherichia coli ATCC Klebsiella pneumoniae ATCC Enterobacter aerogenes ATCC Pseudomonas aeruginosa ATCC Acinetobacter baumannii Mycobacterium tuberculosis H37Rv > 100 > 100 > 100 > 100 > n.i. b n.i. n.i. n.i. n.i. n.i. n.i. n.i c 12.5 > 100 > 100 > 100 > 100 > > 50 > 50 > 100 > 100 > 100 > 100 > 100 > n.i. n.i. n.i. n.i. n.i. n.i. n.i. n.i > 50 > 50 > 100 > 100 > 100 > 100 > 100 n.i. ciprofloxacin n.i. doxycycline n.i. n.i. n.i. vancomycin 1 4 > 64 > 64 > 64 > 64 > 64 n.i. a MIC values are reported for test compounds in µm and for reference antibiotics in µg/ml and indicate the lowest concentration fully inhibiting visible bacterial growth. b n.i. : not investigated. c Growth reduction was already observed at 3.1 µm 60
SUPPORTING INFORMATION
 SUPPORTING INFORMATION Trichodermides A E: New Peptaibols isolated from Australian Termite Nestderived Fungus Trichoderma virens CMB-TN16 Wei-Hua Jiao,, Zeinab Khalil, Pradeep Dewapriya, Angela A. Salim,
SUPPORTING INFORMATION Trichodermides A E: New Peptaibols isolated from Australian Termite Nestderived Fungus Trichoderma virens CMB-TN16 Wei-Hua Jiao,, Zeinab Khalil, Pradeep Dewapriya, Angela A. Salim,
SUPPORTING INFORMATION
 SUPPORTING INFORMATION Heronamides A C, new polyketide macrolactams from an Australian marine-derived Streptomyces sp. A biosynthetic case for synchronized tandem electrocyclization. Ritesh Raju, Andrew
SUPPORTING INFORMATION Heronamides A C, new polyketide macrolactams from an Australian marine-derived Streptomyces sp. A biosynthetic case for synchronized tandem electrocyclization. Ritesh Raju, Andrew
Comparison of carbon-sulfur and carbon-amine bond in therapeutic drug: -S-aromatic heterocyclic podophyllum derivatives display antitumor activity
 Comparison of carbon-sulfur and carbon-amine bond in therapeutic drug: -S-aromatic heterocyclic podophyllum derivatives display antitumor activity Jian-Long Li 1,a, Wei Zhao 1,a, Chen Zhou 1,a, Ya-Xuan
Comparison of carbon-sulfur and carbon-amine bond in therapeutic drug: -S-aromatic heterocyclic podophyllum derivatives display antitumor activity Jian-Long Li 1,a, Wei Zhao 1,a, Chen Zhou 1,a, Ya-Xuan
Malgorzata Korycka-Machala, Marcin Nowosielski, Aneta Kuron, Sebastian Rykowski, Agnieszka Olejniczak, Marcin Hoffmann and Jaroslaw Dziadek
 Molecules 2017, 21, 154; doi:10.3390/molecules22010154 Supplementary Materials: Naphthalimides Selectively Inhibit the Activity of Bacterial, Replicative DNA Ligases and Display Bactericidal Effect against
Molecules 2017, 21, 154; doi:10.3390/molecules22010154 Supplementary Materials: Naphthalimides Selectively Inhibit the Activity of Bacterial, Replicative DNA Ligases and Display Bactericidal Effect against
Supporting Information
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2018 Supporting Information Crotonols A and B, Two Rare Tigliane Diterpenoid
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2018 Supporting Information Crotonols A and B, Two Rare Tigliane Diterpenoid
New Cytotoxic Constituents from the Red Sea Soft Coral Nephthea sp.
 SUPPLEMENTARY MATERIAL New Cytotoxic Constituents from the Red Sea Soft Coral Nephthea sp. Mohamed-Elamir F. Hegazy a,*, Amira M. Gamal-Eldeen b, Tarik A. Mohamed a, Montaser A. Alhammady c, Abuzeid A.
SUPPLEMENTARY MATERIAL New Cytotoxic Constituents from the Red Sea Soft Coral Nephthea sp. Mohamed-Elamir F. Hegazy a,*, Amira M. Gamal-Eldeen b, Tarik A. Mohamed a, Montaser A. Alhammady c, Abuzeid A.
Eremophila spp. by Combined use of Dual High-Resolution PTP1B and α- Glucosidase Inhibition Profiling and HPLC-HRMS-SPE-NMR
 Supporting Information for: Identification of PTP1B and α-glucosidase Inhibitory Serrulatanes from Eremophila spp. by Combined use of Dual High-Resolution PTP1B and α- Glucosidase Inhibition Profiling
Supporting Information for: Identification of PTP1B and α-glucosidase Inhibitory Serrulatanes from Eremophila spp. by Combined use of Dual High-Resolution PTP1B and α- Glucosidase Inhibition Profiling
Supporting Information. Asymmetric Binary-acid Catalysis with Chiral. Phosphoric Acid and MgF 2 : Catalytic
 Supporting Information Asymmetric Binary-acid Catalysis with Chiral Phosphoric Acid and MgF 2 : Catalytic Enantioselective Friedel-Crafts Reactions of β,γ- Unsaturated-α-Ketoesters Jian Lv, Xin Li, Long
Supporting Information Asymmetric Binary-acid Catalysis with Chiral Phosphoric Acid and MgF 2 : Catalytic Enantioselective Friedel-Crafts Reactions of β,γ- Unsaturated-α-Ketoesters Jian Lv, Xin Li, Long
Supporting Information for. A New Diketopiperazine, Cyclo-(4-S-Hydroxy-R-Proline-R-Isoleucine), from an Australian Specimen of the Sponge
 1 Supporting Information for A New Diketopiperazine, Cyclo-(4-S-Hydroxy-R-Proline-R-Isoleucine), from an Australian Specimen of the Sponge Stelletta Sp. Simon P. B. Ovenden, Jonathan L. Nielson, Catherine
1 Supporting Information for A New Diketopiperazine, Cyclo-(4-S-Hydroxy-R-Proline-R-Isoleucine), from an Australian Specimen of the Sponge Stelletta Sp. Simon P. B. Ovenden, Jonathan L. Nielson, Catherine
Supporting Information for. Update of spectroscopic data for 4-hydroxyldictyolactone and dictyol E isolated from a Halimeda stuposa - Dictyota
 1 Supporting Information for Update of spectroscopic data for 4-hydroxyldictyolactone and dictyol E isolated from a Halimeda stuposa - Dictyota sp. Assemblage Simon P. B. Ovenden, Jonathan L. Nielson,
1 Supporting Information for Update of spectroscopic data for 4-hydroxyldictyolactone and dictyol E isolated from a Halimeda stuposa - Dictyota sp. Assemblage Simon P. B. Ovenden, Jonathan L. Nielson,
Synthesis and Biological Evaluation of Novel Acyclic and Cyclic Glyoxamide derivatives as Bacterial Quorum Sensing and Biofilm Inhibitors
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2017 Synthesis and Biological Evaluation of Novel Acyclic and Cyclic Glyoxamide
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2017 Synthesis and Biological Evaluation of Novel Acyclic and Cyclic Glyoxamide
A new ent-kaurane diterpene from Euphorbia stracheyi Boiss
 SUPPLEMENTARY MATERIAL A new ent-kaurane diterpene from Euphorbia stracheyi Boiss Tie Liu a, Qian Liang a,b, Na-Na Xiong a, Lin-Feng Dai a, Jun-Ming Wang a,b, Xiao-Hui Ji c, Wen-Hui Xu a, * a Key Laboratory
SUPPLEMENTARY MATERIAL A new ent-kaurane diterpene from Euphorbia stracheyi Boiss Tie Liu a, Qian Liang a,b, Na-Na Xiong a, Lin-Feng Dai a, Jun-Ming Wang a,b, Xiao-Hui Ji c, Wen-Hui Xu a, * a Key Laboratory
SUPPORTING INFORMATION
 SUPPRTING INFRMATIN Per(-guanidino--deoxy)cyclodextrins: Synthesis, characterisation and binding behaviour toward selected small molecules and DNA. Nikolaos Mourtzis, a Kyriaki Eliadou, a Chrysie Aggelidou,
SUPPRTING INFRMATIN Per(-guanidino--deoxy)cyclodextrins: Synthesis, characterisation and binding behaviour toward selected small molecules and DNA. Nikolaos Mourtzis, a Kyriaki Eliadou, a Chrysie Aggelidou,
Supporting Information. Partial thioamide scan on the lipopeptaibiotic trichogin GA IV. Effects on
 Supporting Information for Partial thioamide scan on the lipopeptaibiotic trichogin GA IV. Effects on folding and bioactivity Marta De Zotti 1, Barbara Biondi 1, Cristina Peggion 1, Matteo De Poli 1, Haleh
Supporting Information for Partial thioamide scan on the lipopeptaibiotic trichogin GA IV. Effects on folding and bioactivity Marta De Zotti 1, Barbara Biondi 1, Cristina Peggion 1, Matteo De Poli 1, Haleh
Direct Transformation of Ethylarenes into Primary Aromatic Amides with N-Bromosuccinimide and I 2 -aq NH 3
 Supporting Information Direct Transformation of Ethylarenes into Primary Aromatic Amides with N-Bromosuccinimide and I 2 -aq NH 3 Shohei Shimokawa, Yuhsuke Kawagoe, Katsuhiko Moriyama, Hideo Togo* Graduate
Supporting Information Direct Transformation of Ethylarenes into Primary Aromatic Amides with N-Bromosuccinimide and I 2 -aq NH 3 Shohei Shimokawa, Yuhsuke Kawagoe, Katsuhiko Moriyama, Hideo Togo* Graduate
Supporting Information One-Pot Approach to Chiral Chromenes via Enantioselective Organocatalytic Domino Oxa-Michael-Aldol Reaction
 Supporting Information ne-pot Approach to Chiral Chromenes via Enantioselective rganocatalytic Domino xa-michael-aldol Reaction Hao Li, Jian Wang, Timiyin E-Nunu, Liansuo Zu, Wei Jiang, Shaohua Wei, *
Supporting Information ne-pot Approach to Chiral Chromenes via Enantioselective rganocatalytic Domino xa-michael-aldol Reaction Hao Li, Jian Wang, Timiyin E-Nunu, Liansuo Zu, Wei Jiang, Shaohua Wei, *
Supporting Information
 Chloromethylhalicyclamine B, a Marine-Derived Protein Kinase CK1δ/ε Inhibitor Germana Esposito, Marie-Lise Bourguet-Kondracki, * Linh H. Mai, Arlette Longeon, Roberta Teta, Laurent Meijer, Rob Van Soest,
Chloromethylhalicyclamine B, a Marine-Derived Protein Kinase CK1δ/ε Inhibitor Germana Esposito, Marie-Lise Bourguet-Kondracki, * Linh H. Mai, Arlette Longeon, Roberta Teta, Laurent Meijer, Rob Van Soest,
Copper-catalyzed formal O-H insertion reaction of α-diazo-1,3-dicarb- onyl compounds to carboxylic acids with the assistance of isocyanide
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Copper-catalyzed formal O-H insertion reaction of α-diazo-1,3-dicarb- onyl compounds to carboxylic
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Copper-catalyzed formal O-H insertion reaction of α-diazo-1,3-dicarb- onyl compounds to carboxylic
Electronic Supplementary Information
 Electronic Supplementary Information NbCl 3 -catalyzed [2+2+2] intermolecular cycloaddition of alkynes and alkenes to 1,3-cyclohexadiene derivatives Yasushi Obora,* Keisuke Takeshita and Yasutaka Ishii*
Electronic Supplementary Information NbCl 3 -catalyzed [2+2+2] intermolecular cycloaddition of alkynes and alkenes to 1,3-cyclohexadiene derivatives Yasushi Obora,* Keisuke Takeshita and Yasutaka Ishii*
Supporting Information. Copyright Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2006
 Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 A new route to enantiopure b-aryl-substituted b-amino acids and 4-aryl-substituted b-lactams through lipase-catalyzed
Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 A new route to enantiopure b-aryl-substituted b-amino acids and 4-aryl-substituted b-lactams through lipase-catalyzed
Electronic Supporting Information. 3-Aminothiophenecarboxylic acid (3-Atc)-induced folding in peptides
 Electronic Supplementary Material (ESI for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2016 Electronic Supporting Information
Electronic Supplementary Material (ESI for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2016 Electronic Supporting Information
Supporting Information
 Supporting Information Molecular Cage Impregnated Palladium Nanoparticles: Efficient, Additive-Free Heterogeneous Catalysts for Cyanation of Aryl Halides. Bijnaneswar Mondal, Koushik Acharyya, Prodip Howlader
Supporting Information Molecular Cage Impregnated Palladium Nanoparticles: Efficient, Additive-Free Heterogeneous Catalysts for Cyanation of Aryl Halides. Bijnaneswar Mondal, Koushik Acharyya, Prodip Howlader
Supporting Information. Research Center for Marine Drugs, Department of Pharmacy, State Key Laboratory
 Supporting Information Dysiherbols A C and Dysideanone E, Cytotoxic and NF-κB Inhibitory Tetracyclic Meroterpenes from a Dysidea sp. Marine Sponge Wei-Hua Jiao,, Guo-Hua Shi,, Ting-Ting Xu,, Guo-Dong Chen,
Supporting Information Dysiherbols A C and Dysideanone E, Cytotoxic and NF-κB Inhibitory Tetracyclic Meroterpenes from a Dysidea sp. Marine Sponge Wei-Hua Jiao,, Guo-Hua Shi,, Ting-Ting Xu,, Guo-Dong Chen,
Synthesis, structural studies and stability of the model, cysteine containing DNA-protein cross-links
 Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2017 ELECTRONIC SUPPLEMENTARY INFORMATION
Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2017 ELECTRONIC SUPPLEMENTARY INFORMATION
Enantioselective Organocatalytic Michael Addition of Isorhodanines. to α, β-unsaturated Aldehydes
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2016 Enantioselective Organocatalytic Michael Addition of Isorhodanines to α,
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2016 Enantioselective Organocatalytic Michael Addition of Isorhodanines to α,
Regioselectivity in the Stille coupling reactions of 3,5- dibromo-2-pyrone.
 Regioselectivity in the Stille coupling reactions of 3,5- dibromo-2-pyrone. Won-Suk Kim, Hyung-Jin Kim and Cheon-Gyu Cho Department of Chemistry, Hanyang University, Seoul 133-791, Korea Experimental Section
Regioselectivity in the Stille coupling reactions of 3,5- dibromo-2-pyrone. Won-Suk Kim, Hyung-Jin Kim and Cheon-Gyu Cho Department of Chemistry, Hanyang University, Seoul 133-791, Korea Experimental Section
Supporting Information. Experimental section
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Experimental section General. Proton nuclear magnetic resonance ( 1
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Experimental section General. Proton nuclear magnetic resonance ( 1
SUPPLEMENTARY MATERIAL
 10.1071/CH16014_AC The Authors 2016 Australian Journal of Chemistry 2016, 69(9), 1049-1053 SUPPLEMENTARY MATERIAL A Green Approach for the Synthesis of Novel 7,11-Dihydro-6H-chromeno[3,4- e]isoxazolo[5,4-b]pyridin-6-one
10.1071/CH16014_AC The Authors 2016 Australian Journal of Chemistry 2016, 69(9), 1049-1053 SUPPLEMENTARY MATERIAL A Green Approach for the Synthesis of Novel 7,11-Dihydro-6H-chromeno[3,4- e]isoxazolo[5,4-b]pyridin-6-one
The Sponge-Derived Fijianolide Polyketide Class: Further Evaluation of Their Structural and Cytotoxicity Properties
 The Sponge-Derived Fijianolide Polyketide Class: Further Evaluation of Their Structural and Cytotoxicity Properties Tyler A. Johnson, Karen Tenney, Robert H. Cichewicz, Brandon I. Morinaka, Kimberly N.
The Sponge-Derived Fijianolide Polyketide Class: Further Evaluation of Their Structural and Cytotoxicity Properties Tyler A. Johnson, Karen Tenney, Robert H. Cichewicz, Brandon I. Morinaka, Kimberly N.
Mandelamide-Zinc Catalyzed Alkyne Addition to Heteroaromatic Aldehydes
 1 Mandelamide-Zinc Catalyzed Alkyne Addition to Heteroaromatic Aldehydes Gonzalo Blay, Isabel Fernández, Alícia Marco-Aleixandre, and José R. Pedro Departament de Química Orgànica, Facultat de Química,
1 Mandelamide-Zinc Catalyzed Alkyne Addition to Heteroaromatic Aldehydes Gonzalo Blay, Isabel Fernández, Alícia Marco-Aleixandre, and José R. Pedro Departament de Química Orgànica, Facultat de Química,
Supporting Information. Palladium Complexes with Bulky Diphosphine. Synthesis of (Bio-) Adipic Acid from Pentenoic. Acid Mixtures.
 Supporting Information Palladium Complexes with Bulky Diphosphine Ligands as Highly Selective Catalysts for the Synthesis of (Bio-) Adipic Acid from Pentenoic Acid Mixtures. Choon Heng Low, James D. Nobbs,*
Supporting Information Palladium Complexes with Bulky Diphosphine Ligands as Highly Selective Catalysts for the Synthesis of (Bio-) Adipic Acid from Pentenoic Acid Mixtures. Choon Heng Low, James D. Nobbs,*
Structure-Metabolism-Relationships in the microsomal clearance of. piperazin-1-ylpyridazines
 Electronic Supplementary Material (ESI) for MedChemComm. This journal is The Royal Society of Chemistry 2017 Structure-Metabolism-Relationships in the microsomal clearance of piperazin-1-ylpyridazines
Electronic Supplementary Material (ESI) for MedChemComm. This journal is The Royal Society of Chemistry 2017 Structure-Metabolism-Relationships in the microsomal clearance of piperazin-1-ylpyridazines
Lewis Acid Catalyzed Propargylation of Arenes with O-Propargyl Trichloroacetimidate: Synthesis of 1,3-Diarylpropynes
 Supporting Information for Lewis Acid Catalyzed Propargylation of Arenes with O-Propargyl Trichloroacetimidate: Synthesis of 1,3-Diarylpropynes Changkun Li and Jianbo Wang* Beijing National Laboratory
Supporting Information for Lewis Acid Catalyzed Propargylation of Arenes with O-Propargyl Trichloroacetimidate: Synthesis of 1,3-Diarylpropynes Changkun Li and Jianbo Wang* Beijing National Laboratory
Natural Products Research Institute, College of Pharmacy, Seoul National University, 1 Gwanak-ro, Gwanak-gu, Seoul 08826, Republic of Korea
 Supporting Information Nicrophorusamides A and B, Antibacterial Chlorinated Cyclic Peptides from a Gut Bacterium of the Carrion Beetle Nicrophorus concolor Yern-Hyerk Shin, Suhyun Bae, Jaehoon Sim,,ǁ Joonseong
Supporting Information Nicrophorusamides A and B, Antibacterial Chlorinated Cyclic Peptides from a Gut Bacterium of the Carrion Beetle Nicrophorus concolor Yern-Hyerk Shin, Suhyun Bae, Jaehoon Sim,,ǁ Joonseong
A facile and general route to 3-((trifluoromethyl)thio)benzofurans and 3-((trifluoromethyl)thio)benzothiophenes
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 A facile and general route to 3-((trifluoromethyl)thio)benzofurans and 3-((trifluoromethyl)thio)benzothiophenes
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 A facile and general route to 3-((trifluoromethyl)thio)benzofurans and 3-((trifluoromethyl)thio)benzothiophenes
A strategy for the identification of combinatorial bioactive compounds. contributing to the holistic effect of herbal medicines
 1 2 Supplementary information 3 4 A strategy for the identification of combinatorial bioactive compounds contributing to the holistic effect of herbal medicines 5 6 Fang Long 1, Hua Yang 1, Yanmin Xu,
1 2 Supplementary information 3 4 A strategy for the identification of combinatorial bioactive compounds contributing to the holistic effect of herbal medicines 5 6 Fang Long 1, Hua Yang 1, Yanmin Xu,
c Key words: cultivation of blood, two-sets blood culture, detection rate of germ Vol. 18 No
 2008 245 2 1) 1) 2) 3) 4) 1) 1) 1) 1) 1), 2) 1) 2) 3) / 4) 20 3 24 20 8 18 2001 2 2 2004 2 59.0 2002 1 2004 12 3 2 22.1 1 14.0 (CNS), Bacillus c 2 p 0.01 2 1 31.3 41.9 21.4 1 2 80 CNS 2 1 74.3 2 Key words:
2008 245 2 1) 1) 2) 3) 4) 1) 1) 1) 1) 1), 2) 1) 2) 3) / 4) 20 3 24 20 8 18 2001 2 2 2004 2 59.0 2002 1 2004 12 3 2 22.1 1 14.0 (CNS), Bacillus c 2 p 0.01 2 1 31.3 41.9 21.4 1 2 80 CNS 2 1 74.3 2 Key words:
Chemical Constituents and Antioxidant Activity of Teucrium barbeyanum Aschers.
 Supporting Information Rec. Nat. Prod. 9:1 (2015) 159-163 Chemical Constituents and Antioxidant Activity of Teucrium barbeyanum Aschers. Mohamed Ali A. Alwahsh, Melati Khairuddean * and Wong Keng Chong
Supporting Information Rec. Nat. Prod. 9:1 (2015) 159-163 Chemical Constituents and Antioxidant Activity of Teucrium barbeyanum Aschers. Mohamed Ali A. Alwahsh, Melati Khairuddean * and Wong Keng Chong
Supporting Information
 Supporting Information Synthesis and Electroactive Properties of Poly(amidoamine) Dendrimers with an Aniline Pentamer Shell Wei-I Hung a, Chih-Bing Hung a, Ya-Han Chang a, Jiun-Kuang Dai a, Yan Li b, Hai
Supporting Information Synthesis and Electroactive Properties of Poly(amidoamine) Dendrimers with an Aniline Pentamer Shell Wei-I Hung a, Chih-Bing Hung a, Ya-Han Chang a, Jiun-Kuang Dai a, Yan Li b, Hai
Supplementary Materials: Development of Amyloseand β-cyclodextrin-based Chiral Fluorescent Sensors Bearing Terthienyl Pendants
 S1 of S12 Supplementary Materials: Development of Amyloseand β-cyclodextrin-based Chiral Fluorescent Sensors Bearing Terthienyl Pendants Tomoyuki Ikai, Changsik Yun, Yutaka Kojima, Daisuke Suzuki, Katsuhiro
S1 of S12 Supplementary Materials: Development of Amyloseand β-cyclodextrin-based Chiral Fluorescent Sensors Bearing Terthienyl Pendants Tomoyuki Ikai, Changsik Yun, Yutaka Kojima, Daisuke Suzuki, Katsuhiro
The Free Internet Journal for Organic Chemistry
 The Free Internet Journal for Organic Chemistry Paper Archive for Organic Chemistry Arkivoc 2018, part iii, S1-S6 Synthesis of dihydropyranones and dihydropyrano[2,3- d][1,3]dioxine-diones by cyclization
The Free Internet Journal for Organic Chemistry Paper Archive for Organic Chemistry Arkivoc 2018, part iii, S1-S6 Synthesis of dihydropyranones and dihydropyrano[2,3- d][1,3]dioxine-diones by cyclization
Highly enantioselective cascade synthesis of spiropyrazolones. Supporting Information. NMR spectra and HPLC traces
 Highly enantioselective cascade synthesis of spiropyrazolones Alex Zea a, Andrea-Nekane R. Alba a, Andrea Mazzanti b, Albert Moyano a and Ramon Rios a,c * Supporting Information NMR spectra and HPLC traces
Highly enantioselective cascade synthesis of spiropyrazolones Alex Zea a, Andrea-Nekane R. Alba a, Andrea Mazzanti b, Albert Moyano a and Ramon Rios a,c * Supporting Information NMR spectra and HPLC traces
Copper-Catalyzed Oxidative Dehydrogenative N-N Bond. Formation for the Synthesis of N,N -Diarylindazol-3-ones
 Electronic Supplementary Material (ESI) for Organic Chemistry Frontiers. This journal is the Partner Organisations 2016 Supporting information Copper-Catalyzed Oxidative Dehydrogenative - Bond Formation
Electronic Supplementary Material (ESI) for Organic Chemistry Frontiers. This journal is the Partner Organisations 2016 Supporting information Copper-Catalyzed Oxidative Dehydrogenative - Bond Formation
Supporting Information
 Supporting Information for AgOTf-catalyzed one-pot reactions of 2-alkynylbenzaldoximes with α,β-unsaturated carbonyl compounds Qiuping Ding 1, Dan Wang 1, Puying Luo* 2, Meiling Liu 1, Shouzhi Pu* 3 and
Supporting Information for AgOTf-catalyzed one-pot reactions of 2-alkynylbenzaldoximes with α,β-unsaturated carbonyl compounds Qiuping Ding 1, Dan Wang 1, Puying Luo* 2, Meiling Liu 1, Shouzhi Pu* 3 and
Supporting Information
 Supporting Information Mitochondria-Targeting Polydopamine Nanocomposites as Chemophotothermal Therapeutics for Cancer Zhuo Wang *,, Yuzhi Chen, Hui Zhang, Yawen Li, Yufan Ma, Jia Huang, Xiaolei Liu, Fang
Supporting Information Mitochondria-Targeting Polydopamine Nanocomposites as Chemophotothermal Therapeutics for Cancer Zhuo Wang *,, Yuzhi Chen, Hui Zhang, Yawen Li, Yufan Ma, Jia Huang, Xiaolei Liu, Fang
Electronic Supplementary Information
 Electronic Supplementary Information Unprecedented Carbon-Carbon Bond Cleavage in Nucleophilic Aziridine Ring Opening Reaction, Efficient Ring Transformation of Aziridines to Imidazolidin-4-ones Jin-Yuan
Electronic Supplementary Information Unprecedented Carbon-Carbon Bond Cleavage in Nucleophilic Aziridine Ring Opening Reaction, Efficient Ring Transformation of Aziridines to Imidazolidin-4-ones Jin-Yuan
Patrycja Miszczyk, Dorota Wieczorek, Joanna Gałęzowska, Błażej Dziuk, Joanna Wietrzyk and Ewa Chmielewska. 1. Spectroscopic Data.
 ; doi:10.3390/molecules22020254 S1 of S23 Supplementary Materials: Reaction of 3-Amino-1,2,4-Triazole with Diethyl Phosphite and Triethyl Orthoformate: Acid-Base Properties and Antiosteoporotic Activities
; doi:10.3390/molecules22020254 S1 of S23 Supplementary Materials: Reaction of 3-Amino-1,2,4-Triazole with Diethyl Phosphite and Triethyl Orthoformate: Acid-Base Properties and Antiosteoporotic Activities
Rudi Hendra 1, Paul A. Keller 1* Telephone: Fax:
 Supporting Information for Phytochemical Studies on Two Australian Anigozanthos Plant Species Rudi Hendra 1, Paul A. Keller 1* 1 School of Chemistry, University of Wollongong, NSW 2522, Australia Telephone:
Supporting Information for Phytochemical Studies on Two Australian Anigozanthos Plant Species Rudi Hendra 1, Paul A. Keller 1* 1 School of Chemistry, University of Wollongong, NSW 2522, Australia Telephone:
Pyrrolo[2,3-d:5,4-d']bisthiazoles: Alternate Synthetic Routes and a Comparative Study to Analogous Fused-ring Bithiophenes
![Pyrrolo[2,3-d:5,4-d']bisthiazoles: Alternate Synthetic Routes and a Comparative Study to Analogous Fused-ring Bithiophenes Pyrrolo[2,3-d:5,4-d']bisthiazoles: Alternate Synthetic Routes and a Comparative Study to Analogous Fused-ring Bithiophenes](/thumbs/92/108867714.jpg) SUPPORTING INFORMATION Pyrrolo[2,3-d:5,4-d']bisthiazoles: Alternate Synthetic Routes and a Comparative Study to Analogous Fused-ring Bithiophenes Eric J. Uzelac, Casey B. McCausland, and Seth C. Rasmussen*
SUPPORTING INFORMATION Pyrrolo[2,3-d:5,4-d']bisthiazoles: Alternate Synthetic Routes and a Comparative Study to Analogous Fused-ring Bithiophenes Eric J. Uzelac, Casey B. McCausland, and Seth C. Rasmussen*
Supporting information. Influence of Aerosol Acidity on the Chemical Composition of Secondary Organic Aerosol from β caryophyllene
 Supporting information Influence of Aerosol Acidity on the Chemical Composition of Secondary Organic Aerosol from β caryophyllene M. N. Chan 1, J. D. Surratt 2,*, A. W. H. Chan 2,**, K. Schilling 2, J.
Supporting information Influence of Aerosol Acidity on the Chemical Composition of Secondary Organic Aerosol from β caryophyllene M. N. Chan 1, J. D. Surratt 2,*, A. W. H. Chan 2,**, K. Schilling 2, J.
9-amino-(9-deoxy)cinchona alkaloids-derived novel chiral phase-transfer catalysts
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 9-amino-(9-deoxy)cinchona alkaloids-derived novel chiral phase-transfer
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 9-amino-(9-deoxy)cinchona alkaloids-derived novel chiral phase-transfer
National d Histoire Naturelle, 57 rue Cuvier (C.P. 54), Paris, France. Contents:
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2017 Supporting Information Synthesis and Biological Evaluation of the Ascidian
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2017 Supporting Information Synthesis and Biological Evaluation of the Ascidian
Phenylpropanoids, Sesquiterpenoids and Flavonoids from Pimpinella tragium Vill. subsp. lithophila (Schischkin) Tutin
 Supporting Information Rec. Nat. Prod. 10:2 (2016) 207-213 Phenylpropanoids, Sesquiterpenoids and Flavonoids from Pimpinella tragium Vill. subsp. lithophila (Schischkin) Tutin Hilal Özbek 1, Zühal Güvenalp
Supporting Information Rec. Nat. Prod. 10:2 (2016) 207-213 Phenylpropanoids, Sesquiterpenoids and Flavonoids from Pimpinella tragium Vill. subsp. lithophila (Schischkin) Tutin Hilal Özbek 1, Zühal Güvenalp
Supporting Information
 Supporting Information Aluminum Complexes of N 2 O 2 3 Formazanate Ligands Supported by Phosphine Oxide Donors Ryan R. Maar, Amir Rabiee Kenaree, Ruizhong Zhang, Yichen Tao, Benjamin D. Katzman, Viktor
Supporting Information Aluminum Complexes of N 2 O 2 3 Formazanate Ligands Supported by Phosphine Oxide Donors Ryan R. Maar, Amir Rabiee Kenaree, Ruizhong Zhang, Yichen Tao, Benjamin D. Katzman, Viktor
Selecting Critical Properties of Terpenes and Terpenoids through Group-Contribution Methods and Equations of State
 Supporting Information Selecting Critical Properties of Terpenes and Terpenoids through Group-Contribution Methods and Equations of State Mónia A. R. Martins, 1,2 Pedro J. Carvalho, 1 André M. Palma, 1
Supporting Information Selecting Critical Properties of Terpenes and Terpenoids through Group-Contribution Methods and Equations of State Mónia A. R. Martins, 1,2 Pedro J. Carvalho, 1 André M. Palma, 1
Heterobimetallic Pd-Sn Catalysis: Michael Addition. Reaction with C-, N-, O-, S- Nucleophiles and In-situ. Diagnostics
 Supporting Information (SI) Heterobimetallic Pd-Sn Catalysis: Michael Addition Reaction with C-, N-, -, S- Nucleophiles and In-situ Diagnostics Debjit Das, a Sanjay Pratihar a,b and Sujit Roy c * a rganometallics
Supporting Information (SI) Heterobimetallic Pd-Sn Catalysis: Michael Addition Reaction with C-, N-, -, S- Nucleophiles and In-situ Diagnostics Debjit Das, a Sanjay Pratihar a,b and Sujit Roy c * a rganometallics
Supporting information
 Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 204 Supporting information Palladium nanoparticles supported on triazine functionalised mesoporous
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 204 Supporting information Palladium nanoparticles supported on triazine functionalised mesoporous
ATP. adenosine triphosphate (ATP) Nontuberculous Mycobacteria (NTM) ATP
 164 ATP 1) 1) 1) 2) 2) 1) 1) 2) 16 4 8 16 9 7 adenosine triphosphate (ATP) Nontuberculous Mycobacteria (NTM) ATP MB ATCC 4 45 ATP 30 60 10 ATP 5 Relative light units (RLU) ratio 7 MB streptomycin, ethambutol,
164 ATP 1) 1) 1) 2) 2) 1) 1) 2) 16 4 8 16 9 7 adenosine triphosphate (ATP) Nontuberculous Mycobacteria (NTM) ATP MB ATCC 4 45 ATP 30 60 10 ATP 5 Relative light units (RLU) ratio 7 MB streptomycin, ethambutol,
Electrolyzed-Reduced Water as Artificial Hot Spring Water
 /-,**- + +/ 0 +, - + + +, - + +. ++,3 +/. +. Electrolyzed-Reduced Water as Artificial Hot Spring Water Shoichi OKOUCHI +, Daisuke TAKEZAKI +, Hideyuki OHNAMI +, Yuhkoh AGISHI,, Yasuo KANROJI -, and Shigeo
/-,**- + +/ 0 +, - + + +, - + +. ++,3 +/. +. Electrolyzed-Reduced Water as Artificial Hot Spring Water Shoichi OKOUCHI +, Daisuke TAKEZAKI +, Hideyuki OHNAMI +, Yuhkoh AGISHI,, Yasuo KANROJI -, and Shigeo
Supplementary Materials for. Kinetic and Computational Studies on Pd(I) Dimer- Mediated Halogen Exchange of Aryl Iodides
 Supplementary Materials for Kinetic and Computational Studies on Pd(I) Dimer- Mediated Halogen Exchange of Aryl Iodides Indrek Kalvet, a Karl J. Bonney, a and Franziska Schoenebeck a * a Institute of Organic
Supplementary Materials for Kinetic and Computational Studies on Pd(I) Dimer- Mediated Halogen Exchange of Aryl Iodides Indrek Kalvet, a Karl J. Bonney, a and Franziska Schoenebeck a * a Institute of Organic
Electronic Supplementary Information. Carbon dioxide as a reversible amine-protecting
 Electronic Supplementary Information Carbon dioxide as a reversible amine-protecting agent in selective Michael additions and acylations Annelies Peeters, Rob Ameloot and Dirk E. De Vos * Centre for Surface
Electronic Supplementary Information Carbon dioxide as a reversible amine-protecting agent in selective Michael additions and acylations Annelies Peeters, Rob Ameloot and Dirk E. De Vos * Centre for Surface
Etest. cefepime. oxacillin Staphylococcus aureus oxacillin. imipenem piperacillin ceftazidime cefpirome
 Etest 3 14 6 6 14 7 31 2000 cefepime 1997 1998 22 22 43 1 Etest 10 10 oxacillin Staphylococcus aureus oxacillin staphylococciceftazidimeescherichia coli imipenem piperacillinceftazidimecefpirome cefoperazonesulbactam
Etest 3 14 6 6 14 7 31 2000 cefepime 1997 1998 22 22 43 1 Etest 10 10 oxacillin Staphylococcus aureus oxacillin staphylococciceftazidimeescherichia coli imipenem piperacillinceftazidimecefpirome cefoperazonesulbactam
Table of Contents 1 Supplementary Data MCD
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2017 Supporting Information for Magnetic circular dichroism and density functional theory
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2017 Supporting Information for Magnetic circular dichroism and density functional theory
Supporting Information. A catalyst-free multicomponent domino sequence for the. diastereoselective synthesis of (E)-3-[2-arylcarbonyl-3-
 Supporting Information for A catalyst-free multicomponent domino sequence for the diastereoselective synthesis of (E)-3-[2-arylcarbonyl-3- (arylamino)allyl]chromen-4-ones Pitchaimani Prasanna 1, Pethaiah
Supporting Information for A catalyst-free multicomponent domino sequence for the diastereoselective synthesis of (E)-3-[2-arylcarbonyl-3- (arylamino)allyl]chromen-4-ones Pitchaimani Prasanna 1, Pethaiah
Supporting Information
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Supporting Information Abnormal N-Heterocyclic Carbene Based Nickel Complex for Catalytic
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Supporting Information Abnormal N-Heterocyclic Carbene Based Nickel Complex for Catalytic
Rh(III)-Catalyzed C-H Amidation with N-hydroxycarbamates: A. new Entry to N-Carbamate Protected Arylamines
 Rh(III)-Catalyzed C-H Amidation with N-hydroxycarbamates: A new Entry to N-Carbamate Protected Arylamines Bing Zhou,* Juanjuan Du, Yaxi Yang,* Huijin Feng, Yuanchao Li Shanghai Institute of Materia Medica,
Rh(III)-Catalyzed C-H Amidation with N-hydroxycarbamates: A new Entry to N-Carbamate Protected Arylamines Bing Zhou,* Juanjuan Du, Yaxi Yang,* Huijin Feng, Yuanchao Li Shanghai Institute of Materia Medica,
Supplementary Information
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2014 Supplementary Information A contribution to the rational design of Ru(CO) 3 Cl 2 L complexes
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2014 Supplementary Information A contribution to the rational design of Ru(CO) 3 Cl 2 L complexes
Supporting Information
 Supporting Information Montmorillonite KSF-Catalyzed One-pot, Three-component, Aza-Diels- Alder Reactions of Methylenecyclopropanes With Arylaldehydes and Aromatic Amines Li-Xiong Shao and Min Shi* General
Supporting Information Montmorillonite KSF-Catalyzed One-pot, Three-component, Aza-Diels- Alder Reactions of Methylenecyclopropanes With Arylaldehydes and Aromatic Amines Li-Xiong Shao and Min Shi* General
Electronic Supplementary Information (ESI)
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information (ESI) Cyclopentadienyl iron dicarbonyl (CpFe(CO) 2 ) derivatives
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information (ESI) Cyclopentadienyl iron dicarbonyl (CpFe(CO) 2 ) derivatives
Supporting Information. Introduction of a α,β-unsaturated carbonyl conjugated pyrene-lactose hybrid
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 16 Supporting Information Introduction of a α,β-unsaturated carbonyl conjugated pyrene-lactose hybrid
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 16 Supporting Information Introduction of a α,β-unsaturated carbonyl conjugated pyrene-lactose hybrid
Electronic Supplementary Information
 Electronic Supplementary Information Cucurbit[7]uril host-guest complexes of the histamine H 2 -receptor antagonist ranitidine Ruibing Wang and Donal H. Macartney* Department of Chemistry, Queen s University,
Electronic Supplementary Information Cucurbit[7]uril host-guest complexes of the histamine H 2 -receptor antagonist ranitidine Ruibing Wang and Donal H. Macartney* Department of Chemistry, Queen s University,
Available online at shd.org.rs/jscs/
 J. Serb. Chem. Soc. 78 (1) S1 S8 (2013) Supplementary material SUPPLEMENTARY MATERIAL TO Metal complexes of N'-[2-hydroxy-5-(phenyldiazenyl)- benzylidene]isonicotinohydrazide. Synthesis, spectroscopic
J. Serb. Chem. Soc. 78 (1) S1 S8 (2013) Supplementary material SUPPLEMENTARY MATERIAL TO Metal complexes of N'-[2-hydroxy-5-(phenyldiazenyl)- benzylidene]isonicotinohydrazide. Synthesis, spectroscopic
Aluminium triflate as a Lewis acid catalyst for the ring opening of epoxides in alcohols
 Aluminium triflate as a Lewis acid catalyst for the ring opening of epoxides in alcohols D. Bradley G. Williams* and Michelle Lawton a Department of Chemistry, University of Johannesburg, P.O. Box 524,
Aluminium triflate as a Lewis acid catalyst for the ring opening of epoxides in alcohols D. Bradley G. Williams* and Michelle Lawton a Department of Chemistry, University of Johannesburg, P.O. Box 524,
Iodine-catalyzed synthesis of sulfur-bridged enaminones and chromones via double C(sp 2 )-H thiolation
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2017 Iodine-catalyzed synthesis of sulfur-bridged enaminones and chromones via
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2017 Iodine-catalyzed synthesis of sulfur-bridged enaminones and chromones via
Nguyen Hien Trang* **
 152 Nippon Shokuhin Kagaku Kogaku Kaishi Vol.., No.., +,+3 (,**1) 10 Nguyen Hien Trang* ** * Department of Food Science and Technology, Hue University of Agriculture and Forestry ** Properties of "Shishibishio
152 Nippon Shokuhin Kagaku Kogaku Kaishi Vol.., No.., +,+3 (,**1) 10 Nguyen Hien Trang* ** * Department of Food Science and Technology, Hue University of Agriculture and Forestry ** Properties of "Shishibishio
Site-Selective Suzuki-Miyaura Cross-Coupling Reactions of 2,3,4,5-Tetrabromofuran
 1 Site-Selective Suzuki-Miyaura Cross-Coupling Reactions of 2,3,4,5-Tetrabromofuran Munawar Hussain, a Rasheed Ahmad Khera, a Nguyen Thai Hung, a Peter Langer* a,b a Institut für Chemie, Universität Rostock,
1 Site-Selective Suzuki-Miyaura Cross-Coupling Reactions of 2,3,4,5-Tetrabromofuran Munawar Hussain, a Rasheed Ahmad Khera, a Nguyen Thai Hung, a Peter Langer* a,b a Institut für Chemie, Universität Rostock,
Supporting Information
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2015 Supporting Information New PKS-NRPS tetramic acids and pyridinone from
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2015 Supporting Information New PKS-NRPS tetramic acids and pyridinone from
Supplementary information:
 Supplementary information: Monitoring changes of docosahexaenoic acid-containing lipids during the recovery process of traumatic brain injury in rat using mass spectrometry imaging Shuai Guo 1, Dan Zhou
Supplementary information: Monitoring changes of docosahexaenoic acid-containing lipids during the recovery process of traumatic brain injury in rat using mass spectrometry imaging Shuai Guo 1, Dan Zhou
Divergent synthesis of various iminocyclitols from D-ribose
 Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 205 Divergent synthesis of various iminocyclitols from D-ribose Ramu Petakamsetty,
Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 205 Divergent synthesis of various iminocyclitols from D-ribose Ramu Petakamsetty,
difluoroboranyls derived from amides carrying donor group Supporting Information
 The influence of the π-conjugated spacer on photophysical properties of difluoroboranyls derived from amides carrying donor group Supporting Information Anna Maria Grabarz a Adèle D. Laurent b, Beata Jędrzejewska
The influence of the π-conjugated spacer on photophysical properties of difluoroboranyls derived from amides carrying donor group Supporting Information Anna Maria Grabarz a Adèle D. Laurent b, Beata Jędrzejewska
Available online at shd.org.rs/jscs/
 J. Serb. Chem. Soc. 78 (12) S131 S136 (2013) Supplementary material SUPPLEMENTARY MATERIAL TO New 9-aminoacridine derivatives as inhibitors of botulinum neurotoxins and P. falciparum malaria MIKLOŠ TOT
J. Serb. Chem. Soc. 78 (12) S131 S136 (2013) Supplementary material SUPPLEMENTARY MATERIAL TO New 9-aminoacridine derivatives as inhibitors of botulinum neurotoxins and P. falciparum malaria MIKLOŠ TOT
Supporting information
 Supporting information Synthesis and biological investigation of oxazole hydroxamates as highly selective histone deacetylase 6 (HDAC6) inhibitors Johanna Senger, Jelena Melesina, Martin Marek, Christophe
Supporting information Synthesis and biological investigation of oxazole hydroxamates as highly selective histone deacetylase 6 (HDAC6) inhibitors Johanna Senger, Jelena Melesina, Martin Marek, Christophe
Nickel and Platinum PCP Pincer Complexes Incorporating an Acyclic Diaminoalkyl Central Moiety Connecting Imidazole or Pyrazole Rings
 ickel and Platinum PCP Pincer Complexes Incorporating an Acyclic Diaminoalkyl Central Moiety Connecting Imidazole or Pyrazole Rings Braulio M. Puerta Lombardi, Rudy M. Braun, Chris Gendy, Chia Yun Chang,
ickel and Platinum PCP Pincer Complexes Incorporating an Acyclic Diaminoalkyl Central Moiety Connecting Imidazole or Pyrazole Rings Braulio M. Puerta Lombardi, Rudy M. Braun, Chris Gendy, Chia Yun Chang,
Electronic Supplementary Information
 Electronic Supplementary Information The preferred all-gauche conformations in 3-fluoro-1,2-propanediol Laize A. F. Andrade, a Josué M. Silla, a Claudimar J. Duarte, b Roberto Rittner, b Matheus P. Freitas*,a
Electronic Supplementary Information The preferred all-gauche conformations in 3-fluoro-1,2-propanediol Laize A. F. Andrade, a Josué M. Silla, a Claudimar J. Duarte, b Roberto Rittner, b Matheus P. Freitas*,a
The effect of curcumin on the stability of Aβ. dimers
 The effect of curcumin on the stability of Aβ dimers Li Na Zhao, See-Wing Chiu, Jérôme Benoit, Lock Yue Chew,, and Yuguang Mu, School of Physical and Mathematical Sciences, Nanyang Technological University,
The effect of curcumin on the stability of Aβ dimers Li Na Zhao, See-Wing Chiu, Jérôme Benoit, Lock Yue Chew,, and Yuguang Mu, School of Physical and Mathematical Sciences, Nanyang Technological University,
VBA Microsoft Excel. J. Comput. Chem. Jpn., Vol. 5, No. 1, pp (2006)
 J. Comput. Chem. Jpn., Vol. 5, No. 1, pp. 29 38 (2006) Microsoft Excel, 184-8588 2-24-16 e-mail: yosimura@cc.tuat.ac.jp (Received: July 28, 2005; Accepted for publication: October 24, 2005; Published on
J. Comput. Chem. Jpn., Vol. 5, No. 1, pp. 29 38 (2006) Microsoft Excel, 184-8588 2-24-16 e-mail: yosimura@cc.tuat.ac.jp (Received: July 28, 2005; Accepted for publication: October 24, 2005; Published on
Protease-catalysed Direct Asymmetric Mannich Reaction in Organic Solvent
 Supplementary information for the paper Protease-catalysed Direct Asymmetric Mannich Reaction in Organic Solvent Yang Xue, Ling-Po Li, Yan-Hong He * & Zhi Guan * School of Chemistry and Chemical Engineering,
Supplementary information for the paper Protease-catalysed Direct Asymmetric Mannich Reaction in Organic Solvent Yang Xue, Ling-Po Li, Yan-Hong He * & Zhi Guan * School of Chemistry and Chemical Engineering,
Available online at
 J. Serb. Chem. Soc. 76 (12) S1 S5 (2011) Supplementary material SUPPLEMENTARY MATERIAL TO Synthesis of quinoline-attached furan-2(3h)-ones having anti-inflammatory and antibacterial properties with reduced
J. Serb. Chem. Soc. 76 (12) S1 S5 (2011) Supplementary material SUPPLEMENTARY MATERIAL TO Synthesis of quinoline-attached furan-2(3h)-ones having anti-inflammatory and antibacterial properties with reduced
Synthesis of Imines from Amines in Aliphatic Alcohols on Pd/ZrO 2 Catalyst at Ambient Conditions
 This journal is The Royal Society of Chemistry 213 Synthesis of Imines from Amines in Aliphatic Alcohols on Pd/ZrO 2 Catalyst at Ambient Conditions Wenjing Cui, a Bao Zhaorigetu,* a Meilin Jia, a and Wulan
This journal is The Royal Society of Chemistry 213 Synthesis of Imines from Amines in Aliphatic Alcohols on Pd/ZrO 2 Catalyst at Ambient Conditions Wenjing Cui, a Bao Zhaorigetu,* a Meilin Jia, a and Wulan
Supporting Information. Route to benzo- and pyrido-fused 1,2,4-triazinyl radicals via N'-(het)aryl- N'-[2-nitro(het)aryl]hydrazides
![Supporting Information. Route to benzo- and pyrido-fused 1,2,4-triazinyl radicals via N'-(het)aryl- N'-[2-nitro(het)aryl]hydrazides Supporting Information. Route to benzo- and pyrido-fused 1,2,4-triazinyl radicals via N'-(het)aryl- N'-[2-nitro(het)aryl]hydrazides](/thumbs/93/112488784.jpg) Supporting Information Route to benzo- and pyrido-fused 1,2,4-triazinyl radicals via N'-(het)aryl- N'-[2-nitro(het)aryl]hydrazides Andrey A. Berezin, Georgia Zissimou, Christos P. Constantinides, Yassine
Supporting Information Route to benzo- and pyrido-fused 1,2,4-triazinyl radicals via N'-(het)aryl- N'-[2-nitro(het)aryl]hydrazides Andrey A. Berezin, Georgia Zissimou, Christos P. Constantinides, Yassine
Supporting Information
 Supporting Information Biomimetic total syntheses of borreverine and flinderole alkaloids. Dattatraya. Dethe,* Rohan D. Erande and Alok Ranjan Department of Chemistry, Indian Institute of Technology-Kanpur,
Supporting Information Biomimetic total syntheses of borreverine and flinderole alkaloids. Dattatraya. Dethe,* Rohan D. Erande and Alok Ranjan Department of Chemistry, Indian Institute of Technology-Kanpur,
Free Radical Initiated Coupling Reaction of Alcohols and. Alkynes: not C-O but C-C Bond Formation. Context. General information 2. Typical procedure 2
 Free Radical Initiated Coupling Reaction of Alcohols and Alkynes: not C-O but C-C Bond Formation Zhongquan Liu,* Liang Sun, Jianguo Wang, Jie Han, Yankai Zhao, Bo Zhou Institute of Organic Chemistry, Gannan
Free Radical Initiated Coupling Reaction of Alcohols and Alkynes: not C-O but C-C Bond Formation Zhongquan Liu,* Liang Sun, Jianguo Wang, Jie Han, Yankai Zhao, Bo Zhou Institute of Organic Chemistry, Gannan
Supporting Information
 Supporting Information for Lewis acid-catalyzed redox-neutral amination of 2-(3-pyrroline-1-yl)benzaldehydes via intramolecular [1,5]-hydride shift/isomerization reaction Chun-Huan Jiang, Xiantao Lei,
Supporting Information for Lewis acid-catalyzed redox-neutral amination of 2-(3-pyrroline-1-yl)benzaldehydes via intramolecular [1,5]-hydride shift/isomerization reaction Chun-Huan Jiang, Xiantao Lei,
of the methanol-dimethylamine complex
 Electronic Supplementary Information for: Fundamental and overtone virational spectroscopy, enthalpy of hydrogen ond formation and equilirium constant determination of the methanol-dimethylamine complex
Electronic Supplementary Information for: Fundamental and overtone virational spectroscopy, enthalpy of hydrogen ond formation and equilirium constant determination of the methanol-dimethylamine complex
Supplementary Information
 9.4 6.60. 8.6 8.8 9.29 20.04 29.6 0.07 6. 40.92 4.64 06.0 6.29 6.0-0.00 Supplementary Information Conformational Analysis, Experimental and GIA-DFT C NMR Chemical Shift Calculation on 2 -Hydroxy-,4,-trimethoxy-chalcone
9.4 6.60. 8.6 8.8 9.29 20.04 29.6 0.07 6. 40.92 4.64 06.0 6.29 6.0-0.00 Supplementary Information Conformational Analysis, Experimental and GIA-DFT C NMR Chemical Shift Calculation on 2 -Hydroxy-,4,-trimethoxy-chalcone
Direct Palladium-Catalyzed Arylations of Aryl Bromides. with 2/9-Substituted Pyrimido[5,4-b]indolizines
![Direct Palladium-Catalyzed Arylations of Aryl Bromides. with 2/9-Substituted Pyrimido[5,4-b]indolizines Direct Palladium-Catalyzed Arylations of Aryl Bromides. with 2/9-Substituted Pyrimido[5,4-b]indolizines](/thumbs/87/95187440.jpg) Direct Palladium-Catalyzed Arylations of Aryl Bromides with 2/9-Substituted Pyrimido[5,4-b]indolizines Min Jiang, Ting Li, Linghua Meng, Chunhao Yang,* Yuyuan Xie*, and Jian Ding State Key Laboratory of
Direct Palladium-Catalyzed Arylations of Aryl Bromides with 2/9-Substituted Pyrimido[5,4-b]indolizines Min Jiang, Ting Li, Linghua Meng, Chunhao Yang,* Yuyuan Xie*, and Jian Ding State Key Laboratory of
SUPPORTING INFORMATION
 SUPPRTING INFRMATIN Dihydroxylation of lefins Catalyzed by Zeolite-Confined smium Nanoclusters: A Novel, Effective, Reusable and Eco-Friendly Method for Preparation of 1,2-cis-Diols Önder Metin,*,a Nurdan
SUPPRTING INFRMATIN Dihydroxylation of lefins Catalyzed by Zeolite-Confined smium Nanoclusters: A Novel, Effective, Reusable and Eco-Friendly Method for Preparation of 1,2-cis-Diols Önder Metin,*,a Nurdan
Metal-free Oxidative Coupling of Amines with Sodium Sulfinates: A Mild Access to Sulfonamides
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting information for Metal-free Oxidative Coupling of Amines with Sodium Sulfinates:
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting information for Metal-free Oxidative Coupling of Amines with Sodium Sulfinates:
Synthesis of α-deoxymono and -Difluorohexopyranosyl. 1-Phosphates and Kinetic Evaluation with Thymidylyl- and. Guanidylyltransferases
 Synthesis of α-deoxymono and -Difluorohexopyranosyl 1-Phosphates and Kinetic Evaluation with Thymidylyl- and Guanidylyltransferases Jian-She Zhu, a Nicole E. McCormick, a Shannon C. Timmons, b,c David
Synthesis of α-deoxymono and -Difluorohexopyranosyl 1-Phosphates and Kinetic Evaluation with Thymidylyl- and Guanidylyltransferases Jian-She Zhu, a Nicole E. McCormick, a Shannon C. Timmons, b,c David
Supporting Information
 Supporting Information Lewis acid catalyzed ring-opening reactions of methylenecyclopropanes with diphenylphosphine oxide in the presence of sulfur or selenium Min Shi,* Min Jiang and Le-Ping Liu State
Supporting Information Lewis acid catalyzed ring-opening reactions of methylenecyclopropanes with diphenylphosphine oxide in the presence of sulfur or selenium Min Shi,* Min Jiang and Le-Ping Liu State
