Comparison of carbon-sulfur and carbon-amine bond in therapeutic drug: -S-aromatic heterocyclic podophyllum derivatives display antitumor activity
|
|
|
- Ἀριστόβουλος Γιαννόπουλος
- 5 χρόνια πριν
- Προβολές:
Transcript
1 Comparison of carbon-sulfur and carbon-amine bond in therapeutic drug: -S-aromatic heterocyclic podophyllum derivatives display antitumor activity Jian-Long Li 1,a, Wei Zhao 1,a, Chen Zhou 1,a, Ya-Xuan Zhang a, Hong-Mei Li a, Ya-Ling Tang b, Xin-Hua Liang b, Tao Chen c, and Ya-Jie Tang a *, a Key Laboratory of Fermentation Engineering (Ministry of Education), Hubei Provincial Cooperative Innovation Center of Industrial Fermentation, Hubei University of Technology, Wuhan , China b State Key Laboratory of Oral Diseases West China Hospital of Stomatology (Sichuan University), Chengdu Sichuan ,People s Republic of China c Key Laboratory of Systems Bioengineering (Ministry of Education), School of Chemical Engineering and Technology, Tianjin University, Tianjin , China *Corresponding author. Tel. & Fax: yajietang@hotmail.com 1 Equally contributed to this work
2 Supporting Information Table S1. Calculated docking of the complex of podophyllum derivatives with tubulin/topo II. Energy Energy Compound H-bond packing Compound ( -kcal/mol) ( -kcal/mol) H-bond packing 1S Ser A178 1'S 6.51 Asp B479 Arg B503, DG D13 1N 4.03 Asn B258 Lys B352 1'N 3.04 Asp B479 Arg B503, DT F9, DA D12, DG D13 2S 4.99 Lys B254 Leu B248 2'S Asp B479 Arg B503, DT F9, DG D13 2N 5.72 Lys B352 Leu B255, Lys B352 2'N 6.15 Asp B479 Arg B503, DA D12, DG D13 3S 4.37 Ser A178 Ala B316 3'S 3.86 Asp B479 Arg B503, DT F9, DG D13 3N 3.87 Asn B258, Thr B353 Leu B248 3'N 5.58 Asp B479 Arg B503, DT F9, DG D13 4S 2.90 Thr A179 Lys B254 4'S 4.52 Asp B479 Arg B503, DT F9, DG D13 4N 2.45 Asn B258 Lys B352 4'N 2.69 Asp B479 Arg B503, DT F9, DG D13 5S 6.45 Lys B254 Lys B254 5'S 5.10 Asp B479 Arg B503, DT F9, DG D13 5N Leu B248 5'N 4.12 Asp B479 Arg B503, DA D12, DG D13 6S 6.28 Lys B352-6'S 8.42 Asp B479 Arg B503, DT F9, DG D13 6N 3.14 Leu B255-6'N 8.10 Asp B479, DG D13 Arg B503, DA D12, DG D13 7S 'S N Thr B353-7'N DC E8 DT F9, DA D12 8S 'S Lys B456 Arg B503 8N Leu B248 8'N Arg B503, DT F9 9S 'S Lys B456 Arg B503 9N Leu B255-9'N Arg B503 PTOX 4.11 Leu B248 Ala B316 VP Asp B479 Arg B503, DC E8, DG D13 2
3 3
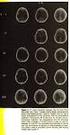
4 Figure S1. Effects of podophyllum derivatives on the HeLa cell cycle arrest and apoptosis induce. (A) Compound 4β-S-(1, 3, 4-trizole-2)-4-deoxy-podophyllotoxin (Compound 1S), Compound 4β-NH-(1, 3, 4-trizole-2)-4-deoxy-podophyllotoxin (Compound 1N), 4β-S-(1, 3, 4-trizole-2)-4-deoxy-4'-demethylepipodophyllotoxin (Compound 1'S) and Compound 4β-NH-(1, 3, 4-trizole-2)-4-deoxy-4'-demethylepipodophyllotoxin (Compound 1'N) arrested cell cycle in HeLa cells in a dose- and time-dependent manner at the concentration of 0, 0.1, 1, and 5 µm for 6, 12, 24, and 48 h, respectively. Compared with the cells incubated with no drug, the treatment of Compounds 1S and 1N did not induce the G 2 /M phase arrest at a lower concentration of 0.1 μm. However, the percentage of G 2 /M phase cells was accumulated significantly to about 80% at 48 h when the concentration was increased to 1 μm. And then the percentage of G 2 /M phase cells continuously increased to the maximum about 80-90% at the higher concentration of 5 μm after the incubation of 48 h. Corresponding to Compounds 1S and 1N, the comparison between Compound 1'S and 1'N showed the similar trend that Compound 1'S was superior than Compound 1'N to arrest the cell cycle. (B) Compound 1S, 1N, 1'S, and 1'N induced cell apoptosis in HeLa cells in a dose- and time-dependent manner at the concentration of 0, 0.1, 1, and 5 µm for 6, 12, 24, and 48 h, respectively. Symbols: the negative control without adding Compounds 1S, 1N, 1'S and 1'N (0 µm), Compound 1S (black triangle, ), Compound 1N (open Compound 1'N (open circle, ). triangle, ), Compound 1'S (black circle, ), and 4
5 Figure S2 Figure S3 5
6 1 Calculated docking of complexes 2 6
7 3 7
8 4 8
9 5 9
10 6 10
11 7 11
12 Copies of 1 H, 13 C NMR and 2D NMR (1H-1H COSY, HMBC, HSQC) Spectra 1. NMR and MS spectrum of compound 3S H NMR spectrum of compound 3S C NMR spectrum of compound 3S H- 1 H COSY spectrums for compound 3S HMBC spectrums for compound 3S HSQC spectrums for compound 3S MS diagram for compound 3S NMR spectrum of compound 4S H NMR spectrum of compound 4S C NMR spectrum of compound 4S H- 1 H COSY spectrums for compound 4S HMBC spectrums for compound 4S HSQC spectrums for compound 4S MS diagram for compound 4S NMR and MS spectrum of compound 5S H NMR spectrum of compound 5S C NMR spectrum of compound 5S H- 1 H COSY spectrums for compound 5S HMBC spectrums for compound 5S HSQC spectrums for compound 5S MS diagram for compound 5S NMR and MS spectrum of compound 6S. 12
13 H NMR spectrum of compound 6S C NMR spectrum of compound 6S H- 1 H COSY spectrums for compound 6S HMBC spectrums for compound 6S HSQC spectrums for compound 6S MS diagram for compound 6S NMR and MS spectrum of compound 3'S H NMR spectrum of compound 3'S C NMR spectrum of compound 3'S H- 1 H COSY spectrums for compound 3'S HMBC spectrums for compound 3'S HSQC spectrums for compound 3'S MS diagram for compound 3'S NMR and MS spectrum of compound 4'S H NMR spectrum of compound 4'S C NMR spectrum of compound 4'S H- 1 H COSY spectrums for compound 4'S HMBC spectrums for compound 4'S HSQC spectrums for compound 4'S MS diagram for compound 4'S NMR and MS spectrum of compound 5'S H NMR spectrum of compound 5'S C NMR spectrum of compound 5'S. 13
14 H- 1 H COSY spectrums for compound 5'S HMBC spectrums for compound 5'S HSQC spectrums for compound 5'S MS diagram for compound 5'S NMR spectrum of compound 6'S H NMR spectrum of compound 6'S C NMR spectrum of compound 6'S H- 1 H COSY spectrums for compound 6'S HMBC spectrums for compound 6'S HSQC spectrums for compound 6'S MS diagram for compound 6'S NMR and MS spectrum of compound 1N H NMR spectrum of compound 1N C NMR spectrum of compound 1N H- 1 H COSY spectrums for compound 1N HMBC spectrums for compound 1N HSQC spectrums for compound 1N MS diagram for compound 1N NMR spectrum of compound 2N H NMR spectrum of compound 2N C NMR spectrum of compound 2N H- 1 H COSY spectrums for compound 2N HMBC spectrums for compound 2N. 14
15 HSQC spectrums for compound 2N MS diagram for compound 2N NMR spectrum of compound 3N H NMR spectrum of compound 3N C NMR spectrum of compound 3N H- 1 H COSY spectrums for compound 3N HMBC spectrums for compound 3N HSQC spectrums for compound 3N MS diagram for compound 3N NMR spectrum of compound 4N H NMR spectrum of compound 4N C NMR spectrum of compound 4N COSY spectrums for compound 4N HMBC spectrums for compound 4N HSQC spectrums for compound 4N MS diagram for compound 4N NMR and MS spectrum of compound 5N H NMR spectrum of compound 5N C NMR spectrum of compound 5N COSY spectrums for compound 5N HMBC spectrums for compound 5N HSQC spectrums for compound 5N MS diagram for compound 5N. 15
16 NMR and MS spectrum of compound 6N H NMR spectrum of compound 6N C NMR spectrum of compound 6N COSY spectrums for compound 6N HMBC spectrums for compound 6N HSQC spectrums for compound 6N MS diagram for compound 6N NMR and MS spectrum of compound 1'N H NMR spectrum of compound 1'N C NMR spectrum of compound 1'N H- 1 H COSY spectrums for compound 1'N HMBC spectrums for compound 1'N HSQC spectrums for compound 1'N MS diagram for compound 1'N NMR spectrum of compound 2'N H NMR spectrum of compound 2'N C NMR spectrum of compound 2'N H- 1 H COSY spectrums for compound 2'N HMBC spectrums for compound 2'N HSQC spectrums for compound 2'N MS diagram for compound 2'N NMR spectrum of compound 3'N H NMR spectrum of compound 3'N. 16
17 C NMR spectrum of compound 3'N H- 1 H COSY spectrums for compound 3'N HMBC spectrums for compound 3'N HSQC spectrums for compound 3'N MS diagram for compound 3'N NMR spectrum of compound 4'N H NMR spectrum of compound 4'N C NMR spectrum of compound 4'N COSY spectrums for compound 4'N HMBC spectrums for compound 4'N HSQC spectrums for compound 4'N MS diagram for compound 4'N NMR and MS spectrum of compound 5'N H NMR spectrum of compound 5'N C NMR spectrum of compound 5'N COSY spectrums for compound 5'N HMBC spectrums for compound 5'N HSQC spectrums for compound 5'N MS diagram for compound 5'N NMR and MS spectrum of compound 6'N H NMR spectrum of compound 6'N C NMR spectrum of compound 6'N COSY spectrums for compound 6'N. 17
18 HMBC spectrums for compound 6'N HSQC spectrums for compound 6'N MS diagram for compound 6'N
19 Copies of 1 H, 13 C NMR and 2D NMR ( 1 H- 1 H COSY, HMBC, HSQC) Spectra H NMR spectrum of compound 3S C NMR spectrum of compound 3S
20 H- 1 H COSY diagram of compound 3S HMBC diagram of compound 3S
21 HSQC diagram of compound 3S MS spectrum of compound 3S β-S-(pyridine-2)-4-deoxy-podophyllotoxin (3S). 1 H NMR (400 MHz, CDCl 3, δ): 8.40 (d, J = 4.0 Hz, 1H), 7.52 (t, J = 4.0 Hz, 1H), 7.18 (d, J = 8.0 Hz, 1H), 7.05 (t, J = 4.0 Hz, 1H), 6.95 (s, 1H), 6.46 (s, 1H), 6.33 (s, 2H), 5.95 (d, J = 8.0 Hz, 2H), 5.57 (d, J = 4.0 Hz, 1H), 4.60 (d, J = 4.0 Hz, 1H), 4.34 (t, J = 8.0 Hz, 1H), 3.86 (t, J = 8.0 Hz, 1H), 3.80 (s, 3H), 3.76 (s, 6H), (m, 2H); 13 C NMR (101 MHz, CDCl 3, δ): , , (2C), , , , , , , , , , , , , (2C), , 71.01, 60.76, (2C), 45.63, 43.77, 42.50, ESI-MS: calc d for C 27 H 25 NO 7 S [M+H] + : , found [M+H] +. 21
22 H NMR spectrum of compound 4S C NMR spectrum of compound 4S
23 H- 1 H COSY diagram of compound 4S HMBC diagram of compound 4S
24 HSQC diagram of compound 4S MS spectrum of compound 4S β-S-(pyrimidine-2)-4-deoxy-podophyllotoxin (4S) 1 H NMR (400 MHz, CDCl 3, δ): 8.57 (d, J = 4.0 Hz, 2H), 7.08 (t, J = 4.0 Hz, 1H), 6.97 (s, 1H), 6.48 (s, 1H), 6.34 (s, 2H), 5.97 (d, J = 8.0 Hz, 2H), 5.45 (d, J = 4.0 Hz, 1H), 4.62 (d, J = 4.0 Hz, 1H), 4.39 (t, J = 8.0 Hz, 1H), 3.88 (t, J = 8.0 Hz, 1H), 3.81 (s, 3H), 3.77 (s, 6H), (m, 2H); 13 C NMR (101 MHz, CDCl 3, δ): , , (2C), (2C), , , , , , , , , , (2C), , 70.68, 60.73, (2C), 47.13, 43.75, 42.40, ESI-MS: calc'd for C 26 H 24 N 2 O 7 S [M+H] + : , found [M+H] + ; calc'd for C 26 H 24 N 2 O 7 S [M+H] + : , found [M+2H] + ; calc'd for C 26 H 24 N 2 O 7 S [M+Na] + : , found [M+Na] + ; calc'd for C 26 H 24 N 2 O 7 S [M+2Na+2K-2H] + : , found [M+2Na+2K-2H]
25 H NMR spectrum of compound 5S C NMR spectrum of compound 5S
26 H- 1 H COSY spectrums for compound 5S HMBC spectrums for compound 5S
27 HSQC spectrums for compound 5S MS spectrum of compound 5S β-S-(benzothiazole-2)-4-deoxy-podophyllotoxin (5S) 1 H NMR (400 MHz, CDCl 3, δ): 7.86 (d, J = 8.0 Hz, 1H), 7.80 (d, J = 8.0 Hz, 1H), 7.45 (t, J = 8.0 Hz, 1H), 7.35 (t, J = 8.0 Hz, 1H), 7.01 (s, 1H), 6.50 (s, 1H), 6.33 (s, 2H), 6.99 (d, J = 8.0 Hz, 2H), 5.77 (d, J = 4.0 Hz, 1H), 4.63 (d, J = 4.0 Hz, 1H), 4.47 (t, J = 8.0 Hz, 1H), 3.99 (t, J = 8.0 Hz, 1H), 3.82 (s, 3H), 3.78 (s, 6H), (m, 1H), (m, 1H); 13 C NMR (101 MHz, CDCl 3, δ): , , (2C), (2C), , , , , , , (2C), , , , , (2C), , 70.79, 60.76, (2C), 49.64, 43.74, 42.58, ESI-MS: calc d for C 29 H 25 NO 7 S 2 [M+H] + : , found [M+H] +. 27
28 H NMR spectrum of compound 6S C NMR spectrum of compound 6S
29 H- 1 H COSY spectrums for compound 6S HMBC spectrums for compound 6S
30 HSQC spectrums for compound 6S MS spectrum of compound 6S β-S-(purine-6)-4-deoxy-podophyllotoxin (6S) 1 H NMR (300 MHz, CDCl 3, δ): 6.87 (s, 1H), 6.54 (s, 1H), 6.27 (s, 2H), 5.99 (d, J = 9.0 Hz, 2H), 5.29 (s, 1H), 4.86 (d, J = 9.0 Hz, 1H), 4.59 (d, J = 9.0 Hz, 1H), (m, 1H), 3.79 (s, 3H), 3.73 (s, 6H), 3.28 (dd, J 1 = 16.0 Hz, J 2 = 6.0 Hz, 1H), (m, 1H); 13 C NMR (75 MHz, CDCl 3, δ): (2C), (2C), , (2C), , (2C), , , (2C), , (2C), 67.61, 66.72, 60.74, (2C), 53.42, , 43.89, 40.47, ESI-MS: calc d for C 27 H 24 N 4 O 7 S [M+H] + : , found [M+H] +. 30
31 H NMR spectrum of compound 3'S C NMR spectrum of compound 3'S
32 H- 1 H COSY spectrums for compound 3'S HMBC spectrums for compound 3'S
33 HSQC spectrums for compound 3'S MS spectrum of compound 3'S β-S-(pyridine-2)-4-deoxy-4'-demethyl-podophyllotoxin (3'S) 1 H NMR (400 MHz, CDCl 3, δ): 8.40 (d, J = 4.0 Hz, 1H), 7.52 (t, J = 8.0 Hz, 1H), 7.18 (d, J = 8.0 Hz, 1H), 7.05 (t, J = 8.0 Hz, 1H), 6.94 (s, 1H), 6.46 (s, 1H), 6.34 (s, 2H), 5.96 (d, J = 12.0 Hz, 2H), 5.56 (d, J = 4.0 Hz, 1H), 5.35 (s, 1H), 4.59 (d, J = 4.0 Hz, 1H), 4.33 (t, J = 8.0 Hz, 1H), 3.85 (t, J = 8.0 Hz, 1H), 3.79 (s, 6H), (m, 2H); 13 C NMR (101 MHz, CDCl 3, δ): , , , , , (2C), , , , , , , , , , (2C), , 70.98, (2C), 45.64, 43.61, 42.61, ESI-MS: calc d for C 26 H 23 NO 7 S [M+H] + : , found [M+H] +. 33
34 H NMR spectrum of compound 4'S C NMR spectrum of compound 4'S
35 H- 1 H COSY spectrums for compound 4'S HMBC spectrums for compound 4'S
36 HSQC spectrums for compound 4'S MS spectrum of compound 4'S β-S-(pyrimidine-2)-4-deoxy-4'-demethyl-podophyllotoxin (4'S) 1 H NMR (300 MHz, CDCl 3, δ): (d, J = 7.2 Hz, 1H), (t, J = 6.9 Hz, 1H), (d, J = 7.8 Hz, 1H), (t, J = 6.0 Hz, 1H), (s, 1H), (s, 1H), (s, 2H), (d, J = 7.2 Hz, 2H), (t, J = 6.0 Hz, 1H), (d, J = 4.2 Hz, 1H), (t, J = 8.1 Hz, 1H), (t, J = 9.0 Hz, 1H), (s, 6H), (m, 2H); 13 C NMR (75 MHz, CDCl 3, δ): , , , , , , , , , , , , , , , (2C), , , (2C), , , , ESI-MS: calc'd for C 25 H 22 N 2 O 7 S [M+H] + : , found [M+H] +. 36
37 H NMR spectrum of compound 5'S C NMR spectrum of compound 5'S
38 H- 1 H COSY spectrums for compound 5'S HMBC spectrums for compound 5'S
39 HSQC spectrums for compound 5'S MS spectrum of compound 5'S. Intens. x MS, 5.9min # m/z 4β-S-(benzothiazole-2)-4-deoxy-4'-demethyl-podophyllotoxin (5'S) 1 H NMR (400 MHz, CDCl 3, δ): 7.88 (d, J = 8.0 Hz, 1H), 7.80 (d, J = 8.0 Hz, 1H), 7.46 (t, J = 8.0 Hz, 1H), 7.35 (t, J = 8.0 Hz, 1H), 7.27 (s, 1H), 6.56 (s, 1H), 6.40 (s, 2H), 6.00 (d, J = 8.0 Hz, 2H), 5.59 (d, J = 4.0 Hz, 1H), 4.65 (d, J = 4.0 Hz, 1H), (m, 2H), 3.80 (s, 6H), (m, 1H), (m, 1H); 13 C NMR(101 MHz, CDCl 3, δ): , , , , , (2C), , , , , , , , , , , , (2C), , 71.55, (2C), 49.75, 47.69, 43.90, ESI-MS: calc d for C 28 H 23 NO 7 S 2 [M+H] + : , found [M+H] +. 39
40 H NMR spectrum of compound 6'S C NMR spectrum of compound 6'S
41 H- 1 H COSY spectrums for compound 6'S HMBC spectrums for compound 6'S
42 HSQC spectrums for compound 6'S MS spectrum of compound 6'S β-S-(purine-6)-4-deoxy-4'-demethyl-podophyllotoxin (6'S) 1 H NMR (400 MHz, DMSO): δ 8.24 (s, 1H), 6.91 (s, 1H), 6.48 (s, 1H), 6.19 (s, 2H), 5.98 (d, J = 8.0 Hz, 2H), 5.42 (d, J = 8.0 Hz, 1H), 4.71 (dd, J = 4.0 Hz, 1H), 4.47 (d, J = 8.0 Hz, 1H), 4.31 (t, J = 8.0 Hz, 1H), 4.15 (t, J = 8.0 Hz, 1H), 3.59 (s, 6H), (m, 1H), (m, 1H); 13 C NMR (101 MHz, DMSO): δ (2C), , (2C), (2C), , (2C), , (2C), , , (2C), , 68.11, 65.41, (2C), 55.36, 43.41, ESI-MS: calc d for C 27 H 25 NO 7 S [M+H] + : , found [M+H] +. 42
43 H NMR spectrum of compound 1N C NMR spectrum of compound 1N
44 H- 1 H COSY spectrums for compound 1N HMBC spectrums for compound 1N
45 HSQC spectrums for compound 1N MS spectrum of compound 1N β-N-(1,2,4-trizole-3)-4-deoxy-podophyllotoxin (1N). 1 H NMR (400 MHz, CDCl 3 ): δ 6.83 (s, 1H), 6.52 (s, 1H), 6.27 (s, 2H), 5.99 (d, J = 16 Hz, 2H), 4.60 (dd, J = 4.0 Hz, 2H), 4.37 (t, J = 4.0 Hz, 1H), 4.24 (t, J = 4.0 Hz, 1H), 3.80 (s, 3H), 3.74 (s, 6H), 3.71 (t, J = 4.0 Hz, 1H), (dd, J = 8.0 Hz, 1H), (m, 1H); 13 C NMR (101 MHz, CDCl 3 ): δ , , (3C), , , , , , , , , (2C), , , , (2C), , , , ; ESI-MS: calc d for C 24 H 24 N 4 O 7 [M+H] + : , found [M+H]
46 H NMR spectrum of compound 2N C NMR spectrum of compound 2N
47 H- 1 H COSY spectrums for compound 2N HMBC spectrums for compound 2N
48 HSQC spectrums for compound 2N MS spectrum of compound 2N β-N-(1,3,4-thiodiazole-2)-4-deoxy-podophyllotoxin (2N). 1 H NMR (400 MHz, CDCl 3 ): δ 6.82 (s, 1H), 6.52 (s, 1H), 6.27 (s, 2H), 5.98 (d, J = 3.6 Hz, 2H), 5.30 (s, 1H), 5.46 (dd, 2H, J = 4.0 Hz), 4.36 (t, 1H, J = 8.0 Hz), 4.24 (t, 1H, J = 8.0 Hz), 3.80 (s, 3H), 3.74 (s, 6H), 3.72 (t, J = 8.0 Hz, 1H), 3.38 (m, J = 4.0 Hz, 1H), (m, 1H); 13 C NMR(101 MHz, CDCl 3 ): δ , (2C), , , , , , , , , (2C), , , 71.31, 71.25, 67.76, 60.73, (2C), 43.86, 41.19, 38.43; ESI-MS: calc d for C 24 H 23 N 3 O 7 S: , found [M+H] +. 48
49 H NMR spectrum of compound 3N C NMR spectrum of compound 3N
50 H- 1 H COSY diagram of compound 3N HMBC diagram of compound 3N
51 HSQC diagram of compound 3N MS spectrum of compound 3N 4β-N-(pyridine-2)-4-deoxy-podophyllotoxin (3N). 1 H NMR (300 MHz, CD 3 OD): δ (d, J = 6.3 Hz, 2H), (m, 2H), (t, J = 7.8 Hz,1H), (s, 2H), (s, 2H), (s, 2H), (s, 1H), (d, J = 4.5 Hz, 3H), (s, 6H), (s, 3H), (m, 2H); 13 C NMR(75 MHz, CD 3 OD): δ , , , , , (2C), , , , , , , , (2C), , , (2C), , , , , ESI-MS: calc d for C 27 H 26 N 2 O 7 [M+H] + : , found [M+H] H NMR spectrum of compound 4N
52 C NMR spectrum of compound 4N H- 1 H COSY diagram of compound 4N
53 HMBC diagram of compound 4N HSQC diagram of compound 4N
54 MS spectrum of compound 4N β-N-(pyrimidine-2)-4-deoxy-podophyllotoxin (4N) 1 H NMR (300 MHz, CDCl 3, δ): (s, 2H), (s, 1H), (s, 1H), (s, 1H), (s, 1H), (s, 2H), (d, J = 8.4 Hz, 2H), (s, 1H), (s, 1H), (t, J = 0.6 Hz, 1H), (s, 3H), (s, 6H), (s, 3H); 13 C NMR(75 MHz, CDCl 3, δ): , , (2C), (2C), , , , , , , , , , (2C), , , , (2C), , , , ESI-MS: calc d for C 26 H 25 N 3 O 7 [M+H] + : , found [M+H] H NMR spectrum of compound 5N
55 H- 1 H COSY diagram of compound 5N HMBC diagram of compound 5N
56 HSQC diagram of compound 5N MS spectrum of compound 5N β-N-(pyrimidine-2)-4-deoxy-podophyllotoxin (5N) 1 H NMR (400 MHz, CDCl 3, δ): 7.62 (d, J = 8.0 Hz, 1H), 7.57 (d, J = 8.0 Hz, 1H), 7.36 (t, J = 8.0 Hz, 1H), 7.19 (t, J = 4.0 Hz, 1H), 6.88 (s, 1H), 6.51 (s, 1H), 6.30 (s, 2H), 5.97 (d, J = 8.0 Hz, 2H), 5.39 (s, 1H), 4.61 (d, J = 4.0 Hz, 1H), 4.50 (t, J = 4.0 Hz, 1H), 4.02 (t, J = 8.0 Hz, H), 3.80 (s, 3H), 3.75 (s, 6H), (m, 2H); 13 C NMR(101 MHz, CDCl 3, δ): , , (2C), , (2C), , , , , , , , (2C), , , (2C), , 69.00, 60.75, (2C), 53.91, 43.69, 41.92, ESI-MS: calc d for C 29 H 26 N 2 O 7 S [M+H] + : , found [M+H] +. 56
57 H NMR spectrum of compound 6N C NMR spectrum of compound 6N
58 H- 1 H COSY diagram of compound 6N HMBC diagram of compound 6N
59 HSQC diagram of compound 6N MS spectrum of compound 6N β-N-(pyrimidine-2)-4-deoxy-podophyllotoxin (6N) 1 H NMR (400 MHz, CDCl 3, δ): 6.87 (s, 1H), 6.54 (s, 1H), 6.27 (s, 2H), 5.99 (d, J = 12.0 Hz, 2H), 5.29 (s, 1H), 4.86 (d, J = 4.0 Hz, 1H), 4.60 (d, J = 4.0 Hz, 1H), (m, 2H), 3.79 (s, 3H), 3.73 (s, 6H), (m, 1H), (m, 1H); 13 C NMR (101 MHz, CDCl 3, δ): (2C), (2C), (2C), , , (2C), , , (2C), , (2C), , 67.61, 66.72, 60.74, (2C), 53.42, 43.89, 40.47, ESI-MS: calc d for C 27 H 25 N 5 O 7 [M+H] + : , found [M+H] +. 59
60 H NMR spectrum of compound 1'N C NMR spectrum of compound 1'N
61 H- 1 H COSY diagram of compound 1'N HMBC diagram of compound 1'N 61
62 HSQC diagram of compound 1'N MS spectrum of compound 1'N β-N-(1,2,4-trizole-3)-4-deoxy-4'-demethyl-podophyllotoxin (1'N) 1 H NMR(400 MHz, CD 3 OD, CD 3 Cl): δ 6.87 (s, 1H), 6.50 (s, 1H), 6.28 (s, 2H), 5.98 (d, 2H, J = 8.0 Hz), (d, 1H, J = 1.8 Hz), (s, 1H), (d, 1H), (d, 1H, J = 2.4 Hz, (t, 1H J = 4.5 Hz,), (t, 1H, J = 4.8 Hz), (s, 6H), (t, 1H, J = 2.1Hz), (dd, 1H, J = 2.4Hz); 13 C NMR (101 MHz, CD 3 OD, CD 3 Cl): δ , , , , , , , , , , , , , 75.14, 75.04, 72.12, 67.42, (2C), 57.26, 47.52, 45.18, ESI-MS: calc d for C 23 H 22 N 4 O 7 [M+H] + : , found [M+H] +. 62
63 H NMR spectrum of compound 2'N C NMR spectrum of compound 2'N
64 H- 1 H COSY diagram of compound 2'N HMBC diagram of compound 2'N
65 HSQC diagram of compound 2'N MS spectrum of compound 2'N β-N-(1,3,4-thiodizole-2)-4-deoxy-4'-demethyl-podophyllotoxin (2'N) 1 H NMR(400 MHz, CDCl 3 ): δ 6.82 (s, 1H), 6.51 (s, 1H), 6.27 (s, 2H), 5.98 (d, J = 16.0 Hz, 2H), 5.38 (s, 1H), 4.59 (dd, J = 4.0 Hz, 2H), 4.34 (t, J = 4.0 Hz, 1H), 4.23 (t, J = 8.0 Hz, 1H), 3.76(s, 6H), 3.71 (t, J = 8.0 Hz, 1H), (m, 1H), (m, 1H); 13 C NMR (101 MHz, CDCl 3 ): δ , , (2C), , , , , , , , (2C), , 71.32, 71.24, 67.76, (2C), 43.69, 41.29, 38.37, ESI-MS: calc d for C 23 H 21 N 3 O 7 S [M+H] + : , found [M+H] +. 65
66 H NMR spectrum of compound 3'N C NMR spectrum of compound 3'N
67 H- 1 H COSY diagram of compound 3'N HMBC diagram of compound 3'N
68 HSQC diagram of compound 3'N MS spectrum of compound 3'N β-N-(pyridine-2)-4-deoxy-4'-demethyl-podophyllotoxin (3'N) 1 H NMR (300 MHz, CD 3 OD): δ (d, J = 6.3 Hz, 2H), (s, 2H), (t, J = 6.6 Hz, 1H), (s, 1H), (s, 2H), (s, 2H), (d, J = 3.0 Hz, 3H), (t, J = 9.0 Hz, 1H), (s, 6H), (s, 2H); 13 C NMR (75 MHz, CD 3 OD): δ , , , (2C), , , , , , , , , , , , (2C), , , (2C), , , , ESI-MS: calc d for C 26 H 24 N 2 O 7 [M+H] + : , found [M+H] +. 68
69 H NMR spectrum of compound 4'N C NMR spectrum of compound 4'N
70 H- 1 H COSY diagram of compound 4'N HMBC diagram of compound 4'N
71 HSQC spectrums for compound 4'N MS spectrum of compound 4'N β-N-(pyrimidine-2)-4-deoxy-4'-demethyl-podophyllotoxin (4'N) 1 H NMR (300 MHz, CDCl 3 ): δ (s, 2H), (s, 1H), (s, 1H), (s, 1H), (s, 2H), (d, J = 9.6 Hz, 2H), (d, J = 2.7 Hz, 1H), (s, 1H), (t, J = 7.8 Hz, 1H), (s, 1H), (s, 1H), (t, J = 5.4 Hz, 1H), (s, 6H), (s, 2H); 13 C NMR (75 MHz, CDCl 3 ): δ , , , , , (2C), , , , , , , , (2C), , , (2C), , , , , ESI-MS: calc d for C 25 H 23 N 3 O 7 [M+H] + : , found [M+H] +. 71
72 H NMR spectrum of compound 5'N C NMR spectrum of compound 5'N
73 H- 1 H COSY diagram of compound 5'N HMBC diagram of compound 5'N
74 HSQC diagram of compound 5'N MS spectrum of compound 5'N β-N-(benzothiazole-2)-4-deoxy-4'-demethyl-podophyllotoxin (5'N) 1 H NMR (400 MHz, CDCl 3 ): δ 7.61 (d, J = 8.0 Hz, 1H), 7.56 (d, J = 8.0 Hz, 1H), 7.34 (t, J = 8.0 Hz, 1H), 7.15 (t, J = 8.0 Hz, 1H), 6.88 (s, 1H), 6.51 (s, 1H), 6.31 (s, 2H), 5.9 7(d, J = 12.0 Hz, 2H), 5.42 (d, J = 4.0 Hz, 1H), 4.58 (d, J = 4.0 Hz, 1H), 4.49 (t, J = 8.0 Hz, 1H), 4.00 (t, J = 8.0 Hz, 1H), 3.77 (s, 6H), (m, 2H); 13 C NMR (101 MHz, CDCl 3 ): δ (2C), , , , , (2C), , , , , , (2C), , , , , (2C), , 69.25, (2C), 53.40, 43.52, 42.02, ESI-MS: calc d for C 28 H 24 N 2 O 7 S [M+H] + : , found [M+H] +. 74
75 H NMR spectrum of compound 6'N C NMR spectrum of compound 6'N
76 H- 1 H COSY diagram of compound 6'N HMBC diagram of compound 6'N
77 HSQC diagram of compound 6'N MS spectrum of compound 6'N H NMR (400 MHz, DMSO): δ 8.24 (s, 1H), 6.91 (s, 1H), 6.48 (s, 1H), 6.19 (s, 2H), 5.98 (d, J = 8.0 Hz, 2H), 5.42 (d, J = 8.0 Hz, 1H), 4.71 (dd, J = 4.0 Hz, 1H), 4.47 (d, J = 8.0 Hz, 1H), 4.31 (t, J = 8.0 Hz, 1H), 4.15 (t, J = 8.0 Hz, 1H), 3.59 (s, 6H), (m, 1H), (m, 1H); 13 C NMR (101 MHz, DMSO): δ (2C), , (2C), (2C), , (2C), , (2C), , , (2C), , 68.11, 65.41, (2C), 55.36, 43.41, ESI-MS: calc d for C 26 H 23 N 5 O 7 [M+H] + : , found [M+H] + 77
Supporting Information
 Supporting Information Synthesis and Electroactive Properties of Poly(amidoamine) Dendrimers with an Aniline Pentamer Shell Wei-I Hung a, Chih-Bing Hung a, Ya-Han Chang a, Jiun-Kuang Dai a, Yan Li b, Hai
Supporting Information Synthesis and Electroactive Properties of Poly(amidoamine) Dendrimers with an Aniline Pentamer Shell Wei-I Hung a, Chih-Bing Hung a, Ya-Han Chang a, Jiun-Kuang Dai a, Yan Li b, Hai
Supporting Information
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2018 Supporting Information Crotonols A and B, Two Rare Tigliane Diterpenoid
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2018 Supporting Information Crotonols A and B, Two Rare Tigliane Diterpenoid
Malgorzata Korycka-Machala, Marcin Nowosielski, Aneta Kuron, Sebastian Rykowski, Agnieszka Olejniczak, Marcin Hoffmann and Jaroslaw Dziadek
 Molecules 2017, 21, 154; doi:10.3390/molecules22010154 Supplementary Materials: Naphthalimides Selectively Inhibit the Activity of Bacterial, Replicative DNA Ligases and Display Bactericidal Effect against
Molecules 2017, 21, 154; doi:10.3390/molecules22010154 Supplementary Materials: Naphthalimides Selectively Inhibit the Activity of Bacterial, Replicative DNA Ligases and Display Bactericidal Effect against
Supporting Information
 Supporting Information Mitochondria-Targeting Polydopamine Nanocomposites as Chemophotothermal Therapeutics for Cancer Zhuo Wang *,, Yuzhi Chen, Hui Zhang, Yawen Li, Yufan Ma, Jia Huang, Xiaolei Liu, Fang
Supporting Information Mitochondria-Targeting Polydopamine Nanocomposites as Chemophotothermal Therapeutics for Cancer Zhuo Wang *,, Yuzhi Chen, Hui Zhang, Yawen Li, Yufan Ma, Jia Huang, Xiaolei Liu, Fang
Supporting Information
 Supporting Information for AgOTf-catalyzed one-pot reactions of 2-alkynylbenzaldoximes with α,β-unsaturated carbonyl compounds Qiuping Ding 1, Dan Wang 1, Puying Luo* 2, Meiling Liu 1, Shouzhi Pu* 3 and
Supporting Information for AgOTf-catalyzed one-pot reactions of 2-alkynylbenzaldoximes with α,β-unsaturated carbonyl compounds Qiuping Ding 1, Dan Wang 1, Puying Luo* 2, Meiling Liu 1, Shouzhi Pu* 3 and
Supporting Information
 Supporting Information for Lewis acid-catalyzed redox-neutral amination of 2-(3-pyrroline-1-yl)benzaldehydes via intramolecular [1,5]-hydride shift/isomerization reaction Chun-Huan Jiang, Xiantao Lei,
Supporting Information for Lewis acid-catalyzed redox-neutral amination of 2-(3-pyrroline-1-yl)benzaldehydes via intramolecular [1,5]-hydride shift/isomerization reaction Chun-Huan Jiang, Xiantao Lei,
Discovery of multi-target receptor tyrosine kinase inhibitors as novel anti-angiogenesis agents
 Discovery of multi-target receptor tyrosine kinase inhibitors as novel anti-angiogenesis agents Jinfeng Wang, Lin Zhang, Xiaoyan Pan, Bingling Dai, Ying Sun, Chuansheng Li, Jie Zhang School of Pharmacy,
Discovery of multi-target receptor tyrosine kinase inhibitors as novel anti-angiogenesis agents Jinfeng Wang, Lin Zhang, Xiaoyan Pan, Bingling Dai, Ying Sun, Chuansheng Li, Jie Zhang School of Pharmacy,
SUPPORTING INFORMATION
 SUPPORTING INFORMATION Heronamides A C, new polyketide macrolactams from an Australian marine-derived Streptomyces sp. A biosynthetic case for synchronized tandem electrocyclization. Ritesh Raju, Andrew
SUPPORTING INFORMATION Heronamides A C, new polyketide macrolactams from an Australian marine-derived Streptomyces sp. A biosynthetic case for synchronized tandem electrocyclization. Ritesh Raju, Andrew
A new ent-kaurane diterpene from Euphorbia stracheyi Boiss
 SUPPLEMENTARY MATERIAL A new ent-kaurane diterpene from Euphorbia stracheyi Boiss Tie Liu a, Qian Liang a,b, Na-Na Xiong a, Lin-Feng Dai a, Jun-Ming Wang a,b, Xiao-Hui Ji c, Wen-Hui Xu a, * a Key Laboratory
SUPPLEMENTARY MATERIAL A new ent-kaurane diterpene from Euphorbia stracheyi Boiss Tie Liu a, Qian Liang a,b, Na-Na Xiong a, Lin-Feng Dai a, Jun-Ming Wang a,b, Xiao-Hui Ji c, Wen-Hui Xu a, * a Key Laboratory
Supporting Information. Research Center for Marine Drugs, Department of Pharmacy, State Key Laboratory
 Supporting Information Dysiherbols A C and Dysideanone E, Cytotoxic and NF-κB Inhibitory Tetracyclic Meroterpenes from a Dysidea sp. Marine Sponge Wei-Hua Jiao,, Guo-Hua Shi,, Ting-Ting Xu,, Guo-Dong Chen,
Supporting Information Dysiherbols A C and Dysideanone E, Cytotoxic and NF-κB Inhibitory Tetracyclic Meroterpenes from a Dysidea sp. Marine Sponge Wei-Hua Jiao,, Guo-Hua Shi,, Ting-Ting Xu,, Guo-Dong Chen,
Supporting Information for. Update of spectroscopic data for 4-hydroxyldictyolactone and dictyol E isolated from a Halimeda stuposa - Dictyota
 1 Supporting Information for Update of spectroscopic data for 4-hydroxyldictyolactone and dictyol E isolated from a Halimeda stuposa - Dictyota sp. Assemblage Simon P. B. Ovenden, Jonathan L. Nielson,
1 Supporting Information for Update of spectroscopic data for 4-hydroxyldictyolactone and dictyol E isolated from a Halimeda stuposa - Dictyota sp. Assemblage Simon P. B. Ovenden, Jonathan L. Nielson,
Free Radical Initiated Coupling Reaction of Alcohols and. Alkynes: not C-O but C-C Bond Formation. Context. General information 2. Typical procedure 2
 Free Radical Initiated Coupling Reaction of Alcohols and Alkynes: not C-O but C-C Bond Formation Zhongquan Liu,* Liang Sun, Jianguo Wang, Jie Han, Yankai Zhao, Bo Zhou Institute of Organic Chemistry, Gannan
Free Radical Initiated Coupling Reaction of Alcohols and Alkynes: not C-O but C-C Bond Formation Zhongquan Liu,* Liang Sun, Jianguo Wang, Jie Han, Yankai Zhao, Bo Zhou Institute of Organic Chemistry, Gannan
Copper-catalyzed formal O-H insertion reaction of α-diazo-1,3-dicarb- onyl compounds to carboxylic acids with the assistance of isocyanide
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Copper-catalyzed formal O-H insertion reaction of α-diazo-1,3-dicarb- onyl compounds to carboxylic
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Copper-catalyzed formal O-H insertion reaction of α-diazo-1,3-dicarb- onyl compounds to carboxylic
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information Syntheses and structures of copper complexes of
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information Syntheses and structures of copper complexes of
Supporting Information for
 Supporting Information for An atom-economic route to densely functionalized thiophenes via base-catalyzed rearrangement of 5-propargyl-2H-thiopyran-4(3H)-ones Chunlin Tang a, Jian Qin b, Xingqi Li *a a
Supporting Information for An atom-economic route to densely functionalized thiophenes via base-catalyzed rearrangement of 5-propargyl-2H-thiopyran-4(3H)-ones Chunlin Tang a, Jian Qin b, Xingqi Li *a a
Supporting Information
 Chloromethylhalicyclamine B, a Marine-Derived Protein Kinase CK1δ/ε Inhibitor Germana Esposito, Marie-Lise Bourguet-Kondracki, * Linh H. Mai, Arlette Longeon, Roberta Teta, Laurent Meijer, Rob Van Soest,
Chloromethylhalicyclamine B, a Marine-Derived Protein Kinase CK1δ/ε Inhibitor Germana Esposito, Marie-Lise Bourguet-Kondracki, * Linh H. Mai, Arlette Longeon, Roberta Teta, Laurent Meijer, Rob Van Soest,
Table of Contents 1 Supplementary Data MCD
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2017 Supporting Information for Magnetic circular dichroism and density functional theory
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2017 Supporting Information for Magnetic circular dichroism and density functional theory
Eremophila spp. by Combined use of Dual High-Resolution PTP1B and α- Glucosidase Inhibition Profiling and HPLC-HRMS-SPE-NMR
 Supporting Information for: Identification of PTP1B and α-glucosidase Inhibitory Serrulatanes from Eremophila spp. by Combined use of Dual High-Resolution PTP1B and α- Glucosidase Inhibition Profiling
Supporting Information for: Identification of PTP1B and α-glucosidase Inhibitory Serrulatanes from Eremophila spp. by Combined use of Dual High-Resolution PTP1B and α- Glucosidase Inhibition Profiling
D-Glucosamine-derived copper catalyst for Ullmann-type C- N coupling reaction: theoretical and experimental study
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 D-Glucosamine-derived copper catalyst for Ullmann-type C- N coupling reaction: theoretical
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 D-Glucosamine-derived copper catalyst for Ullmann-type C- N coupling reaction: theoretical
Divergent synthesis of various iminocyclitols from D-ribose
 Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 205 Divergent synthesis of various iminocyclitols from D-ribose Ramu Petakamsetty,
Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 205 Divergent synthesis of various iminocyclitols from D-ribose Ramu Petakamsetty,
New Cytotoxic Constituents from the Red Sea Soft Coral Nephthea sp.
 SUPPLEMENTARY MATERIAL New Cytotoxic Constituents from the Red Sea Soft Coral Nephthea sp. Mohamed-Elamir F. Hegazy a,*, Amira M. Gamal-Eldeen b, Tarik A. Mohamed a, Montaser A. Alhammady c, Abuzeid A.
SUPPLEMENTARY MATERIAL New Cytotoxic Constituents from the Red Sea Soft Coral Nephthea sp. Mohamed-Elamir F. Hegazy a,*, Amira M. Gamal-Eldeen b, Tarik A. Mohamed a, Montaser A. Alhammady c, Abuzeid A.
C H Activation of Cp* Ligand Coordinated to Ruthenium. Center: Synthesis and Reactivity of a Thiolate-Bridged
 Supporting Information C H Activation of Cp* Ligand Coordinated to Ruthenium Center: Synthesis and Reactivity of a Thiolate-Bridged Diruthenium Complex Featuring Fulvene-like Cp* Ligand Xiaoxiao Ji, Dawei
Supporting Information C H Activation of Cp* Ligand Coordinated to Ruthenium Center: Synthesis and Reactivity of a Thiolate-Bridged Diruthenium Complex Featuring Fulvene-like Cp* Ligand Xiaoxiao Ji, Dawei
Metal-free Oxidative Coupling of Amines with Sodium Sulfinates: A Mild Access to Sulfonamides
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting information for Metal-free Oxidative Coupling of Amines with Sodium Sulfinates:
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting information for Metal-free Oxidative Coupling of Amines with Sodium Sulfinates:
Phosphorus Oxychloride as an Efficient Coupling Reagent for the Synthesis of Ester, Amide and Peptide under Mild Conditions
 Supplementary Information for Phosphorus xychloride as an Efficient Coupling Reagent for the Synthesis of Ester, Amide and Peptide under Mild Conditions u Chen,* a,b Xunfu Xu, a Liu Liu, a Guo Tang,* a
Supplementary Information for Phosphorus xychloride as an Efficient Coupling Reagent for the Synthesis of Ester, Amide and Peptide under Mild Conditions u Chen,* a,b Xunfu Xu, a Liu Liu, a Guo Tang,* a
Supporting Information
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2017 Modular Synthesis of Propargylamine Modified Cyclodextrins by a Gold(III)-catalyzed Three Component
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2017 Modular Synthesis of Propargylamine Modified Cyclodextrins by a Gold(III)-catalyzed Three Component
Copper-Catalyzed Oxidative Dehydrogenative N-N Bond. Formation for the Synthesis of N,N -Diarylindazol-3-ones
 Electronic Supplementary Material (ESI) for Organic Chemistry Frontiers. This journal is the Partner Organisations 2016 Supporting information Copper-Catalyzed Oxidative Dehydrogenative - Bond Formation
Electronic Supplementary Material (ESI) for Organic Chemistry Frontiers. This journal is the Partner Organisations 2016 Supporting information Copper-Catalyzed Oxidative Dehydrogenative - Bond Formation
A facile and general route to 3-((trifluoromethyl)thio)benzofurans and 3-((trifluoromethyl)thio)benzothiophenes
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 A facile and general route to 3-((trifluoromethyl)thio)benzofurans and 3-((trifluoromethyl)thio)benzothiophenes
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 A facile and general route to 3-((trifluoromethyl)thio)benzofurans and 3-((trifluoromethyl)thio)benzothiophenes
SUPPLEMENTARY MATERIAL
 10.1071/CH16014_AC The Authors 2016 Australian Journal of Chemistry 2016, 69(9), 1049-1053 SUPPLEMENTARY MATERIAL A Green Approach for the Synthesis of Novel 7,11-Dihydro-6H-chromeno[3,4- e]isoxazolo[5,4-b]pyridin-6-one
10.1071/CH16014_AC The Authors 2016 Australian Journal of Chemistry 2016, 69(9), 1049-1053 SUPPLEMENTARY MATERIAL A Green Approach for the Synthesis of Novel 7,11-Dihydro-6H-chromeno[3,4- e]isoxazolo[5,4-b]pyridin-6-one
Enantioselective Organocatalytic Michael Addition of Isorhodanines. to α, β-unsaturated Aldehydes
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2016 Enantioselective Organocatalytic Michael Addition of Isorhodanines to α,
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2016 Enantioselective Organocatalytic Michael Addition of Isorhodanines to α,
Synthesis, structural studies and stability of the model, cysteine containing DNA-protein cross-links
 Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2017 ELECTRONIC SUPPLEMENTARY INFORMATION
Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2017 ELECTRONIC SUPPLEMENTARY INFORMATION
Supplementary Information. Living Ring-Opening Polymerization of Lactones by N-Heterocyclic Olefin/Al(C 6 F 5 ) 3
 Supplementary Information Living Ring-Opening Polymerization of Lactones by N-Heterocyclic Olefin/Al(C 6 F 5 ) 3 Lewis Pairs: Structures of Intermediates, Kinetics, and Mechanism Qianyi Wang, Wuchao Zhao,
Supplementary Information Living Ring-Opening Polymerization of Lactones by N-Heterocyclic Olefin/Al(C 6 F 5 ) 3 Lewis Pairs: Structures of Intermediates, Kinetics, and Mechanism Qianyi Wang, Wuchao Zhao,
Supporting Information. Copyright Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2007
 Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2007 Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Supporting Information for High-Throughput Substrate
Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2007 Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Supporting Information for High-Throughput Substrate
SUPPORTING INFORMATION
 SUPPORTING INFORMATION Trichodermides A E: New Peptaibols isolated from Australian Termite Nestderived Fungus Trichoderma virens CMB-TN16 Wei-Hua Jiao,, Zeinab Khalil, Pradeep Dewapriya, Angela A. Salim,
SUPPORTING INFORMATION Trichodermides A E: New Peptaibols isolated from Australian Termite Nestderived Fungus Trichoderma virens CMB-TN16 Wei-Hua Jiao,, Zeinab Khalil, Pradeep Dewapriya, Angela A. Salim,
Table S1. Summary of data collections and structure refinements for crystals 1Rb-1h, 1Rb-2h, and 1Rb-4h.
 Supporting Information [NH 3 CH 3 ] [In SbS 9 SH]: A novel methylamine-directed indium thioantimonate with Rb + ion-exchange property Kai-Yao Wang a,b, Mei-Ling Feng a, Jian-Rong Li a and Xiao-Ying Huang
Supporting Information [NH 3 CH 3 ] [In SbS 9 SH]: A novel methylamine-directed indium thioantimonate with Rb + ion-exchange property Kai-Yao Wang a,b, Mei-Ling Feng a, Jian-Rong Li a and Xiao-Ying Huang
Supporting Information. Asymmetric Binary-acid Catalysis with Chiral. Phosphoric Acid and MgF 2 : Catalytic
 Supporting Information Asymmetric Binary-acid Catalysis with Chiral Phosphoric Acid and MgF 2 : Catalytic Enantioselective Friedel-Crafts Reactions of β,γ- Unsaturated-α-Ketoesters Jian Lv, Xin Li, Long
Supporting Information Asymmetric Binary-acid Catalysis with Chiral Phosphoric Acid and MgF 2 : Catalytic Enantioselective Friedel-Crafts Reactions of β,γ- Unsaturated-α-Ketoesters Jian Lv, Xin Li, Long
Supporting Information. Microwave-assisted construction of triazole-linked amino acid - glucoside conjugates as novel PTP1B inhibitors
 Supporting Information Microwave-assisted construction of triazole-linked amino acid - glucoside conjugates as novel PTP1B inhibitors Xiao-Peng He, abd Cui Li, d Xiao-Ping Jin, b Zhuo Song, b Hai-Lin Zhang,
Supporting Information Microwave-assisted construction of triazole-linked amino acid - glucoside conjugates as novel PTP1B inhibitors Xiao-Peng He, abd Cui Li, d Xiao-Ping Jin, b Zhuo Song, b Hai-Lin Zhang,
Direct Palladium-Catalyzed Arylations of Aryl Bromides. with 2/9-Substituted Pyrimido[5,4-b]indolizines
![Direct Palladium-Catalyzed Arylations of Aryl Bromides. with 2/9-Substituted Pyrimido[5,4-b]indolizines Direct Palladium-Catalyzed Arylations of Aryl Bromides. with 2/9-Substituted Pyrimido[5,4-b]indolizines](/thumbs/87/95187440.jpg) Direct Palladium-Catalyzed Arylations of Aryl Bromides with 2/9-Substituted Pyrimido[5,4-b]indolizines Min Jiang, Ting Li, Linghua Meng, Chunhao Yang,* Yuyuan Xie*, and Jian Ding State Key Laboratory of
Direct Palladium-Catalyzed Arylations of Aryl Bromides with 2/9-Substituted Pyrimido[5,4-b]indolizines Min Jiang, Ting Li, Linghua Meng, Chunhao Yang,* Yuyuan Xie*, and Jian Ding State Key Laboratory of
Supporting Information for. A New Diketopiperazine, Cyclo-(4-S-Hydroxy-R-Proline-R-Isoleucine), from an Australian Specimen of the Sponge
 1 Supporting Information for A New Diketopiperazine, Cyclo-(4-S-Hydroxy-R-Proline-R-Isoleucine), from an Australian Specimen of the Sponge Stelletta Sp. Simon P. B. Ovenden, Jonathan L. Nielson, Catherine
1 Supporting Information for A New Diketopiperazine, Cyclo-(4-S-Hydroxy-R-Proline-R-Isoleucine), from an Australian Specimen of the Sponge Stelletta Sp. Simon P. B. Ovenden, Jonathan L. Nielson, Catherine
Supporting Information
 Supporting Information Lewis acid catalyzed ring-opening reactions of methylenecyclopropanes with diphenylphosphine oxide in the presence of sulfur or selenium Min Shi,* Min Jiang and Le-Ping Liu State
Supporting Information Lewis acid catalyzed ring-opening reactions of methylenecyclopropanes with diphenylphosphine oxide in the presence of sulfur or selenium Min Shi,* Min Jiang and Le-Ping Liu State
Supplementary material
 Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2015 Supplementary material Recyclable
Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2015 Supplementary material Recyclable
Supporting Information
 Supporting Information Molecular Cage Impregnated Palladium Nanoparticles: Efficient, Additive-Free Heterogeneous Catalysts for Cyanation of Aryl Halides. Bijnaneswar Mondal, Koushik Acharyya, Prodip Howlader
Supporting Information Molecular Cage Impregnated Palladium Nanoparticles: Efficient, Additive-Free Heterogeneous Catalysts for Cyanation of Aryl Halides. Bijnaneswar Mondal, Koushik Acharyya, Prodip Howlader
Electronic Supplementary Information
 Electronic Supplementary Information Unprecedented Carbon-Carbon Bond Cleavage in Nucleophilic Aziridine Ring Opening Reaction, Efficient Ring Transformation of Aziridines to Imidazolidin-4-ones Jin-Yuan
Electronic Supplementary Information Unprecedented Carbon-Carbon Bond Cleavage in Nucleophilic Aziridine Ring Opening Reaction, Efficient Ring Transformation of Aziridines to Imidazolidin-4-ones Jin-Yuan
Selective mono reduction of bisphosphine
 Griffith Research Online https://research-repository.griffith.edu.au Selective mono reduction of bisphosphine oxides under mild conditions Author etersson, Maria, Loughlin, Wendy, Jenkins, Ian ublished
Griffith Research Online https://research-repository.griffith.edu.au Selective mono reduction of bisphosphine oxides under mild conditions Author etersson, Maria, Loughlin, Wendy, Jenkins, Ian ublished
Chemical Constituents and Antioxidant Activity of Teucrium barbeyanum Aschers.
 Supporting Information Rec. Nat. Prod. 9:1 (2015) 159-163 Chemical Constituents and Antioxidant Activity of Teucrium barbeyanum Aschers. Mohamed Ali A. Alwahsh, Melati Khairuddean * and Wong Keng Chong
Supporting Information Rec. Nat. Prod. 9:1 (2015) 159-163 Chemical Constituents and Antioxidant Activity of Teucrium barbeyanum Aschers. Mohamed Ali A. Alwahsh, Melati Khairuddean * and Wong Keng Chong
Uncovering the impact of capsule shaped amine-type ligands on. Am(III)/Eu(III) separation
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 Uncovering the impact of capsule shaped amine-type ligands on Am(III)/Eu(III) separation
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 Uncovering the impact of capsule shaped amine-type ligands on Am(III)/Eu(III) separation
Lewis Acid Catalyzed Propargylation of Arenes with O-Propargyl Trichloroacetimidate: Synthesis of 1,3-Diarylpropynes
 Supporting Information for Lewis Acid Catalyzed Propargylation of Arenes with O-Propargyl Trichloroacetimidate: Synthesis of 1,3-Diarylpropynes Changkun Li and Jianbo Wang* Beijing National Laboratory
Supporting Information for Lewis Acid Catalyzed Propargylation of Arenes with O-Propargyl Trichloroacetimidate: Synthesis of 1,3-Diarylpropynes Changkun Li and Jianbo Wang* Beijing National Laboratory
Room Temperature Highly Diastereoselective Zn-Mediated. Allylation of Chiral N-tert-Butanesulfinyl Imines: Remarkable Reaction Condition Controlled
 Supporting Information for: Room Temperature Highly Diastereoselective Zn-Mediated Allylation of Chiral N-tert-Butanesulfinyl Imines: Remarkable Reaction Condition Controlled Stereoselectivity Reversal
Supporting Information for: Room Temperature Highly Diastereoselective Zn-Mediated Allylation of Chiral N-tert-Butanesulfinyl Imines: Remarkable Reaction Condition Controlled Stereoselectivity Reversal
Electronic Supplementary Information (ESI)
 Electronic Supplementary Information (ESI) Lanthanide metal-organic frameworks constructed by asymmetric 2-nitro-biphenyl-4,4 -dicarboxylate ligand: syntheses, structures, luminescence and magnetic investigations
Electronic Supplementary Information (ESI) Lanthanide metal-organic frameworks constructed by asymmetric 2-nitro-biphenyl-4,4 -dicarboxylate ligand: syntheses, structures, luminescence and magnetic investigations
Electronic Supporting Information. 3-Aminothiophenecarboxylic acid (3-Atc)-induced folding in peptides
 Electronic Supplementary Material (ESI for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2016 Electronic Supporting Information
Electronic Supplementary Material (ESI for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2016 Electronic Supporting Information
Highly enantioselective cascade synthesis of spiropyrazolones. Supporting Information. NMR spectra and HPLC traces
 Highly enantioselective cascade synthesis of spiropyrazolones Alex Zea a, Andrea-Nekane R. Alba a, Andrea Mazzanti b, Albert Moyano a and Ramon Rios a,c * Supporting Information NMR spectra and HPLC traces
Highly enantioselective cascade synthesis of spiropyrazolones Alex Zea a, Andrea-Nekane R. Alba a, Andrea Mazzanti b, Albert Moyano a and Ramon Rios a,c * Supporting Information NMR spectra and HPLC traces
and Selective Allylic Reduction of Allylic Alcohols and Their Derivatives with Benzyl Alcohol
 FeCl 3 6H 2 O-Catalyzed Disproportionation of Allylic Alcohols and Selective Allylic Reduction of Allylic Alcohols and Their Derivatives with Benzyl Alcohol Jialiang Wang, Wen Huang, Zhengxing Zhang, Xu
FeCl 3 6H 2 O-Catalyzed Disproportionation of Allylic Alcohols and Selective Allylic Reduction of Allylic Alcohols and Their Derivatives with Benzyl Alcohol Jialiang Wang, Wen Huang, Zhengxing Zhang, Xu
phase: synthesis of biaryls, terphenyls and polyaryls
 Supporting Information for Studies on Pd/NiFe 2 O 4 catalyzed ligand-free Suzuki reaction in aqueous phase: synthesis of biaryls, terphenyls and polyaryls Sanjay R. Borhade and Suresh B. Waghmode* Address:
Supporting Information for Studies on Pd/NiFe 2 O 4 catalyzed ligand-free Suzuki reaction in aqueous phase: synthesis of biaryls, terphenyls and polyaryls Sanjay R. Borhade and Suresh B. Waghmode* Address:
Supporting information
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 Supporting information Copper-catalysed intramolecular O-arylation: a simple
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 Supporting information Copper-catalysed intramolecular O-arylation: a simple
Natural Products Research Institute, College of Pharmacy, Seoul National University, 1 Gwanak-ro, Gwanak-gu, Seoul 08826, Republic of Korea
 Supporting Information Nicrophorusamides A and B, Antibacterial Chlorinated Cyclic Peptides from a Gut Bacterium of the Carrion Beetle Nicrophorus concolor Yern-Hyerk Shin, Suhyun Bae, Jaehoon Sim,,ǁ Joonseong
Supporting Information Nicrophorusamides A and B, Antibacterial Chlorinated Cyclic Peptides from a Gut Bacterium of the Carrion Beetle Nicrophorus concolor Yern-Hyerk Shin, Suhyun Bae, Jaehoon Sim,,ǁ Joonseong
9-amino-(9-deoxy)cinchona alkaloids-derived novel chiral phase-transfer catalysts
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 9-amino-(9-deoxy)cinchona alkaloids-derived novel chiral phase-transfer
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 9-amino-(9-deoxy)cinchona alkaloids-derived novel chiral phase-transfer
Supporting Information for. Catalytic C H α-trifluoromethylation of α,β-unsaturated Carbonyl Compounds
 Supporting Information for Catalytic C H α-trifluoromethylation of α,β-unsaturated Carbonyl Compounds Zhongxue Fang, a Yongquan Ning, a Pengbing Mi, a Peiqiu Liao, a Xihe Bi* a,b a Department of Chemistry,
Supporting Information for Catalytic C H α-trifluoromethylation of α,β-unsaturated Carbonyl Compounds Zhongxue Fang, a Yongquan Ning, a Pengbing Mi, a Peiqiu Liao, a Xihe Bi* a,b a Department of Chemistry,
Supporting Information. Partial thioamide scan on the lipopeptaibiotic trichogin GA IV. Effects on
 Supporting Information for Partial thioamide scan on the lipopeptaibiotic trichogin GA IV. Effects on folding and bioactivity Marta De Zotti 1, Barbara Biondi 1, Cristina Peggion 1, Matteo De Poli 1, Haleh
Supporting Information for Partial thioamide scan on the lipopeptaibiotic trichogin GA IV. Effects on folding and bioactivity Marta De Zotti 1, Barbara Biondi 1, Cristina Peggion 1, Matteo De Poli 1, Haleh
Supporting Information. A catalyst-free multicomponent domino sequence for the. diastereoselective synthesis of (E)-3-[2-arylcarbonyl-3-
 Supporting Information for A catalyst-free multicomponent domino sequence for the diastereoselective synthesis of (E)-3-[2-arylcarbonyl-3- (arylamino)allyl]chromen-4-ones Pitchaimani Prasanna 1, Pethaiah
Supporting Information for A catalyst-free multicomponent domino sequence for the diastereoselective synthesis of (E)-3-[2-arylcarbonyl-3- (arylamino)allyl]chromen-4-ones Pitchaimani Prasanna 1, Pethaiah
Supporting Information
 Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2014 A Family of Click Nucleosides for Metal-Mediated Base Pairing: Unravelling the Principles of Highly Stabilizing Metal-Mediated
Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2014 A Family of Click Nucleosides for Metal-Mediated Base Pairing: Unravelling the Principles of Highly Stabilizing Metal-Mediated
Quantum dot sensitized solar cells with efficiency over 12% based on tetraethyl orthosilicate additive in polysulfide electrolyte
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supplementary Information (SI) Quantum dot sensitized solar cells with
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supplementary Information (SI) Quantum dot sensitized solar cells with
Synthesis and evaluation of novel aza-caged Garcinia xanthones
 Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry Synthesis and evaluation of novel aza-caged Garcinia xanthones Xiaojin Zhang, a,1 Xiang Li, a,1 Haopeng Sun, * b Zhengyu Jiang,
Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry Synthesis and evaluation of novel aza-caged Garcinia xanthones Xiaojin Zhang, a,1 Xiang Li, a,1 Haopeng Sun, * b Zhengyu Jiang,
Electronic supplementary information for
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry C. This journal is The Royal Society of Chemistry 2017 Electronic supplementary information for Highly responsive ethylenediamine
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry C. This journal is The Royal Society of Chemistry 2017 Electronic supplementary information for Highly responsive ethylenediamine
Iodine-catalyzed synthesis of sulfur-bridged enaminones and chromones via double C(sp 2 )-H thiolation
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2017 Iodine-catalyzed synthesis of sulfur-bridged enaminones and chromones via
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2017 Iodine-catalyzed synthesis of sulfur-bridged enaminones and chromones via
Supporting Information
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2018 Supporting Information Silver or Cerium-Promoted Free Radical Cascade Difunctionalization
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2018 Supporting Information Silver or Cerium-Promoted Free Radical Cascade Difunctionalization
Supporting Information
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Synthesis of 3-omosubstituted Pyrroles via Palladium- Catalyzed Intermolecular Oxidative Cyclization
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Synthesis of 3-omosubstituted Pyrroles via Palladium- Catalyzed Intermolecular Oxidative Cyclization
Electronic Supplementary Information
 Electronic Supplementary Information NbCl 3 -catalyzed [2+2+2] intermolecular cycloaddition of alkynes and alkenes to 1,3-cyclohexadiene derivatives Yasushi Obora,* Keisuke Takeshita and Yasutaka Ishii*
Electronic Supplementary Information NbCl 3 -catalyzed [2+2+2] intermolecular cycloaddition of alkynes and alkenes to 1,3-cyclohexadiene derivatives Yasushi Obora,* Keisuke Takeshita and Yasutaka Ishii*
SUPPORTING INFORMATION. Polystyrene-immobilized DABCO as a highly efficient and recyclable organocatalyst for Knoevenagel condensation
 Electronic Supplementary Material (ESI) for RSC Advances This journal is The Royal Society of Chemistry 213 SUPPORTING INFORMATION Polystyrene-immobilized DABCO as a highly efficient and recyclable organocatalyst
Electronic Supplementary Material (ESI) for RSC Advances This journal is The Royal Society of Chemistry 213 SUPPORTING INFORMATION Polystyrene-immobilized DABCO as a highly efficient and recyclable organocatalyst
Tributylphosphine-Catalyzed Cycloaddition of Aziridines with Carbon Disulfide and Isothiocyanate
 upporting Information Tributylphosphine-Catalyzed Cycloaddition of Aziridines with Carbon Disulfide and Isothiocyanate Jing-Yu Wu, Zhi-Bin Luo, Li-Xin Dai and Xue-Long Hou* a tate Key Laboratory of Organometallic
upporting Information Tributylphosphine-Catalyzed Cycloaddition of Aziridines with Carbon Disulfide and Isothiocyanate Jing-Yu Wu, Zhi-Bin Luo, Li-Xin Dai and Xue-Long Hou* a tate Key Laboratory of Organometallic
Rh(III)-Catalyzed C-H Amidation with N-hydroxycarbamates: A. new Entry to N-Carbamate Protected Arylamines
 Rh(III)-Catalyzed C-H Amidation with N-hydroxycarbamates: A new Entry to N-Carbamate Protected Arylamines Bing Zhou,* Juanjuan Du, Yaxi Yang,* Huijin Feng, Yuanchao Li Shanghai Institute of Materia Medica,
Rh(III)-Catalyzed C-H Amidation with N-hydroxycarbamates: A new Entry to N-Carbamate Protected Arylamines Bing Zhou,* Juanjuan Du, Yaxi Yang,* Huijin Feng, Yuanchao Li Shanghai Institute of Materia Medica,
Supporting Information for
 Supporting Information for Acceptorless Dehydrogenation of N-Heterocycles and Secondary Alcohols by Ru(II)-NNC Complexes Bearing a Pyrazoyl-Indolyl-Pyridine Ligand Qingfu Wang,, Huining Chai,, and Zhengkun
Supporting Information for Acceptorless Dehydrogenation of N-Heterocycles and Secondary Alcohols by Ru(II)-NNC Complexes Bearing a Pyrazoyl-Indolyl-Pyridine Ligand Qingfu Wang,, Huining Chai,, and Zhengkun
Supporting Information One-Pot Approach to Chiral Chromenes via Enantioselective Organocatalytic Domino Oxa-Michael-Aldol Reaction
 Supporting Information ne-pot Approach to Chiral Chromenes via Enantioselective rganocatalytic Domino xa-michael-aldol Reaction Hao Li, Jian Wang, Timiyin E-Nunu, Liansuo Zu, Wei Jiang, Shaohua Wei, *
Supporting Information ne-pot Approach to Chiral Chromenes via Enantioselective rganocatalytic Domino xa-michael-aldol Reaction Hao Li, Jian Wang, Timiyin E-Nunu, Liansuo Zu, Wei Jiang, Shaohua Wei, *
Electronic Supplementary Information
 Electronic Supplementary Information The preferred all-gauche conformations in 3-fluoro-1,2-propanediol Laize A. F. Andrade, a Josué M. Silla, a Claudimar J. Duarte, b Roberto Rittner, b Matheus P. Freitas*,a
Electronic Supplementary Information The preferred all-gauche conformations in 3-fluoro-1,2-propanediol Laize A. F. Andrade, a Josué M. Silla, a Claudimar J. Duarte, b Roberto Rittner, b Matheus P. Freitas*,a
Synthesis of novel 1,2,3-triazolyl derivatives of pregnane, androstane and D-homoandrostane. Tandem Click reaction/cu-catalyzed D-homo rearrangement
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 Supporting Information Synthesis of novel 1,2,3-triazolyl derivatives of
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 Supporting Information Synthesis of novel 1,2,3-triazolyl derivatives of
Supporting information
 Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 204 Supporting information Palladium nanoparticles supported on triazine functionalised mesoporous
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 204 Supporting information Palladium nanoparticles supported on triazine functionalised mesoporous
Supporting Information
 Supporting Information Ceric Ammonium Nitrate (CAN) catalyzed efficient one-pot three component aza-diels-alder reactions for a facile synthesis of tetrahydropyranoquinoline derivatives Ravinder Goud Puligoundla
Supporting Information Ceric Ammonium Nitrate (CAN) catalyzed efficient one-pot three component aza-diels-alder reactions for a facile synthesis of tetrahydropyranoquinoline derivatives Ravinder Goud Puligoundla
Supporting Information for Iron-catalyzed decarboxylative alkenylation of cycloalkanes with arylvinylic carboxylic acids via a radical process
 Supporting Information for Iron-catalyzed decarboxylative alkenylation of cycloalkanes with arylvinylic carboxylic acids via a radical process Jincan Zhao 1, Hong Fang 1, Jianlin Han* 1,2 and Yi Pan* 1
Supporting Information for Iron-catalyzed decarboxylative alkenylation of cycloalkanes with arylvinylic carboxylic acids via a radical process Jincan Zhao 1, Hong Fang 1, Jianlin Han* 1,2 and Yi Pan* 1
Available online at shd.org.rs/jscs/
 J. Serb. Chem. Soc. 78 (1) S1 S8 (2013) Supplementary material SUPPLEMENTARY MATERIAL TO Metal complexes of N'-[2-hydroxy-5-(phenyldiazenyl)- benzylidene]isonicotinohydrazide. Synthesis, spectroscopic
J. Serb. Chem. Soc. 78 (1) S1 S8 (2013) Supplementary material SUPPLEMENTARY MATERIAL TO Metal complexes of N'-[2-hydroxy-5-(phenyldiazenyl)- benzylidene]isonicotinohydrazide. Synthesis, spectroscopic
Nguyen Hien Trang* **
 152 Nippon Shokuhin Kagaku Kogaku Kaishi Vol.., No.., +,+3 (,**1) 10 Nguyen Hien Trang* ** * Department of Food Science and Technology, Hue University of Agriculture and Forestry ** Properties of "Shishibishio
152 Nippon Shokuhin Kagaku Kogaku Kaishi Vol.., No.., +,+3 (,**1) 10 Nguyen Hien Trang* ** * Department of Food Science and Technology, Hue University of Agriculture and Forestry ** Properties of "Shishibishio
Phenylpropanoids, Sesquiterpenoids and Flavonoids from Pimpinella tragium Vill. subsp. lithophila (Schischkin) Tutin
 Supporting Information Rec. Nat. Prod. 10:2 (2016) 207-213 Phenylpropanoids, Sesquiterpenoids and Flavonoids from Pimpinella tragium Vill. subsp. lithophila (Schischkin) Tutin Hilal Özbek 1, Zühal Güvenalp
Supporting Information Rec. Nat. Prod. 10:2 (2016) 207-213 Phenylpropanoids, Sesquiterpenoids and Flavonoids from Pimpinella tragium Vill. subsp. lithophila (Schischkin) Tutin Hilal Özbek 1, Zühal Güvenalp
Supplementary Information
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2014 Supplementary Information A contribution to the rational design of Ru(CO) 3 Cl 2 L complexes
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2014 Supplementary Information A contribution to the rational design of Ru(CO) 3 Cl 2 L complexes
Supporting Information
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2017 Supporting Information Phosphorescent Pt(II) complexes bearing a monoanionic C^N^N luminophore
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2017 Supporting Information Phosphorescent Pt(II) complexes bearing a monoanionic C^N^N luminophore
Supporting Information. Experimental section
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Experimental section General. Proton nuclear magnetic resonance ( 1
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Experimental section General. Proton nuclear magnetic resonance ( 1
Lyotropic Liquid Crystals in Amino acid derived Protic Ionic Liquids: Physicochemical Properties and Behaviour as Amphiphile Self-Assembly Media
 Lyotropic Liquid Crystals in Amino acid derived Protic Ionic Liquids: Physicochemical Properties and Behaviour as Amphiphile Self-Assembly Media Jiayi Wang, [a,b] Tamar L. Greaves, [b] Danielle F. Kennedy,
Lyotropic Liquid Crystals in Amino acid derived Protic Ionic Liquids: Physicochemical Properties and Behaviour as Amphiphile Self-Assembly Media Jiayi Wang, [a,b] Tamar L. Greaves, [b] Danielle F. Kennedy,
Fluorinative Ring-opening of Cyclopropanes by Hypervalent Iodine Reagents. An Efficient Method for 1,3- Oxyfluorination and 1,3-Difluorination
 Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2016 Supporting Information Fluorinative Ring-opening of Cyclopropanes by Hypervalent Iodine
Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2016 Supporting Information Fluorinative Ring-opening of Cyclopropanes by Hypervalent Iodine
Direct Transformation of Ethylarenes into Primary Aromatic Amides with N-Bromosuccinimide and I 2 -aq NH 3
 Supporting Information Direct Transformation of Ethylarenes into Primary Aromatic Amides with N-Bromosuccinimide and I 2 -aq NH 3 Shohei Shimokawa, Yuhsuke Kawagoe, Katsuhiko Moriyama, Hideo Togo* Graduate
Supporting Information Direct Transformation of Ethylarenes into Primary Aromatic Amides with N-Bromosuccinimide and I 2 -aq NH 3 Shohei Shimokawa, Yuhsuke Kawagoe, Katsuhiko Moriyama, Hideo Togo* Graduate
Supporting Information
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Supporting Information Abnormal N-Heterocyclic Carbene Based Nickel Complex for Catalytic
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Supporting Information Abnormal N-Heterocyclic Carbene Based Nickel Complex for Catalytic
Novel and Selective Palladium-Catalyzed Annulation of 2-Alkynylphenols to Form 2-Substituted 3-Halobenzo[b]furans. Supporting Information
![Novel and Selective Palladium-Catalyzed Annulation of 2-Alkynylphenols to Form 2-Substituted 3-Halobenzo[b]furans. Supporting Information Novel and Selective Palladium-Catalyzed Annulation of 2-Alkynylphenols to Form 2-Substituted 3-Halobenzo[b]furans. Supporting Information](/thumbs/64/50937700.jpg) Novel and Selective Palladium-Catalyzed Annulation of 2-Alkynylphenols to Form 2-Substituted 3-Halobenzo[b]furans Liang Yun, Shi Tang, Xu-Dong Zhang, Li-Qiu Mao, Ye-Xiang Xie and Jin-Heng Li* Key Laboratory
Novel and Selective Palladium-Catalyzed Annulation of 2-Alkynylphenols to Form 2-Substituted 3-Halobenzo[b]furans Liang Yun, Shi Tang, Xu-Dong Zhang, Li-Qiu Mao, Ye-Xiang Xie and Jin-Heng Li* Key Laboratory
LUO, Hong2Qun LIU, Shao2Pu Ξ LI, Nian2Bing
 2003 61 3, 435 439 ACTA CHIMICA SINICA Vol 61, 2003 No 3, 435 439 2 ΞΞ ( 400715), 2, 2, 2, 3/ 2 2,, 2,, Ne w Methods for the Determination of the Inclusion Constant between Procaine Hydrochloride and 2Cyclodextrin
2003 61 3, 435 439 ACTA CHIMICA SINICA Vol 61, 2003 No 3, 435 439 2 ΞΞ ( 400715), 2, 2, 2, 3/ 2 2,, 2,, Ne w Methods for the Determination of the Inclusion Constant between Procaine Hydrochloride and 2Cyclodextrin
Electronic Supplementary Information
 Electronic Supplementary Information Regenerative Labeling of Saccharides Hao-Yu Wen, Peng-Hao Hsu, Guei-San Chen and Jim-Min Fang,, * Department of Chemistry, National Taiwan University, Taipei 106, Taiwan
Electronic Supplementary Information Regenerative Labeling of Saccharides Hao-Yu Wen, Peng-Hao Hsu, Guei-San Chen and Jim-Min Fang,, * Department of Chemistry, National Taiwan University, Taipei 106, Taiwan
Heavier chalcogenone complexes of bismuth(iii)trihalides: Potential catalysts for acylative cleavage of cyclic ethers. Supporting Information
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Heavier chalcogenone complexes of bismuth(iii)trihalides: Potential catalysts for acylative
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Heavier chalcogenone complexes of bismuth(iii)trihalides: Potential catalysts for acylative
Supporting Information
 Supporting Information Montmorillonite KSF-Catalyzed One-pot, Three-component, Aza-Diels- Alder Reactions of Methylenecyclopropanes With Arylaldehydes and Aromatic Amines Li-Xiong Shao and Min Shi* General
Supporting Information Montmorillonite KSF-Catalyzed One-pot, Three-component, Aza-Diels- Alder Reactions of Methylenecyclopropanes With Arylaldehydes and Aromatic Amines Li-Xiong Shao and Min Shi* General
Electronic Supplementary Information (ESI)
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information (ESI) Cyclopentadienyl iron dicarbonyl (CpFe(CO) 2 ) derivatives
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information (ESI) Cyclopentadienyl iron dicarbonyl (CpFe(CO) 2 ) derivatives
Available online at shd.org.rs/jscs/
 J. Serb. Chem. Soc. 78 (12) S131 S136 (2013) Supplementary material SUPPLEMENTARY MATERIAL TO New 9-aminoacridine derivatives as inhibitors of botulinum neurotoxins and P. falciparum malaria MIKLOŠ TOT
J. Serb. Chem. Soc. 78 (12) S131 S136 (2013) Supplementary material SUPPLEMENTARY MATERIAL TO New 9-aminoacridine derivatives as inhibitors of botulinum neurotoxins and P. falciparum malaria MIKLOŠ TOT
Synthesis and Biological Evaluation of Novel Acyclic and Cyclic Glyoxamide derivatives as Bacterial Quorum Sensing and Biofilm Inhibitors
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2017 Synthesis and Biological Evaluation of Novel Acyclic and Cyclic Glyoxamide
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2017 Synthesis and Biological Evaluation of Novel Acyclic and Cyclic Glyoxamide
A strategy for the identification of combinatorial bioactive compounds. contributing to the holistic effect of herbal medicines
 1 2 Supplementary information 3 4 A strategy for the identification of combinatorial bioactive compounds contributing to the holistic effect of herbal medicines 5 6 Fang Long 1, Hua Yang 1, Yanmin Xu,
1 2 Supplementary information 3 4 A strategy for the identification of combinatorial bioactive compounds contributing to the holistic effect of herbal medicines 5 6 Fang Long 1, Hua Yang 1, Yanmin Xu,
Structure-Metabolism-Relationships in the microsomal clearance of. piperazin-1-ylpyridazines
 Electronic Supplementary Material (ESI) for MedChemComm. This journal is The Royal Society of Chemistry 2017 Structure-Metabolism-Relationships in the microsomal clearance of piperazin-1-ylpyridazines
Electronic Supplementary Material (ESI) for MedChemComm. This journal is The Royal Society of Chemistry 2017 Structure-Metabolism-Relationships in the microsomal clearance of piperazin-1-ylpyridazines
Cyclic Cystine-Bridged Peptides from the Marine. Sponge Clathria basilana Induce Apoptosis in. Tumor Cells and Depolarize the Bacterial
 Supporting information Cyclic Cystine-Bridged Peptides from the Marine Sponge Clathria basilana Induce Apoptosis in Tumor Cells and Depolarize the Bacterial Cytoplasmic Membrane Amin Mokhlesi,, Fabian
Supporting information Cyclic Cystine-Bridged Peptides from the Marine Sponge Clathria basilana Induce Apoptosis in Tumor Cells and Depolarize the Bacterial Cytoplasmic Membrane Amin Mokhlesi,, Fabian
Design and Solid Phase Synthesis of New DOTA Conjugated (+)-Biotin Dimers Planned to Develop Molecular Weight-Tuned Avidin Oligomers
 Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2015 Supplementary Information Design and Solid Phase Synthesis of New DTA Conjugated
Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2015 Supplementary Information Design and Solid Phase Synthesis of New DTA Conjugated
Supporting Information. for. A novel application of 2-silylated 1,3-dithiolanes for the. synthesis of aryl/hetaryl-substituted ethenes and
 Supporting Information for A novel application of 2-silylated 1,3-dithiolanes for the synthesis of aryl/hetaryl-substituted ethenes and dibenzofulvenes Grzegorz Mlostoń *1, Paulina Pipiak 1, Róża Hamera-Fałdyga
Supporting Information for A novel application of 2-silylated 1,3-dithiolanes for the synthesis of aryl/hetaryl-substituted ethenes and dibenzofulvenes Grzegorz Mlostoń *1, Paulina Pipiak 1, Róża Hamera-Fałdyga
SUPPORTING INFORMATION
 SUPPRTING INFRMATIN Per(-guanidino--deoxy)cyclodextrins: Synthesis, characterisation and binding behaviour toward selected small molecules and DNA. Nikolaos Mourtzis, a Kyriaki Eliadou, a Chrysie Aggelidou,
SUPPRTING INFRMATIN Per(-guanidino--deoxy)cyclodextrins: Synthesis, characterisation and binding behaviour toward selected small molecules and DNA. Nikolaos Mourtzis, a Kyriaki Eliadou, a Chrysie Aggelidou,
