Supporting Information. for
|
|
|
- Φιλύρη Ζωγράφου
- 7 χρόνια πριν
- Προβολές:
Transcript
1 Supporting Information for A general synthetic route to [Cu(X)(NHC)] (NHC = N- heterocyclic carbene, X =Cl, Br, I) complexes Orlando Santoro, Alba Collado, Alexandra M. Z. Slawin, Steven P. Nolan and Catherine S. J. Cazin* EaStCHEM School of Chemistry, University of St Andrews, St Andrews, KY16 9ST, UK. cc111@st-andrews.ac.uk S1
2 Table of Contents General considerations... S3 Synthesis of the [Cu(X)(NHC)] complexes... S3 Screening of the reaction conditions... S3 General Procedure... S3 Small scale:... S3 Large scale... S4 Synthesis of [Cu(Cl)(IPr)] (2a)... S4 Synthesis of [Cu(Cl)(SIPr)] (2b)... S5 Synthesis of [Cu(Cl)(IMes)] (2c)... S5 Synthesis of [Cu(Cl)(SIMes)] (2d)... S6 Synthesis of [Cu(Cl)(IPr*)] (2e)... S6 Synthesis of [Cu(Cl)(I t Bu)] (2f)... S7 Synthesis of [Cu(Cl)(ICy)] (2g)... S7 Synthesis of [Cu(Cl)(SICy)] (2h)... S8 Synthesis of [Cu(Br)(IPr)] (2i)... S9 Synthesis of [Cu(I)(IPr)] (2j)... S9 Synthesis of the [(NHC)H][CuXY] salts (3a-3k)... S10 General Procedure... S10 Synthesis of [IPrH][CuCl 2 ] (3a)... S10 Synthesis of [SIPrH][CuCl 2 ] (3b)... S11 Synthesis of [IMesH][CuCl 2 ] (3c)... S12 Synthesis of [SIMesH][CuCl 2 ] (3d)... S12 Synthesis of [ICyH][CuCl 2 ] (3g)... S13 Synthesis of [IPrH][CuClBr] (3i)... S14 Synthesis of [IPrH][CuClI] (3j)... S14 Synthesis of [IPrH][CuBrI] (3k)... S15 1 H and 13 C-{1H} NMR spectra... S16 Crystal data and Structure refinement... S34 S2
3 General considerations All reactions were carried under air and technical grade solvent were used unless otherwise stated. K 2 CO 3 and KHCO 3 were used as received without further purification. 1 H, and 13 C- { 1 H} Nuclear Magnetic Resonance (NMR) spectra were recorded on a Bruker ADVANCE 300 MHz and Bruker ADVANCE 400 MHz spectrometer using the residual solvent peak as reference (CHCl 3 : δ H = 7.26 ppm, δ C = ppm, CH 2 Cl 2 : δ H = 5.32 ppm, δ C = ppm) at 298K. Elemental analyses were performed at London Metropolitan University , Holloway Road, London, N7 8DB. Synthesis of the [Cu(X)(NHC)] complexes Screening of the reaction conditions Entry Base (equiv.) Solvent T ( C) Time (h) Conversion (%) a 1 K 2 CO 3 (1.5) acetone rt K 2 CO 3 (2.0) acetone rt K 2 CO 3 (1.0) acetone K 2 CO 3 (2.0) acetone >99 5 K 2 CO 3 (10) acetone 60 1 >99 6 KHCO 3 (2.0) acetone K 2 CO 3 (2.0) THF a Conversion determined by 1 H NMR analysis. General Procedure Small scale: A vial was charged with NHC. HCl (1.0 equiv.), CuX (1.0 equiv.) and K 2 CO 3 (2.0 equiv.). The mixture was dissolved in acetone (1.0 ml) and stirred at 60 C for 24 hours. The solution was then filtered through silica which was washed with dichloromethane (3 x 1.0 ml). The solvent was concentrated under vacuum and pentane (3.0 ml) was added thereby precipitating the desired product that was washed with further portions of pentane (3 x 1.0 ml) and dried under vacuum. S3
4 Large scale: a round bottom flask equipped with a condenser was charged with IPr. HCl (1.0 equiv.), CuX (X = Cl, Br, I, 1.0 equiv.) and K 2 CO 3 (3.0 equiv.). The mixture was dissolved in acetone and stirred for 8-15 h at 60 C. The same work-up as the small scale procedure was carried out. Synthesis of [Cu(Cl)(IPr)] (2a) Reaction between IPr. HCl (1a) (100 mg, 0.23 mmol), CuCl (23 mg, 0.23 mmol) and K 2 CO 3 (66 mg, 0.46 mmol) led to the isolation of 2a as a white solid in 92% isolated yield (103 mg, 0.21 mmol). 1 H NMR (400 MHz, CDCl 3, 298 K): δ = 1.23 (d, 3 J H-H = 6.9 Hz, 12H, CH-CH 3 ), 1.30 (d, 3 J H- H = 6.9 Hz, 12H, CH-CH 3 ), 2.56 (sept, 3 J H-H = 6.9 Hz, 4H, CH-CH 3 ), 7.13 (s, 2H, H 4 and H 5 ), 7.29 (d, 3 J H-H = 7.8 Hz, 4H, CH phenyl), 7.49 (t, 3 J H-H = 7.8 Hz, 2H, CH phenyl). 13 C-{ 1 H} NMR (75 MHz, CDCl 3, 298 K): δ = 24.0 (s, CH-CH 3 ), 24.9 (s, CH-CH 3 ), 28.8 (s, CH-CH 3 ), (s, C IV Ar), (s, CH Ar), (s, C 4 and C 5 ), (s, C IV Ar), (s, CH Ar), (s, C 2 ). Elem. Anal. Calcd. for C 27 H 37 ClCuN 2 : C, 66.37; H, 7.63, N, Found: C, 66.62, H, 7.31, N, 5.84 S4
5 Synthesis of [Cu(Cl)(SIPr)] (2b) Reaction between SIPr. HCl (1b) (100 mg, 0.23 mmol), CuCl (23 mg, 0.23 mmol) and K 2 CO 3 (66 mg, 0.46 mmol) led to the isolation of 2b as a white solid in 84% isolated yield (94 mg, 0.19 mmol). 1 H NMR (400 MHz, CDCl 3, 298 K): δ = 1.34 (d, 3 J H-H = 7.0 Hz, 12H, CH-CH 3 ), 1.36 (d, 3 J H- H = 7.0 Hz, 12H, CH-CH 3 ), 3.06 (sept, 3 J H-H = 6.9 Hz, 4H, CH-CH 3 ), 4.01 (s, 4H, H 4 and H 5 ), 7.24 (d, 3 J H-H = 7.8 Hz, 4H, CH phenyl), 7.39 (t, 3 J H-H = 7.8 Hz, 2H, CH phenyl). 13 C-{ 1 H} NMR (75 MHz, CDCl 3, 298 K): δ = 24.0 (s, CH-CH 3 ), 25.6 (s, CH-CH 3 ), 29.0 (s, CH-CH 3 ), 53.8 (s, C 4 and C 5 ), (s, CH Ar), (s, C IV CH Ar), (s, C IV Ar), (s, CH Ar), (s, C 2 ). Synthesis of [Cu(Cl)(IMes)] (2c) Reaction between IMes. HCl (1c) (100 mg, 0.29 mmol), CuCl (29 mg, 0.29 mmol) and K 2 CO 3 (80 mg, 0.58 mmol) led to the isolation of 2c as a white solid in 76% isolated yield (89 mg, 0.22 mmol). 1 H NMR (400 MHz, CDCl 3, 298 K): δ = 2.10 (s, 12H, CH 3 ), 2.34 (s, 6H, CH 3 ), 6.99 (s, 4H, CH phenyl), 7.05 (s, 2H, H 4 and H 5 ). 13 C-{ 1 H} NMR (75 MHz, CDCl 3, 298 K): δ = 17.9 (s, CH 3 ), 21.2 (s, CH 3 ), (s, C IV Ar), (s, CH Ar), (s, C 4 and C 5 ), (s, C IV Ar), (s, C IV Ar), (s, C 2 ). S5
6 Synthesis of [Cu(Cl)(SIMes)] (2d) Reaction between SIMes. HCl (1d) (100 mg, 0.29 mmol), CuCl (29 mg, 0.29 mmol) and K 2 CO 3 (80 mg, 0.58 mmol) led to the isolation of 2d as a white solid in 94% isolated yield (110 mg, 0.27 mmol). 1 H NMR (400 MHz, CDCl 3, 298 K): δ = 2.29 (s, 6H, CH 3 ), 2.30 (s, 12H, CH 3 ), 3.93 (s, 2H, H 4 and H 5 ), 6.93 (s, 4H, CH phenyl). 13 C-{ 1 H} NMR (75 MHz, CDCl 3, 298 K): δ = 18.1 (s, CH 3 ), 21.1 (s, CH 3 ), 51.0 (s, C 4 and C 5 ), (s, CH Ar), (s, C IV Ar), (s, C IV Ar), (s, C IV Ar), (s, C 2 ). Synthesis of [Cu(Cl)(IPr*)] (2e) Reaction between IPr*. HCl (1e) (100 mg, 0.10 mmol), CuCl (9.90 mg, 0.10 mmol) and K 2 CO 3 (27.6 mg, 0.20 mmol) led to the isolation of 2e as a white solid in 70% isolated yield (71 mg, 0.07 mmol). 1 H NMR (400 MHz, CDCl 3, 298 K): δ = 2.23 (s, 6H, CH 3 ), 5.21 (s, 4H, CHPh 2 ), 5.83 (s, 2H, H 4 and H 5 ), 6.86 (s, 4H, CH phenyl), (m, 8H, CH phenyl), 7.03 (m, 8H, CH aryl), (m, 24H, CH aryl). 13 C-{ 1 H} NMR (75 MHz, CDCl 3, 298 K): δ = 21.9 (s, CH 3 ), 51.3 (s, CHPh 2 ), (s, C 4 and C 5 ), (s, CH Ar), (s, CH Ar), (s, CH Ar), (s, CH Ar), (s, CH Ar), (s, CH Ar), (s, CH Ar), (s, C IV Ar), (s, C IV Ar), (s, C IV Ar), (s, C IV Ar), (s, C IV Ar), (s, C 2 ). S6
7 Synthesis of [Cu(Cl)(I t Bu)] (2f) Reaction (carried out under Ar atmosphere) between I t Bu. HCl (1f) (100 mg, 0.37 mmol), CuCl (36.6 mg, 0.37 mmol) and K 2 CO 3 (99.4 mg, 0.74 mmol) led to the isolation of 2f as a white solid in 55% isolated yield (47 mg, 0.21 mmol). 1 H NMR (400 MHz, CDCl 3, 298 K): δ = 1.75 (s, 18H, CH 3 ), 7.03 (s, 2H, H 4 and H 5 ). 13 C-{ 1 H} NMR (75 MHz, CDCl 3, 298 K): δ = 32.1 (s, CH 3 ), (s, C 4 and C 5 ), (s, C 2 ). Synthesis of [Cu(Cl)(ICy)] (2g) Reaction between ICy. HCl (1g) (100 mg, 0.37 mmol), CuCl (36.7 mg, 0.37 mmol) and K 2 CO 3 (99.4 mg, 0.74 mmol) led to the isolation of 2g as a white solid in 80% isolated yield (98 mg, 0.30 mmol). 1 H NMR (400 MHz, CDCl 3, 298 K): δ = 1.20 (m, 2H, CH 2 ), 1.40 (m, 4H, CH 2 ), (m, 6H, CH 2 ), 1.84 (m, 4H, CH 2 ), 2.00 (m, 4H, CH 2 ), 4.24 (m, 2H, CH), 6.91 (s, 2H, H 4 and H 5 ). 13 C-{ 1 H} NMR (75 MHz, CDCl 3, 298 K): δ = 25.0 (s, CH 2 ), 25.4 (s, CH 2 ), 34.7 (s, CH 2 ), 61.1 (s, CH), (s, C 4 and C 5 ), (s, C 2 ). S7
8 Synthesis of [Cu(Cl)(SICy)] (2h) Under Ar, a vial was charged with SICy. HCl (1h) (100 mg, 0.37 mmol, 1.0 equiv.), CuCl (36.6 mg, 0.37 mmol, 1.0 equiv.) and K 2 CO 3 (99.4 mg, 0.74 mmol, 2.0 equiv.) The mixture was dissolved in dry dichloromethane (1.0 ml) and stirred at 60 C for 24 hours. The solution was then filtered through silica which was washed with dichloromethane (3 x 1 ml). Evaporation under reduced pressure of the volatiles led to the isolation of 2h as a white solid in 49% isolated yield (60 mg, 0.19 mmol). 1 H NMR (400 MHz, CDCl 3, 298 K): δ = 1.04 (m, 2H, CH 2 ), 1.29 (m, 4H, CH 2 ), 1.43 (m, 4H, CH 2 ), 1.61 (m, 2H, CH 2 ), 1.76 (m, 8H, CH 2 ), 3.45 (s, 3 J H-H = 12.0 Hz, 4H, H 4 and H 5 ), 3.77 (m, 2H, CH). 13 C-{ 1 H} NMR (75 MHz, CDCl 3, 298 K): δ = 25.2 (s, CH 2 ), 25.3 (s, CH 2 ), 31.9 (s, CH 2 ), 44.2 (s, C 4 and C 5 ), 59.6 (s, CH), (s, C 2 ). S8
9 Synthesis of [Cu(Br)(IPr)] (2i) Reaction between IPr. HCl (1a) (100 mg, 0.23 mmol), CuBr (33 mg, 0.23 mmol) and K 2 CO 3 (66 mg, 0.46 mmol) led to the isolation of 2i as a white solid in 88% isolated yield (106 mg, 0.20 mmol). 1 H NMR (400 MHz, CDCl 3, 298 K): δ = 1.22 (d, 3 J H-H = 6.9 Hz, 12H, CH-CH 3 ), 1.30 (d, 3 J H- H = 6.9 Hz, 12H, CH-CH 3 ), 2.56 (sept, 3 J H-H = 6.9 Hz, 4H, CH-CH 3 ), 7.14 (s, 2H, H 4 and H 5 ), 7.29 (d, 3 J H-H = 7.8 Hz, 4H, CH phenyl), 7.48 (t, 3 J H-H = 7.8 Hz, 2H, CH phenyl). 13 C-{ 1 H} NMR (75 MHz, CDCl 3, 298 K): δ = 24.0 (s, CH-CH 3 ), 24.9 (s, CH-CH 3 ), 28.8 (s,ch-ch 3 ), (s, C IV Ar), (s, CH Ar), (s, C 4 and C 5 ), (s, C IV Ar), (s, CH Ar), (s, C 2 ). Synthesis of [Cu(I)(IPr)] (2j) Reaction between IPr. HCl (1a) (100 mg, 0.23 mmol), CuI (44 mg, 0.23 mmol) and K 2 CO 3 (66 mg, 0.46 mmol) led to the isolation of 2j as a white solid in 77% isolated yield (104 mg, 0.18 mmol). 1 H NMR (400 MHz, CDCl 3, 298 K): δ = 1.23 (d, 3 J H-H = 6.9 Hz, 12H, CH-CH 3 ), 1.30 (d, 3 J H- H = 6.9 Hz, 12H, CH-CH 3 ), 2.57 (sept, 3 J H-H = 6.9 Hz, 4H, CH-CH 3 ), 7.14 (s, 2H, H 4 and H 5 ), 7.30 (d, 3 J H-H = 7.8 Hz, 4H, CH phenyl), 7.49 (t, 3 J H-H = 7.8 Hz, 2H, CH phenyl). S9
10 13 C-{ 1 H} NMR (75 MHz, CDCl 3, 298 K): δ = 24.0 (s, CH-CH 3 ), 25.0 (s, CH-CH 3 ), 28.8 (s, CH-CH 3 ), (s, C IV Ar), (s, CH Ar), (s, C 4 and C 5 ), (s, C IV Ar), (s, CH Ar), (s, C 2 ). Synthesis of the [(NHC)H][CuXY] salts (3a-3k) General Procedure NHC HX and CuY (X, Y = Cl, Br, I) were charged in a vial equipped with a magnetic stirring bar. The final solids mixture was dissolved in acetone (3.0 ml) and stirred for 10 minutes at room temperature. After this time the solvent was removed under reduced pressure affording the product. Synthesis of [IPrH][CuCl 2 ] (3a) Reaction between IPr HCl (1a) (300 mg, 0.70 mmol) and CuCl (69 mg, 0.70 mmol) led to the isolation of 3a as a white solid in 95% isolated yield (350 mg, 0.66 mmol). 1 H NMR (400 MHz, CD 2 Cl 2, 298 K): δ = 1.22 (d, 3 J H-H = 7.1 Hz, 12H, CH-CH 3 ), 1.28 (d, 3 J H-H = 7.1 Hz, 12H, CH-CH 3 ), 2.39 (sept, 3 J H-H = 7.1 Hz, 4H, CH-CH 3 ), 7.39 (d, 3 J H-H = 7.8 Hz, 4H, CH phenyl), 7.62 (t, 3 J H-H = 7.8 Hz, 2H, CH phenyl), 7.79 (s, 2H, H 4 and H 5 ) (s, 1H, H 2 ). 13 C-{ 1 H} NMR (75 MHz, CD 2 Cl 2, 298 K): δ = 23.8 (s, CH-CH 3 ), 24.7 (s, CH-CH 3 ), 29.5 (s, CH-CH 3 ), (s, CH Ar), (s, CH Ar), (s, C IV Ar), (s, C 4 and C 5 ), (s, C 2 ), (s, C IV Ar). S10
11 Elem. Anal.: Calcd. for C 27 H 37 Cl 2 CuN 2 : C, 61.88; H, 7.12, N, Found: C, 61.96, H, 7.15, N, 5.41 Synthesis of [SIPrH][CuCl 2 ] (3b) Reaction between SIPr. HCl (1b) (100 mg, 0.23 mmol), CuCl (23 mg, 0.23 mmol) led to the isolation of 3b as a white solid in 87% isolated yield (109 mg, 0.20 mmol). Crystals suitable for X-ray diffraction were grown by slow diffusion of pentane in a saturated solution of 3b in dichloromethane. 1 H NMR (400 MHz, CD 2 Cl 2, 298 K): δ = 1.26 (d, 3 J H-H = 6.9 Hz, 12H, CH-CH 3 ), 1.39 (d, 3 J H-H = 6.9 Hz, 12H, CH-CH 3 ), 3.00 (sept, 3 J H-H = 7.0 Hz, 4H, CH-CH 3 ), 4.59 (s, 4H, H 4 and H 5 ), 7.32 (d, 3 J H-H = 7.9 Hz, 4H, CH phenyl), 7.52 (t, 3 J H-H = 7.9 Hz, 2H, CH phenyl), 8.63 (s, 1H, H 2 ). 13 C-{ 1 H} NMR (75 MHz, CD 2 Cl 2, 298 K): δ = 24.1 (s, CH-CH 3 ), 25.5 (s, CH-CH 3 ), 29.6 (s, CH-CH 3 ), 54.6 (s, C 4 and C 5 ), (s, CH Ar), (s, C IV CH Ar), (s, C IV Ar), (s, CH Ar), (s, C 2 ). Elem. Anal.: Calcd. for C 27 H 39 Cl 2 CuN 2 : C, 61.65; H, 7.47, N, Found: C, 61.75, H, 7.57, N, 5.40 S11
12 Synthesis of [IMesH][CuCl 2 ] (3c) Reaction between IMes. HCl (1c) (100 mg, 0.29 mmol), CuCl (29 mg, 0.29 mmol) led to the isolation of 3c as a white solid in 95% isolated yield (112 mg, 0.28 mmol). Crystals suitable for X-ray diffraction were grown by slow diffusion of pentane in a saturated solution of 3c in dichloromethane. 1 H NMR (400 MHz, CD 2 Cl 2, 298 K): δ = 2.16 (s, 12H, CH 3 ), 2.39 (s, 6H, CH 3 ), 7.11 (s, 4H, CH phenyl), 7.65 (s, 2H, H 4 and H 5 ), 9.55 (s, 1H, H 2 ). 13 C-{ 1 H} NMR (75 MHz, CD2Cl2, 298 K): δ = 17.8 (s, CH 3 ), 21.3 (s, CH 3 ), (s, C 4 and C 5 ), (s, CH Ar), (s, C IV Ar), (s, C 2 ), (s, C IV Ar), (s, C IV Ar). Elem. Anal.: Calcd. for C 21 H 25 Cl 2 CuN 2 : C, 57.34; H, 5.73, N, Found: C, 57.44, H, 5.82, N, 6.46 Synthesis of [SIMesH][CuCl 2 ] (3d) Reaction between SIMes. HCl (100 mg, 0.29 mmol), CuCl (29 mg, 0.29 mmol) led to the isolation of 3d as a white solid in 94% isolated yield (110 mg, 0.27 mmol). Crystals suitable for X-ray diffraction were grown by slow diffusion of pentane in a saturated solution of 3d in dichloromethane. 1 H NMR (400 MHz, CD 2 Cl 2, 298 K): δ = 2.33 (s, 6H, CH 3 ), 2.39 (s, 12H, CH 3 ), 4.53 (s, 4H, H 4 and H 5 ), 7.04 (s, 4H, CH phenyl), 8.47 (s, 1H, H 2 ). S12
13 13 C-{ 1 H} NMR (75 MHz, CD 2 Cl 2, 298 K): δ = 18.3 (s, CH 3 ), 21.2 (s, CH 3 ), 52.0 (s, C 4 and C 5 ), (s, C IV Ar), (s, CH Ar), (s, C IV Ar), (s, C IV Ar), (s, C 2 ). Elem. Anal.: Calcd. for C 21 H 27 Cl 2 CuN 2 : C, 57.08; H, 6.16, N, Found: C, 57.16, H, 6.08, N, 6.40 Synthesis of [ICyH][CuCl 2 ] (3g) Reaction between ICy. HCl (1g) (100 mg, 0.37 mmol), CuCl (36.7 mg, 0.37 mmol) led to the isolation of 3g as a white solid in 95% isolated yield (130 mg, 0.35 mmol). Crystals suitable for X-ray diffraction were grown by slow diffusion of pentane in a saturated solution of 3g in dichloromethane. 1 H NMR (400 MHz, CDCl 3, 298 K): δ = 1.29 (m, 2H, CH 2 ), 1.53 (m, 4H, CH 2 ), (m, 8H, CH 2 ), 1.94 (m, 4H, CH 2 ), 2.27 (m, 4H, CH 2 ), 4.52 (m, 2H, CH), 7.30 (s, 2H, H 4 and H 5 ), (s, 1H, H 2 ). 13 C-{ 1 H} NMR (75 MHz, CDCl 3, 298 K): δ = 24.6 (s, CH 2 ), 24.9 (s, CH 2 ), 33.6 (s, CH 2 ), 60.2 (s, CH), (s, C 4 and C 5 ), (s, C 2 ). Elem. Anal.: Calcd. for C 15 H 25 Cl 2 CuN 2: C, 48.98; H, 6.85, N, Found: C, 48.97, H, 6.73, N, S13
14 Synthesis of [IPrH][CuClBr] (3i) Reaction between IPr HCl (300 mg, 0.70 mmol) and CuBr (100 mg, 0.70 mmol) led to the isolation of 3i as a white solid in 93% isolated yield (339 mg, 0.65 mmol). 1 H NMR (400 MHz, CD 2 Cl 2, 298 K): δ = 1.24 (d, 3 J H-H = 7.1 Hz, 12H, CH-CH 3 ), 1.30 (d, 3 J H-H = 7.1 Hz, 12H, CH-CH 3 ), 2.40 (sept, 3 J H-H = 7.1 Hz, 4H, CH-CH 3 ), 7.41 (d, 3 J H-H = 7.8 Hz, 4H, CH phenyl), 7.65 (t, 3 J H-H = 7.8 Hz, 2H, CH phenyl), 7.80 (s, 2H, H 4 and H 5 ), 9.16 (s, 1H, H 2 ). 13 C-{ 1 H} NMR (75 MHz, CD 2 Cl 2, 298 K): δ = 24.0 (s, CH-CH 3 ), 24.8 (s, CH-CH 3 ), 29.5 (s, CH-CH 3 ), (s, CH Ar), (s, CH Ar), (s, C IV Ar), (s, C 4 and C 5 ), (s, C 2 ),145.3 (s, C IV Ar). Elem. Anal.: Calcd. for C 27 H 37 BrClCuN 2: C, 57.04; H, 6.56, N, Found: C, 56.91, H, 6.63, N, 5.01 Synthesis of [IPrH][CuClI] (3j) Reaction between IPr HCl (300 mg, 0.70 mmol) and CuI (130 mg, 0.70 mmol) led to the isolation of 3j as a white solid in 95% isolated yield (400 mg, 0.66 mmol). 1 H NMR (400 MHz, CD 2 Cl 2, 298 K): δ = 1.23 (d, 3 J H-H = 7.1 Hz, 12H, CH-CH 3 ), 1.30 (d, 3 J H-H = 7.1 Hz, 12H, CH-CH 3 ), 2.41 (sept, 3 J H-H = 7.1 Hz, 4H, CH-CH 3 isopropyl), 7.42 (d, 3 J H-H = 7.8 Hz, 4H, CH phenyl), 7.63 (t, 3 J H-H = 7.8 Hz, 2H, CH phenyl), 7.84 (d, 2H, H 4 and H 5 ), 9.10 (t, 1H, H 2 ). S14
15 13 C-{ 1 H} NMR (75 MHz, CD 2 Cl 2, 298 K): δ = 24.0 (s, CH-CH 3 ), 24.8 (s, CH-CH 3 ), 29.5 (s, CH-CH 3 ), (s, CH Ar), (s, CH Ar), (s, C IV Ar), (s, C 4 and C 5 ), (s, C 2 ), (s, C IV Ar). Elem. Anal.: Calcd. for C 27 H 37 ClCuIN 2: C, 52.69; H, 6.06, N, Found: C, 52.78, H, 6.07, N, 4.61 Synthesis of [IPrH][CuBrI] (3k) Reaction between IPr HBr (300 mg, 0.70 mmol) and CuI (120 mg, 0.70 mmol) led to the isolation of 3k as a white solid in 94% isolated yield (410 mg, 0.66 mmol). 1 H NMR (400 MHz, CD 2 Cl 2, 298 K): δ = 1.24 (d, 3 J H-H = 7.1 Hz, 12H, CH-CH 3 ), 1.32 (d, 3 J H-H = 7.1 Hz, 12H, CH-CH 3 ), 2.41 (sept, 3 J H-H = 7.1 Hz, 4H, CH-CH 3 ), 7.44 (d, 3 J H-H = 7.8 Hz, 4H, CH phenyl), 7.67 (t, 3 J H-H = 7.8 Hz, 2H, CH phenyl), 7.83 (s, 2H, H 4 and H 5 ), 8.87 (s, 1H, H 2 ). 13 C-{ 1 H} NMR (75 MHz, CD 2 Cl 2, 298 K): δ = 24.0 (s, CH-CH 3 ), 24.8 (s, CH-CH 3 ), 29.5 (s, CH-CH 3 ), (s, CH Ar), (s, CH Ar), (s, C IV Ar), (s, C 4 and C 5 ), (s, C 2,145.3 (s, C IV Ar). Elem. Anal.: Calcd. for C 27 H 37 BrCuIN 2: C, 49.14; H, 5.65, N, Found: C, 49.04, H, 5.59, N, 4.27 S15
16 1 H and 13 C-{1H} NMR spectra [Cu(Cl)(IPr)] 2a, 1 H NMR, CDCl 3, 298K and 13 C-{ 1 H} NMR, CDCl 3, 298 K ppm ppm S16
17 [Cu(Cl)(SIPr)] 2b, 1 H NMR, CDCl 3, 298K and 13 C-{ 1 H} NMR, CDCl 3, 298 K ppm ppm S17
18 [Cu(Cl)(IMes)] 2c, 1 H NMR, CDCl 3, 298K and 13 C-{ 1 H} NMR, CDCl 3, 298 K ppm ppm S18
19 [Cu(Cl)(SIMes)] 2d, 1 H NMR, CDCl 3, 298K and 13 C-{ 1 H} NMR, CDCl 3, 298 K ppm ppm S19
20 [Cu(Cl)(IPr*)] 2e, 1 H NMR, CDCl 3, 298K and 13 C-{ 1 H} NMR, CDCl 3, 298 K ppm ppm S20
21 [Cu(Cl)(I t Bu)] 2f, 1 H NMR, CDCl 3, 298K and 13 C-{ 1 H} NMR, CDCl 3, 298 K ppm ppm S21
22 [Cu(Cl)(ICy)] 2g, 1 H NMR, CDCl 3, 298K and 13 C-{ 1 H} NMR, CDCl 3, 298 K ppm ppm S22
23 [Cu(Cl)(SICy)] 2h, 1 H NMR, CDCl 3, 298K and 13 C-{ 1 H} NMR, CDCl 3, 298 K ppm ppm S23
24 [Cu(Br)(IPr)] 2i, 1 H NMR, CDCl 3, 298K.and 13 C-{ 1 H} NMR, CDCl 3, 298 K ppm ppm S24
25 [Cu(I)(IPr)] 2j, 1 H NMR, CDCl 3, 298K.and 13 C-{ 1 H} NMR, CDCl 3, 298 K ppm ppm S25
26 [IPrH][CuCl 2 ] 3a, 1 H NMR, CD 2 Cl 2, 298K.and 13 C-{ 1 H} NMR, CD 2 Cl 2, 298 K ppm ppm S26
27 [SIPrH][CuCl 2 ] 3b, 1 H NMR, CD 2 Cl 2, 298K.and 13 C-{ 1 H} NMR, CD 2 Cl 2, 298 K ppm ppm S27
28 [IMesH][CuCl 2 ] 3c, 1 H NMR, CD 2 Cl 2, 298K.and 13 C-{ 1 H} NMR, CD 2 Cl 2, 298 K ppm ppm S28
29 [SIMesH][CuCl 2 ] 3d, 1 H NMR, CD 2 Cl 2, 298K.and 13 C-{ 1 H} NMR, CD 2 Cl 2, 298 K ppm ppm S29
30 [ICyH][CuCl 2 ] 3g, 1 H NMR, CDCl 3, 298K.and 13 C-{ 1 H} NMR, CDCl 3, 298 K ppm ppm S30
31 [IPrH][CuClBr] 3i, 1 H NMR, CD 2 Cl 2, 298K.and 13 C-{ 1 H} NMR, CD 2 Cl 2, 298 K ppm ppm S31
32 [IPrH][CuClI] 3j, 1 H NMR, CD 2 Cl 2, 298K.and 13 C-{ 1 H} NMR, CD 2 Cl 2, 298 K ppm ppm S32
33 [IPrH][CuBrI] 3k, 1 H NMR, CD 2 Cl 2, 298K.and 13 C-{ 1 H} NMR, CD 2 Cl 2, 298 K ppm ppm S33
34 Crystal data and Structure refinement [SIPrH][CuCl 2 ]/CCDC (3b) [IMesH][CuCl 2 ]/CCDC (3c) S34
35 [SIMes][CuCl 2 ]/CCDC (3d) [SIMes][CuCl 2 ]/CCDC (3g) S35
36 CCDC/ b CCDC/ c CCDC/ d CCDC g Empirical formula C 27 H 39 Cl 2 CuN 2 C 21 H 25 Cl 2 CuN 2 C 21 H 27 Cl 2 CuN 2 C 15 H 25 Cl 2 CuN 2 Formula weight Temperature (K) Wavelength (Å) Crystal system orthorhombic monoclinic monoclinic monoclinic Space group Pca2 1 P2 1 /c P2 1 /c C2/c a (Å) b (Å) c (Å) (4) (4) (4) 8.572(4) (7) (6) 8.388(3) (5) (5) (12) 9.877(4) c (8) α, β, γ ( ) 90, 90, 90 90, (13), 90 90, (7), 90 90, (11), 90 Volume (Å) (2) (15) (11) 3450(3) Z Density calculated (g/cm 3 ) Absorption coefficient (mm -1 ) F(000) Crystal size (mm 3 ) 0.20 x 0.20 x Theta range for data collection ( ) 2.0 to to to to 25.4 Index ranges Reflections collected Independent reflections Completeness to theta Max. and min. transmission Refinement method -13<h<17-22<k<23-23<l<18-10<h<10-19<k<13-19<l<19-10<h<8-19<k<12-19<l<18-27<h<27-9<k<11-19<l< and and and and full-matrix leastsquares refinement on F 2 full-matrix leastsquares refinement on F 2 full-matrix leastsquares refinement on F 2 full-matrix leastsquares refinement on F 2 Data/ restraints/ parameters 9616 / 1 / / 0 / / 0 / / 0 / 181 Goodness-of-fit on F R1 [I>2σ(I)] R indices (all data) R = and R = and R = and R = and wr2 = wr2 = wr2 = wr2 = Largest diff. peak and hole (e.å -3 ) 0.41 and and and and 0.48 Flack parameter none none none none S36
Enantioselective Organocatalytic Michael Addition of Isorhodanines. to α, β-unsaturated Aldehydes
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2016 Enantioselective Organocatalytic Michael Addition of Isorhodanines to α,
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2016 Enantioselective Organocatalytic Michael Addition of Isorhodanines to α,
chlorostibine Iou-Sheng Ke and François P. Gabbai Department of Chemistry, Texas A&M University, College Station, TX
 σ-donor/acceptor confused ligands: The case of a chlorostibine Iou-Sheng Ke and François P. Gabbai Department of Chemistry, Texas A&M University, College Station, TX 77843-3255. *To whom correspondence
σ-donor/acceptor confused ligands: The case of a chlorostibine Iou-Sheng Ke and François P. Gabbai Department of Chemistry, Texas A&M University, College Station, TX 77843-3255. *To whom correspondence
A facile and general route to 3-((trifluoromethyl)thio)benzofurans and 3-((trifluoromethyl)thio)benzothiophenes
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 A facile and general route to 3-((trifluoromethyl)thio)benzofurans and 3-((trifluoromethyl)thio)benzothiophenes
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 A facile and general route to 3-((trifluoromethyl)thio)benzofurans and 3-((trifluoromethyl)thio)benzothiophenes
Supporting Information for. Catalytic C H α-trifluoromethylation of α,β-unsaturated Carbonyl Compounds
 Supporting Information for Catalytic C H α-trifluoromethylation of α,β-unsaturated Carbonyl Compounds Zhongxue Fang, a Yongquan Ning, a Pengbing Mi, a Peiqiu Liao, a Xihe Bi* a,b a Department of Chemistry,
Supporting Information for Catalytic C H α-trifluoromethylation of α,β-unsaturated Carbonyl Compounds Zhongxue Fang, a Yongquan Ning, a Pengbing Mi, a Peiqiu Liao, a Xihe Bi* a,b a Department of Chemistry,
Supporting Information
 Supporting Information Montmorillonite KSF-Catalyzed One-pot, Three-component, Aza-Diels- Alder Reactions of Methylenecyclopropanes With Arylaldehydes and Aromatic Amines Li-Xiong Shao and Min Shi* General
Supporting Information Montmorillonite KSF-Catalyzed One-pot, Three-component, Aza-Diels- Alder Reactions of Methylenecyclopropanes With Arylaldehydes and Aromatic Amines Li-Xiong Shao and Min Shi* General
Cycloaddition of Homochiral Dihydroimidazoles: A 1,3-Dipolar Cycloaddition Route to Optically Active Pyrrolo[1,2-a]imidazoles
![Cycloaddition of Homochiral Dihydroimidazoles: A 1,3-Dipolar Cycloaddition Route to Optically Active Pyrrolo[1,2-a]imidazoles Cycloaddition of Homochiral Dihydroimidazoles: A 1,3-Dipolar Cycloaddition Route to Optically Active Pyrrolo[1,2-a]imidazoles](/thumbs/89/100111450.jpg) X-Ray crystallographic data tables for paper: Supplementary Material (ESI) for Organic & Biomolecular Chemistry Cycloaddition of Homochiral Dihydroimidazoles: A 1,3-Dipolar Cycloaddition Route to Optically
X-Ray crystallographic data tables for paper: Supplementary Material (ESI) for Organic & Biomolecular Chemistry Cycloaddition of Homochiral Dihydroimidazoles: A 1,3-Dipolar Cycloaddition Route to Optically
Copper-catalyzed formal O-H insertion reaction of α-diazo-1,3-dicarb- onyl compounds to carboxylic acids with the assistance of isocyanide
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Copper-catalyzed formal O-H insertion reaction of α-diazo-1,3-dicarb- onyl compounds to carboxylic
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Copper-catalyzed formal O-H insertion reaction of α-diazo-1,3-dicarb- onyl compounds to carboxylic
Supporting Information
 Supporting Information Transition-metal-free Ring Expansion Reactions of Indene-1,3-dione: Synthesis of Functionalized Benzoannulated Seven-Membered Ring Compounds Qiyi Yao, Lingkai Kong, Mengdan Wang,
Supporting Information Transition-metal-free Ring Expansion Reactions of Indene-1,3-dione: Synthesis of Functionalized Benzoannulated Seven-Membered Ring Compounds Qiyi Yao, Lingkai Kong, Mengdan Wang,
Nickel and Platinum PCP Pincer Complexes Incorporating an Acyclic Diaminoalkyl Central Moiety Connecting Imidazole or Pyrazole Rings
 ickel and Platinum PCP Pincer Complexes Incorporating an Acyclic Diaminoalkyl Central Moiety Connecting Imidazole or Pyrazole Rings Braulio M. Puerta Lombardi, Rudy M. Braun, Chris Gendy, Chia Yun Chang,
ickel and Platinum PCP Pincer Complexes Incorporating an Acyclic Diaminoalkyl Central Moiety Connecting Imidazole or Pyrazole Rings Braulio M. Puerta Lombardi, Rudy M. Braun, Chris Gendy, Chia Yun Chang,
Photo-Induced Self-Assembly of Pt(II)-Linked Rings and Cages via the Photolabilization of a Pt(II) Pyridine Bond
 Photo-Induced Self-Assembly of Pt(II)-Linked Rings and Cages via the Photolabilization of a Pt(II) Pyridine Bond Ken-ichi Yamashita, Kei-ichi Sato, Masaki Kawano and Makoto Fujita* Contents; Figure S1.
Photo-Induced Self-Assembly of Pt(II)-Linked Rings and Cages via the Photolabilization of a Pt(II) Pyridine Bond Ken-ichi Yamashita, Kei-ichi Sato, Masaki Kawano and Makoto Fujita* Contents; Figure S1.
Electronic Supplementary Information (ESI)
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information (ESI) Cyclopentadienyl iron dicarbonyl (CpFe(CO) 2 ) derivatives
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information (ESI) Cyclopentadienyl iron dicarbonyl (CpFe(CO) 2 ) derivatives
Enantioselective Synthesis of the Anti-inflammatory Agent ( )-Acanthoic Acid
 Enantioselective Synthesis of the Anti-inflammatory Agent ( )-Acanthoic Acid Taotao Ling, a Chinmay Chowdhury, a Bryan A. Kramer, a Binh G. Vong, a Michael A. Palladino b and Emmanuel A. Theodorakis a
Enantioselective Synthesis of the Anti-inflammatory Agent ( )-Acanthoic Acid Taotao Ling, a Chinmay Chowdhury, a Bryan A. Kramer, a Binh G. Vong, a Michael A. Palladino b and Emmanuel A. Theodorakis a
SUPPLEMENTARY MATERIAL. A Facile and Convenient Approach for the Synthesis of Novel Sesamol-Oxazine and Quinoline- Oxazine Hybrids
 10.1071/CH17272_AC CSIRO 2017 Australian Journal of Chemistry 2017, 70(12), 1285-1290 SUPPLEMENTARY MATERIAL A Facile and Convenient Approach for the Synthesis of Novel Sesamol-Oxazine and Quinoline- Oxazine
10.1071/CH17272_AC CSIRO 2017 Australian Journal of Chemistry 2017, 70(12), 1285-1290 SUPPLEMENTARY MATERIAL A Facile and Convenient Approach for the Synthesis of Novel Sesamol-Oxazine and Quinoline- Oxazine
IV. ANHANG 179. Anhang 178
 Anhang 178 IV. ANHANG 179 1. Röntgenstrukturanalysen (Tabellen) 179 1.1. Diastereomer A (Diplomarbeit) 179 1.2. Diastereomer B (Diplomarbeit) 186 1.3. Aldoladdukt 5A 193 1.4. Aldoladdukt 13A 200 1.5. Aldoladdukt
Anhang 178 IV. ANHANG 179 1. Röntgenstrukturanalysen (Tabellen) 179 1.1. Diastereomer A (Diplomarbeit) 179 1.2. Diastereomer B (Diplomarbeit) 186 1.3. Aldoladdukt 5A 193 1.4. Aldoladdukt 13A 200 1.5. Aldoladdukt
The Free Internet Journal for Organic Chemistry
 The Free Internet Journal for Organic Chemistry Paper Archive for Organic Chemistry Arkivoc 2018, part iii, S1-S6 Synthesis of dihydropyranones and dihydropyrano[2,3- d][1,3]dioxine-diones by cyclization
The Free Internet Journal for Organic Chemistry Paper Archive for Organic Chemistry Arkivoc 2018, part iii, S1-S6 Synthesis of dihydropyranones and dihydropyrano[2,3- d][1,3]dioxine-diones by cyclization
Supporting Information. A catalyst-free multicomponent domino sequence for the. diastereoselective synthesis of (E)-3-[2-arylcarbonyl-3-
 Supporting Information for A catalyst-free multicomponent domino sequence for the diastereoselective synthesis of (E)-3-[2-arylcarbonyl-3- (arylamino)allyl]chromen-4-ones Pitchaimani Prasanna 1, Pethaiah
Supporting Information for A catalyst-free multicomponent domino sequence for the diastereoselective synthesis of (E)-3-[2-arylcarbonyl-3- (arylamino)allyl]chromen-4-ones Pitchaimani Prasanna 1, Pethaiah
Supporting Information for
 Supporting Information for An atom-economic route to densely functionalized thiophenes via base-catalyzed rearrangement of 5-propargyl-2H-thiopyran-4(3H)-ones Chunlin Tang a, Jian Qin b, Xingqi Li *a a
Supporting Information for An atom-economic route to densely functionalized thiophenes via base-catalyzed rearrangement of 5-propargyl-2H-thiopyran-4(3H)-ones Chunlin Tang a, Jian Qin b, Xingqi Li *a a
Copper-Catalyzed Oxidative Dehydrogenative N-N Bond. Formation for the Synthesis of N,N -Diarylindazol-3-ones
 Electronic Supplementary Material (ESI) for Organic Chemistry Frontiers. This journal is the Partner Organisations 2016 Supporting information Copper-Catalyzed Oxidative Dehydrogenative - Bond Formation
Electronic Supplementary Material (ESI) for Organic Chemistry Frontiers. This journal is the Partner Organisations 2016 Supporting information Copper-Catalyzed Oxidative Dehydrogenative - Bond Formation
Supporting Information for Substituent Effects on the Properties of Borafluorenes
 Supporting Information for Substituent Effects on the Properties of Borafluorenes Mallory F. Smith, S. Joel Cassidy, Ian A. Adams, Monica Vasiliu, Deidra L. Gerlach, David Dixon*, Paul A. Rupar* Department
Supporting Information for Substituent Effects on the Properties of Borafluorenes Mallory F. Smith, S. Joel Cassidy, Ian A. Adams, Monica Vasiliu, Deidra L. Gerlach, David Dixon*, Paul A. Rupar* Department
Supporting Information
 Supporting Information Gold-catalyzed Cycloisomerization of 1,6-Diyne-4-en-3-ols to form Naphthyl Ketone Derivatives. Jian-Jou Lian and Rai-Shung Liu* Department of Chemistry, National Tsing-Hua University,
Supporting Information Gold-catalyzed Cycloisomerization of 1,6-Diyne-4-en-3-ols to form Naphthyl Ketone Derivatives. Jian-Jou Lian and Rai-Shung Liu* Department of Chemistry, National Tsing-Hua University,
Supplementary Information. Living Ring-Opening Polymerization of Lactones by N-Heterocyclic Olefin/Al(C 6 F 5 ) 3
 Supplementary Information Living Ring-Opening Polymerization of Lactones by N-Heterocyclic Olefin/Al(C 6 F 5 ) 3 Lewis Pairs: Structures of Intermediates, Kinetics, and Mechanism Qianyi Wang, Wuchao Zhao,
Supplementary Information Living Ring-Opening Polymerization of Lactones by N-Heterocyclic Olefin/Al(C 6 F 5 ) 3 Lewis Pairs: Structures of Intermediates, Kinetics, and Mechanism Qianyi Wang, Wuchao Zhao,
Supporting Information
 Supporting Information for AgOTf-catalyzed one-pot reactions of 2-alkynylbenzaldoximes with α,β-unsaturated carbonyl compounds Qiuping Ding 1, Dan Wang 1, Puying Luo* 2, Meiling Liu 1, Shouzhi Pu* 3 and
Supporting Information for AgOTf-catalyzed one-pot reactions of 2-alkynylbenzaldoximes with α,β-unsaturated carbonyl compounds Qiuping Ding 1, Dan Wang 1, Puying Luo* 2, Meiling Liu 1, Shouzhi Pu* 3 and
Supporting Information
 Supporting Information Vinylogous elimination/heck coupling/allylation domino reactions: access to 2- substituted 2,3-dihydrobenzofurans and indolines Jianguo Yang, *, Hanjie Mo, Xiuxiu Jin, Dongdong Cao,
Supporting Information Vinylogous elimination/heck coupling/allylation domino reactions: access to 2- substituted 2,3-dihydrobenzofurans and indolines Jianguo Yang, *, Hanjie Mo, Xiuxiu Jin, Dongdong Cao,
9-amino-(9-deoxy)cinchona alkaloids-derived novel chiral phase-transfer catalysts
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 9-amino-(9-deoxy)cinchona alkaloids-derived novel chiral phase-transfer
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 9-amino-(9-deoxy)cinchona alkaloids-derived novel chiral phase-transfer
Supporting Information One-Pot Approach to Chiral Chromenes via Enantioselective Organocatalytic Domino Oxa-Michael-Aldol Reaction
 Supporting Information ne-pot Approach to Chiral Chromenes via Enantioselective rganocatalytic Domino xa-michael-aldol Reaction Hao Li, Jian Wang, Timiyin E-Nunu, Liansuo Zu, Wei Jiang, Shaohua Wei, *
Supporting Information ne-pot Approach to Chiral Chromenes via Enantioselective rganocatalytic Domino xa-michael-aldol Reaction Hao Li, Jian Wang, Timiyin E-Nunu, Liansuo Zu, Wei Jiang, Shaohua Wei, *
E-H (E = B, Si, C) Bond Activation by Tuning Structural and Electronic Properties of Phosphenium Cations
 E-H (E = B, Si, C) Bond Activation by Tuning Structural and Electronic Properties of Phosphenium Cations Nemanja Đorđević, Rakesh Ganguly, Milena Petković, and Dragoslav Vidović Email: drasko.vidovic@monash.edu
E-H (E = B, Si, C) Bond Activation by Tuning Structural and Electronic Properties of Phosphenium Cations Nemanja Đorđević, Rakesh Ganguly, Milena Petković, and Dragoslav Vidović Email: drasko.vidovic@monash.edu
Room Temperature Highly Diastereoselective Zn-Mediated. Allylation of Chiral N-tert-Butanesulfinyl Imines: Remarkable Reaction Condition Controlled
 Supporting Information for: Room Temperature Highly Diastereoselective Zn-Mediated Allylation of Chiral N-tert-Butanesulfinyl Imines: Remarkable Reaction Condition Controlled Stereoselectivity Reversal
Supporting Information for: Room Temperature Highly Diastereoselective Zn-Mediated Allylation of Chiral N-tert-Butanesulfinyl Imines: Remarkable Reaction Condition Controlled Stereoselectivity Reversal
Direct Transformation of Ethylarenes into Primary Aromatic Amides with N-Bromosuccinimide and I 2 -aq NH 3
 Supporting Information Direct Transformation of Ethylarenes into Primary Aromatic Amides with N-Bromosuccinimide and I 2 -aq NH 3 Shohei Shimokawa, Yuhsuke Kawagoe, Katsuhiko Moriyama, Hideo Togo* Graduate
Supporting Information Direct Transformation of Ethylarenes into Primary Aromatic Amides with N-Bromosuccinimide and I 2 -aq NH 3 Shohei Shimokawa, Yuhsuke Kawagoe, Katsuhiko Moriyama, Hideo Togo* Graduate
Supporting Information. Experimental section
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Experimental section General. Proton nuclear magnetic resonance ( 1
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Experimental section General. Proton nuclear magnetic resonance ( 1
Supplementary Figure S1. Single X-ray structure 3a at probability ellipsoids of 20%.
 Supplementary Figure S1. Single X-ray structure 3a at probability ellipsoids of 20%. S1 Supplementary Figure S2. Single X-ray structure 5a at probability ellipsoids of 20%. S2 H 15 Ph Ac Ac I AcH Ph Ac
Supplementary Figure S1. Single X-ray structure 3a at probability ellipsoids of 20%. S1 Supplementary Figure S2. Single X-ray structure 5a at probability ellipsoids of 20%. S2 H 15 Ph Ac Ac I AcH Ph Ac
Pyrrolo[2,3-d:5,4-d']bisthiazoles: Alternate Synthetic Routes and a Comparative Study to Analogous Fused-ring Bithiophenes
![Pyrrolo[2,3-d:5,4-d']bisthiazoles: Alternate Synthetic Routes and a Comparative Study to Analogous Fused-ring Bithiophenes Pyrrolo[2,3-d:5,4-d']bisthiazoles: Alternate Synthetic Routes and a Comparative Study to Analogous Fused-ring Bithiophenes](/thumbs/92/108867714.jpg) SUPPORTING INFORMATION Pyrrolo[2,3-d:5,4-d']bisthiazoles: Alternate Synthetic Routes and a Comparative Study to Analogous Fused-ring Bithiophenes Eric J. Uzelac, Casey B. McCausland, and Seth C. Rasmussen*
SUPPORTING INFORMATION Pyrrolo[2,3-d:5,4-d']bisthiazoles: Alternate Synthetic Routes and a Comparative Study to Analogous Fused-ring Bithiophenes Eric J. Uzelac, Casey B. McCausland, and Seth C. Rasmussen*
Supporting Information for: Intramolecular Hydrogen Bonding-Assisted Cyclocondensation of. 1,2,3-Triazole Synthesis
 Supporting Information for: Intramolecular Hydrogen Bonding-Assisted Cyclocondensation of α-diazoketones with Various Amines: A Strategy for Catalytic Wolff 1,2,3-Triazole Synthesis Zikun Wang, a Xihe
Supporting Information for: Intramolecular Hydrogen Bonding-Assisted Cyclocondensation of α-diazoketones with Various Amines: A Strategy for Catalytic Wolff 1,2,3-Triazole Synthesis Zikun Wang, a Xihe
Supporting Information
 Supporting Information Lewis acid catalyzed ring-opening reactions of methylenecyclopropanes with diphenylphosphine oxide in the presence of sulfur or selenium Min Shi,* Min Jiang and Le-Ping Liu State
Supporting Information Lewis acid catalyzed ring-opening reactions of methylenecyclopropanes with diphenylphosphine oxide in the presence of sulfur or selenium Min Shi,* Min Jiang and Le-Ping Liu State
The N,S-Bidentate Ligand Assisted Pd-Catalyzed C(sp 2 )-H. Carbonylation using Langlois Reagent as CO Source. Supporting Information.
 Electronic upplementary Material (EI) for rganic & Biomolecular Chemistry. This journal is The Royal ociety of Chemistry 2018 The,-Bidentate Ligand Assisted Pd-Catalyzed C(sp 2 )-H Carbonylation using
Electronic upplementary Material (EI) for rganic & Biomolecular Chemistry. This journal is The Royal ociety of Chemistry 2018 The,-Bidentate Ligand Assisted Pd-Catalyzed C(sp 2 )-H Carbonylation using
Supporting Information. Research Center for Marine Drugs, Department of Pharmacy, State Key Laboratory
 Supporting Information Dysiherbols A C and Dysideanone E, Cytotoxic and NF-κB Inhibitory Tetracyclic Meroterpenes from a Dysidea sp. Marine Sponge Wei-Hua Jiao,, Guo-Hua Shi,, Ting-Ting Xu,, Guo-Dong Chen,
Supporting Information Dysiherbols A C and Dysideanone E, Cytotoxic and NF-κB Inhibitory Tetracyclic Meroterpenes from a Dysidea sp. Marine Sponge Wei-Hua Jiao,, Guo-Hua Shi,, Ting-Ting Xu,, Guo-Dong Chen,
Supporting Information
 Supporting Information Wiley-VCH 10 69451 Weinheim, Germany Selective and Bond Formations through Copper- Catalyzed Aerobic xidative Dehydrogenative Couplings of H-hosphonates Yongbo Zhou, Shuangfeng Yin,
Supporting Information Wiley-VCH 10 69451 Weinheim, Germany Selective and Bond Formations through Copper- Catalyzed Aerobic xidative Dehydrogenative Couplings of H-hosphonates Yongbo Zhou, Shuangfeng Yin,
and Selective Allylic Reduction of Allylic Alcohols and Their Derivatives with Benzyl Alcohol
 FeCl 3 6H 2 O-Catalyzed Disproportionation of Allylic Alcohols and Selective Allylic Reduction of Allylic Alcohols and Their Derivatives with Benzyl Alcohol Jialiang Wang, Wen Huang, Zhengxing Zhang, Xu
FeCl 3 6H 2 O-Catalyzed Disproportionation of Allylic Alcohols and Selective Allylic Reduction of Allylic Alcohols and Their Derivatives with Benzyl Alcohol Jialiang Wang, Wen Huang, Zhengxing Zhang, Xu
Metal-free Oxidative Coupling of Amines with Sodium Sulfinates: A Mild Access to Sulfonamides
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting information for Metal-free Oxidative Coupling of Amines with Sodium Sulfinates:
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting information for Metal-free Oxidative Coupling of Amines with Sodium Sulfinates:
Supporting Information. Asymmetric Binary-acid Catalysis with Chiral. Phosphoric Acid and MgF 2 : Catalytic
 Supporting Information Asymmetric Binary-acid Catalysis with Chiral Phosphoric Acid and MgF 2 : Catalytic Enantioselective Friedel-Crafts Reactions of β,γ- Unsaturated-α-Ketoesters Jian Lv, Xin Li, Long
Supporting Information Asymmetric Binary-acid Catalysis with Chiral Phosphoric Acid and MgF 2 : Catalytic Enantioselective Friedel-Crafts Reactions of β,γ- Unsaturated-α-Ketoesters Jian Lv, Xin Li, Long
Fluorinative Ring-opening of Cyclopropanes by Hypervalent Iodine Reagents. An Efficient Method for 1,3- Oxyfluorination and 1,3-Difluorination
 Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2016 Supporting Information Fluorinative Ring-opening of Cyclopropanes by Hypervalent Iodine
Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2016 Supporting Information Fluorinative Ring-opening of Cyclopropanes by Hypervalent Iodine
Supporting Information
 S1 Supporting Information Synthesis of 2-Arylated Hydroxytyrosol Derivatives via Suzuki-Myaura Cross-Coupling Roberta Bernini, a Sandro Cacchi, b* Giancarlo Fabrizi, b* Eleonora Filisti b a Dipartimento
S1 Supporting Information Synthesis of 2-Arylated Hydroxytyrosol Derivatives via Suzuki-Myaura Cross-Coupling Roberta Bernini, a Sandro Cacchi, b* Giancarlo Fabrizi, b* Eleonora Filisti b a Dipartimento
Site-Selective Suzuki-Miyaura Cross-Coupling Reactions of 2,3,4,5-Tetrabromofuran
 1 Site-Selective Suzuki-Miyaura Cross-Coupling Reactions of 2,3,4,5-Tetrabromofuran Munawar Hussain, a Rasheed Ahmad Khera, a Nguyen Thai Hung, a Peter Langer* a,b a Institut für Chemie, Universität Rostock,
1 Site-Selective Suzuki-Miyaura Cross-Coupling Reactions of 2,3,4,5-Tetrabromofuran Munawar Hussain, a Rasheed Ahmad Khera, a Nguyen Thai Hung, a Peter Langer* a,b a Institut für Chemie, Universität Rostock,
Supporting Information
 Supporting Information for Lewis acid-catalyzed redox-neutral amination of 2-(3-pyrroline-1-yl)benzaldehydes via intramolecular [1,5]-hydride shift/isomerization reaction Chun-Huan Jiang, Xiantao Lei,
Supporting Information for Lewis acid-catalyzed redox-neutral amination of 2-(3-pyrroline-1-yl)benzaldehydes via intramolecular [1,5]-hydride shift/isomerization reaction Chun-Huan Jiang, Xiantao Lei,
Supporting information for
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting information for Palladium-Catalyzed Benzothieno[2,3-b]indole Formation via Dehydrative-Dehydrogenative
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting information for Palladium-Catalyzed Benzothieno[2,3-b]indole Formation via Dehydrative-Dehydrogenative
Practical Pd(II)-catalyzed C H Alkylation with Epoxides: One-step Syntheses of 3,4-Dihydroisocoumarins
 Practical Pd(II)-catalyzed C H Alkylation with Epoxides: One-step Syntheses of 3,4-Dihydroisocoumarins Guolin Cheng, Tuan-Jie Li, and Jin-Quan Yu* Department of Chemistry, The Scripps Research Institute,
Practical Pd(II)-catalyzed C H Alkylation with Epoxides: One-step Syntheses of 3,4-Dihydroisocoumarins Guolin Cheng, Tuan-Jie Li, and Jin-Quan Yu* Department of Chemistry, The Scripps Research Institute,
Regioselectivity in the Stille coupling reactions of 3,5- dibromo-2-pyrone.
 Regioselectivity in the Stille coupling reactions of 3,5- dibromo-2-pyrone. Won-Suk Kim, Hyung-Jin Kim and Cheon-Gyu Cho Department of Chemistry, Hanyang University, Seoul 133-791, Korea Experimental Section
Regioselectivity in the Stille coupling reactions of 3,5- dibromo-2-pyrone. Won-Suk Kim, Hyung-Jin Kim and Cheon-Gyu Cho Department of Chemistry, Hanyang University, Seoul 133-791, Korea Experimental Section
Supporting Information. Synthesis and biological evaluation of nojirimycin- and
 Supporting Information for Synthesis and biological evaluation of nojirimycin- and pyrrolidine-based trehalase inhibitors Davide Bini 1, Francesca Cardona 2, Matilde Forcella 1, Camilla Parmeggiani 2,3,
Supporting Information for Synthesis and biological evaluation of nojirimycin- and pyrrolidine-based trehalase inhibitors Davide Bini 1, Francesca Cardona 2, Matilde Forcella 1, Camilla Parmeggiani 2,3,
Divergent synthesis of various iminocyclitols from D-ribose
 Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 205 Divergent synthesis of various iminocyclitols from D-ribose Ramu Petakamsetty,
Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 205 Divergent synthesis of various iminocyclitols from D-ribose Ramu Petakamsetty,
Table of Contents 1 Supplementary Data MCD
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2017 Supporting Information for Magnetic circular dichroism and density functional theory
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2017 Supporting Information for Magnetic circular dichroism and density functional theory
Experimental procedure
 Supporting Information for Direct electrophilic N-trifluoromethylthiolation of amines with trifluoromethanesulfenamide Sébastien Alazet 1,2, Kevin Ollivier 1 and Thierry Billard* 1,2 Address: 1 Institute
Supporting Information for Direct electrophilic N-trifluoromethylthiolation of amines with trifluoromethanesulfenamide Sébastien Alazet 1,2, Kevin Ollivier 1 and Thierry Billard* 1,2 Address: 1 Institute
Electronic supplementary information (ESI) Bodipy functionalized ortho-carborane dyads for low-energy photosensitization
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2014 Electronic supplementary information (ESI) Bodipy functionalized ortho-carborane dyads
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2014 Electronic supplementary information (ESI) Bodipy functionalized ortho-carborane dyads
Supporting Information
 Supporting Information Co(III)-Catalyzed Synthesis of Quinazolines via C-H Activation of -Sulfinylimines and Benzimidates Fen Wang, He Wang, Qiang Wang, Songjie Yu, Xingwei Li* Dalian Institute of Chemical
Supporting Information Co(III)-Catalyzed Synthesis of Quinazolines via C-H Activation of -Sulfinylimines and Benzimidates Fen Wang, He Wang, Qiang Wang, Songjie Yu, Xingwei Li* Dalian Institute of Chemical
Four- and Five-membered Cobaltacycles by Regioselective Cyclometalation. of Benzylsulfide Derivatives via Co(V) intermediates
 Electronic Supplementary Information for Dalton Transactions This journal is The Royal Society of Chemistry 2008 Supporting Information for: Four- and Five-membered Cobaltacycles by Regioselective Cyclometalation
Electronic Supplementary Information for Dalton Transactions This journal is The Royal Society of Chemistry 2008 Supporting Information for: Four- and Five-membered Cobaltacycles by Regioselective Cyclometalation
Heavier chalcogenone complexes of bismuth(iii)trihalides: Potential catalysts for acylative cleavage of cyclic ethers. Supporting Information
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Heavier chalcogenone complexes of bismuth(iii)trihalides: Potential catalysts for acylative
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Heavier chalcogenone complexes of bismuth(iii)trihalides: Potential catalysts for acylative
Supporting Information. Copyright Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2006
 Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 1 A Facile Way to Synthesize 2H-Chromenes: Reconsideration of the Reaction Mechanism between Salicylic Aldehyde and
Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 1 A Facile Way to Synthesize 2H-Chromenes: Reconsideration of the Reaction Mechanism between Salicylic Aldehyde and
Lewis Acid Catalyzed Propargylation of Arenes with O-Propargyl Trichloroacetimidate: Synthesis of 1,3-Diarylpropynes
 Supporting Information for Lewis Acid Catalyzed Propargylation of Arenes with O-Propargyl Trichloroacetimidate: Synthesis of 1,3-Diarylpropynes Changkun Li and Jianbo Wang* Beijing National Laboratory
Supporting Information for Lewis Acid Catalyzed Propargylation of Arenes with O-Propargyl Trichloroacetimidate: Synthesis of 1,3-Diarylpropynes Changkun Li and Jianbo Wang* Beijing National Laboratory
Supplementary information
 Electronic Supplementary Material (ESI) for MedChemComm. This journal is The Royal Society of Chemistry 2015 Supplementary information Synthesis of carboxyimidamide-substituted benzo[c][1,2,5]oxadiazoles
Electronic Supplementary Material (ESI) for MedChemComm. This journal is The Royal Society of Chemistry 2015 Supplementary information Synthesis of carboxyimidamide-substituted benzo[c][1,2,5]oxadiazoles
Palladium-Catalyzed Direct ortho-sulfonylation of. Azobenzenes with Arylsulfonyl Chlorides via C H. Table of Contents
 Electronic upplementary Material (EI) for RC Advances. This journal is The Royal ociety of Chemistry 205 upporting Information Palladium-Catalyzed Direct ortho-ulfonylation of Azobenzenes with Arylsulfonyl
Electronic upplementary Material (EI) for RC Advances. This journal is The Royal ociety of Chemistry 205 upporting Information Palladium-Catalyzed Direct ortho-ulfonylation of Azobenzenes with Arylsulfonyl
Electronic Supplementary Information (ESI)
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information (ESI) CPh 3 as a functional group in P-heterocyclic
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information (ESI) CPh 3 as a functional group in P-heterocyclic
Supporting Information File 2. Crystallographic data of syn-bis-quinoxaline, 16c CH 3 CO 2 C 2 H 5 ;
 Supporting Information File 2 Crystallographic data of syn-bis-quinoxaline, 16c CH 3 CO 2 C 2 H 5 ; Preparation, structures and host guest chemistry of fluorinated syn-bisquinoxaline molecular tweezers
Supporting Information File 2 Crystallographic data of syn-bis-quinoxaline, 16c CH 3 CO 2 C 2 H 5 ; Preparation, structures and host guest chemistry of fluorinated syn-bisquinoxaline molecular tweezers
A straightforward metal-free synthesis of 2-substituted thiazolines in air
 Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2015 Supporting Information for A straightforward metal-free synthesis of 2-substituted thiazolines
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2015 Supporting Information for A straightforward metal-free synthesis of 2-substituted thiazolines
Supporting Information. Experimental section
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Experimental section General. Anhydrous solvents were transferred by
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Experimental section General. Anhydrous solvents were transferred by
ESI for. A simple and efficient protocol for the palladium-catalyzed. ligand-free Suzuki reaction at room temperature in aqueous DMF.
 ESI for A simple and efficient protocol for the palladium-catalyzed ligand-free Suzuki reaction at room temperature in aqueous DMF Chun Liu,* Qijian i, Fanying Bao and Jieshan Qiu State Key Laboratory
ESI for A simple and efficient protocol for the palladium-catalyzed ligand-free Suzuki reaction at room temperature in aqueous DMF Chun Liu,* Qijian i, Fanying Bao and Jieshan Qiu State Key Laboratory
Vilsmeier Haack reagent-promoted formyloxylation of α-chloro-narylacetamides
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 205 Vilsmeier aack reagent-promoted formyloxylation of α-chloro-arylacetamides by formamide Jiann-Jyh
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 205 Vilsmeier aack reagent-promoted formyloxylation of α-chloro-arylacetamides by formamide Jiann-Jyh
Supporting Information
 Supporting Information Copper/Silver Cocatalyzed Oxidative Coupling of Vinylarenes with ICH 2 CF 3 or ICH 2 CHF 2 Leading to β-cf 3 /CHF 2 -Substituted Ketones Niannian Yi, Hao Zhang, Chonghui Xu, Wei
Supporting Information Copper/Silver Cocatalyzed Oxidative Coupling of Vinylarenes with ICH 2 CF 3 or ICH 2 CHF 2 Leading to β-cf 3 /CHF 2 -Substituted Ketones Niannian Yi, Hao Zhang, Chonghui Xu, Wei
Supporting Information
 Supporting Information Wiley-VC 007 9 Weinheim, Germany ew ear Infrared Dyes and Fluorophores Based on Diketopyrrolopyrroles Dipl.-Chem. Georg M. Fischer, Dipl.-Chem. Andreas P. Ehlers, Prof. Dr. Andreas
Supporting Information Wiley-VC 007 9 Weinheim, Germany ew ear Infrared Dyes and Fluorophores Based on Diketopyrrolopyrroles Dipl.-Chem. Georg M. Fischer, Dipl.-Chem. Andreas P. Ehlers, Prof. Dr. Andreas
Patrycja Miszczyk, Dorota Wieczorek, Joanna Gałęzowska, Błażej Dziuk, Joanna Wietrzyk and Ewa Chmielewska. 1. Spectroscopic Data.
 ; doi:10.3390/molecules22020254 S1 of S23 Supplementary Materials: Reaction of 3-Amino-1,2,4-Triazole with Diethyl Phosphite and Triethyl Orthoformate: Acid-Base Properties and Antiosteoporotic Activities
; doi:10.3390/molecules22020254 S1 of S23 Supplementary Materials: Reaction of 3-Amino-1,2,4-Triazole with Diethyl Phosphite and Triethyl Orthoformate: Acid-Base Properties and Antiosteoporotic Activities
Facile construction of the functionalized 4H-chromene via tandem. benzylation and cyclization. Jinmin Fan and Zhiyong Wang*
 Facile construction of the functionalized 4H-chromene via tandem benzylation and cyclization Jinmin Fan and Zhiyong Wang* Hefei National Laboratory for Physical Science at Microscale, Joint- Lab of Green
Facile construction of the functionalized 4H-chromene via tandem benzylation and cyclization Jinmin Fan and Zhiyong Wang* Hefei National Laboratory for Physical Science at Microscale, Joint- Lab of Green
Supporting information
 Electronic upplementary Material (EI) for New Journal of Chemistry. This journal is The Royal ociety of Chemistry and the Centre National de la Recherche cientifique 7 upporting information Lipase catalyzed,-addition
Electronic upplementary Material (EI) for New Journal of Chemistry. This journal is The Royal ociety of Chemistry and the Centre National de la Recherche cientifique 7 upporting information Lipase catalyzed,-addition
Supporting Information for. Palladium-catalyzed Addition Reaction of Aroyl/Heteroaroyl Acid Anhydrides to Norbornenes
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting Information for Palladium-catalyzed Addition Reaction of Aroyl/Heteroaroyl Acid Anhydrides
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting Information for Palladium-catalyzed Addition Reaction of Aroyl/Heteroaroyl Acid Anhydrides
Supporting Information for Iron-catalyzed decarboxylative alkenylation of cycloalkanes with arylvinylic carboxylic acids via a radical process
 Supporting Information for Iron-catalyzed decarboxylative alkenylation of cycloalkanes with arylvinylic carboxylic acids via a radical process Jincan Zhao 1, Hong Fang 1, Jianlin Han* 1,2 and Yi Pan* 1
Supporting Information for Iron-catalyzed decarboxylative alkenylation of cycloalkanes with arylvinylic carboxylic acids via a radical process Jincan Zhao 1, Hong Fang 1, Jianlin Han* 1,2 and Yi Pan* 1
multicomponent synthesis of 5-amino-4-
 Supporting Informartion for Molecular iodine-catalyzed one-pot multicomponent synthesis of 5-amino-4- (arylselanyl)-1h-pyrazoles Camila S. Pires 1, Daniela H. de Oliveira 1, Maria R. B. Pontel 1, Jean
Supporting Informartion for Molecular iodine-catalyzed one-pot multicomponent synthesis of 5-amino-4- (arylselanyl)-1h-pyrazoles Camila S. Pires 1, Daniela H. de Oliveira 1, Maria R. B. Pontel 1, Jean
Tributylphosphine-Catalyzed Cycloaddition of Aziridines with Carbon Disulfide and Isothiocyanate
 upporting Information Tributylphosphine-Catalyzed Cycloaddition of Aziridines with Carbon Disulfide and Isothiocyanate Jing-Yu Wu, Zhi-Bin Luo, Li-Xin Dai and Xue-Long Hou* a tate Key Laboratory of Organometallic
upporting Information Tributylphosphine-Catalyzed Cycloaddition of Aziridines with Carbon Disulfide and Isothiocyanate Jing-Yu Wu, Zhi-Bin Luo, Li-Xin Dai and Xue-Long Hou* a tate Key Laboratory of Organometallic
Supporting Information
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Supporting Information Abnormal N-Heterocyclic Carbene Based Nickel Complex for Catalytic
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Supporting Information Abnormal N-Heterocyclic Carbene Based Nickel Complex for Catalytic
Supporting Information
 Supporting Information Ceric Ammonium Nitrate (CAN) catalyzed efficient one-pot three component aza-diels-alder reactions for a facile synthesis of tetrahydropyranoquinoline derivatives Ravinder Goud Puligoundla
Supporting Information Ceric Ammonium Nitrate (CAN) catalyzed efficient one-pot three component aza-diels-alder reactions for a facile synthesis of tetrahydropyranoquinoline derivatives Ravinder Goud Puligoundla
Supporting Information
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2018 Supporting Information Silver or Cerium-Promoted Free Radical Cascade Difunctionalization
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2018 Supporting Information Silver or Cerium-Promoted Free Radical Cascade Difunctionalization
Hiyama Cross-Coupling of Chloro-, Fluoroand Methoxy- pyridyl trimethylsilanes : Room-temperature Novel Access to Functional Bi(het)aryl
 Hiyama Cross-Coupling of Chloro-, Fluoroand Methoxy- pyridyl trimethylsilanes : Room-temperature Novel Access to Functional Bi(het)aryl Philippe Pierrat, Philippe Gros* and Yves Fort Synthèse Organométallique
Hiyama Cross-Coupling of Chloro-, Fluoroand Methoxy- pyridyl trimethylsilanes : Room-temperature Novel Access to Functional Bi(het)aryl Philippe Pierrat, Philippe Gros* and Yves Fort Synthèse Organométallique
Supporting Information
 Supporting Information Aluminum Complexes of N 2 O 2 3 Formazanate Ligands Supported by Phosphine Oxide Donors Ryan R. Maar, Amir Rabiee Kenaree, Ruizhong Zhang, Yichen Tao, Benjamin D. Katzman, Viktor
Supporting Information Aluminum Complexes of N 2 O 2 3 Formazanate Ligands Supported by Phosphine Oxide Donors Ryan R. Maar, Amir Rabiee Kenaree, Ruizhong Zhang, Yichen Tao, Benjamin D. Katzman, Viktor
Supporting Information
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Synthesis of 3-omosubstituted Pyrroles via Palladium- Catalyzed Intermolecular Oxidative Cyclization
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Synthesis of 3-omosubstituted Pyrroles via Palladium- Catalyzed Intermolecular Oxidative Cyclization
Combined Spectroscopic and Quantum Chemical Study of [trans-ru(c CC 6 H 4 R 1 -
 Combined Spectroscopic and Quantum Chemical Study of [trans-ru(c CC 6 H 4 R 1-4) 2 (dppe) 2 ] n+ and [trans-ru(c CC 6 H 4 R 1-4)(C CC 6 H 4 R 2-4)(dppe) 2 ] n+ (n = 0, 1) Complexes: Interpretations Beyond
Combined Spectroscopic and Quantum Chemical Study of [trans-ru(c CC 6 H 4 R 1-4) 2 (dppe) 2 ] n+ and [trans-ru(c CC 6 H 4 R 1-4)(C CC 6 H 4 R 2-4)(dppe) 2 ] n+ (n = 0, 1) Complexes: Interpretations Beyond
Supporting Information. Synthesis and biological evaluation of 2,3-Bis(het)aryl-4-azaindoles Derivatives as protein kinases inhibitors
 Supporting Information Synthesis and biological evaluation of 2,3-Bis(het)aryl-4-azaindoles Derivatives as protein kinases inhibitors Frédéric Pin, a Frédéric Buron, a Fabienne Saab, a Lionel Colliandre,
Supporting Information Synthesis and biological evaluation of 2,3-Bis(het)aryl-4-azaindoles Derivatives as protein kinases inhibitors Frédéric Pin, a Frédéric Buron, a Fabienne Saab, a Lionel Colliandre,
Aminofluorination of Fluorinated Alkenes
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 Synthesis of ɑ CF 3 and ɑ CF 2 H Amines via Aminofluorination of Fluorinated Alkenes Ling Yang,
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 Synthesis of ɑ CF 3 and ɑ CF 2 H Amines via Aminofluorination of Fluorinated Alkenes Ling Yang,
Rh(III)-Catalyzed C-H Amidation with N-hydroxycarbamates: A. new Entry to N-Carbamate Protected Arylamines
 Rh(III)-Catalyzed C-H Amidation with N-hydroxycarbamates: A new Entry to N-Carbamate Protected Arylamines Bing Zhou,* Juanjuan Du, Yaxi Yang,* Huijin Feng, Yuanchao Li Shanghai Institute of Materia Medica,
Rh(III)-Catalyzed C-H Amidation with N-hydroxycarbamates: A new Entry to N-Carbamate Protected Arylamines Bing Zhou,* Juanjuan Du, Yaxi Yang,* Huijin Feng, Yuanchao Li Shanghai Institute of Materia Medica,
Highly enantioselective cascade synthesis of spiropyrazolones. Supporting Information. NMR spectra and HPLC traces
 Highly enantioselective cascade synthesis of spiropyrazolones Alex Zea a, Andrea-Nekane R. Alba a, Andrea Mazzanti b, Albert Moyano a and Ramon Rios a,c * Supporting Information NMR spectra and HPLC traces
Highly enantioselective cascade synthesis of spiropyrazolones Alex Zea a, Andrea-Nekane R. Alba a, Andrea Mazzanti b, Albert Moyano a and Ramon Rios a,c * Supporting Information NMR spectra and HPLC traces
Kishore Natte, Jianbin Chen, Helfried Neumann, Matthias Beller, and Xiao-Feng Wu*
 Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 204 Kishore Natte, Jianbin Chen, Helfried Neumann, Matthias Beller, and Xiao-Feng
Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 204 Kishore Natte, Jianbin Chen, Helfried Neumann, Matthias Beller, and Xiao-Feng
Multifunctinality and Crystal Dynamics of Highly Stable Porous Metal-Organic Framework [Zn 4 O(NTB) 2 ]
![Multifunctinality and Crystal Dynamics of Highly Stable Porous Metal-Organic Framework [Zn 4 O(NTB) 2 ] Multifunctinality and Crystal Dynamics of Highly Stable Porous Metal-Organic Framework [Zn 4 O(NTB) 2 ]](/thumbs/92/109256432.jpg) Supporting Information Multifunctinality and Crystal Dynamics of Highly Stable Porous Metal-Organic Framework [Zn 4 O(NTB) 2 ] Eun Young Lee, Seung Yeon Jang, and Myunghyun Paik Suh* School of Chemistry,
Supporting Information Multifunctinality and Crystal Dynamics of Highly Stable Porous Metal-Organic Framework [Zn 4 O(NTB) 2 ] Eun Young Lee, Seung Yeon Jang, and Myunghyun Paik Suh* School of Chemistry,
Phosphorus Oxychloride as an Efficient Coupling Reagent for the Synthesis of Ester, Amide and Peptide under Mild Conditions
 Supplementary Information for Phosphorus xychloride as an Efficient Coupling Reagent for the Synthesis of Ester, Amide and Peptide under Mild Conditions u Chen,* a,b Xunfu Xu, a Liu Liu, a Guo Tang,* a
Supplementary Information for Phosphorus xychloride as an Efficient Coupling Reagent for the Synthesis of Ester, Amide and Peptide under Mild Conditions u Chen,* a,b Xunfu Xu, a Liu Liu, a Guo Tang,* a
SUPPORTING INFORMATION. Diastereoselective synthesis of nitroso acetals from (S,E)- -aminated
 SUPPORTING INFORMATION for Diastereoselective synthesis of nitroso acetals from (S,E)- -aminated nitroalkenes via multicomponent [4 + 2]/[3 + 2] cycloadditions promoted by LiCl or LiClO 4 Leandro Lara
SUPPORTING INFORMATION for Diastereoselective synthesis of nitroso acetals from (S,E)- -aminated nitroalkenes via multicomponent [4 + 2]/[3 + 2] cycloadditions promoted by LiCl or LiClO 4 Leandro Lara
Electronic Supplementary Information
 Electronic Supplementary Information Unprecedented Carbon-Carbon Bond Cleavage in Nucleophilic Aziridine Ring Opening Reaction, Efficient Ring Transformation of Aziridines to Imidazolidin-4-ones Jin-Yuan
Electronic Supplementary Information Unprecedented Carbon-Carbon Bond Cleavage in Nucleophilic Aziridine Ring Opening Reaction, Efficient Ring Transformation of Aziridines to Imidazolidin-4-ones Jin-Yuan
Supporting Information for Synthesis of Fused N-Heterocycles via Tandem C-H Activation
 This journal is The Royal Society of Chemistry 212 Supporting Information for Synthesis of Fused -Heterocycles via Tandem C-H Activation Ge Meng, Hong-Ying iu, Gui-Rong Qu, John S. Fossey, Jian-Ping Li,*
This journal is The Royal Society of Chemistry 212 Supporting Information for Synthesis of Fused -Heterocycles via Tandem C-H Activation Ge Meng, Hong-Ying iu, Gui-Rong Qu, John S. Fossey, Jian-Ping Li,*
Supporting Information. Palladium Complexes with Bulky Diphosphine. Synthesis of (Bio-) Adipic Acid from Pentenoic. Acid Mixtures.
 Supporting Information Palladium Complexes with Bulky Diphosphine Ligands as Highly Selective Catalysts for the Synthesis of (Bio-) Adipic Acid from Pentenoic Acid Mixtures. Choon Heng Low, James D. Nobbs,*
Supporting Information Palladium Complexes with Bulky Diphosphine Ligands as Highly Selective Catalysts for the Synthesis of (Bio-) Adipic Acid from Pentenoic Acid Mixtures. Choon Heng Low, James D. Nobbs,*
Supporting Information. Chemoselective Acylation of 2-Amino-8-quinolinol in the Generation of C2-Amides or C8-Esters
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2017 Supporting Information Chemoselective Acylation of 2-Amino-8-quinolinol in the Generation of
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2017 Supporting Information Chemoselective Acylation of 2-Amino-8-quinolinol in the Generation of
Enantioselective Organocatalyzed Direct α-thiocyanation of. Cyclic β-ketoesters by N-Thiocyanatophthalimide
 Supporting Information Enantioselective rganocatalyzed Direct α-thiocyanation of Cyclic β-ketoesters by N-Thiocyanatophthalimide Jiashen Qiu, Di Wu, Pran Gopal Karmaker, Hongquan Yin, and Fu-Xue Chen*
Supporting Information Enantioselective rganocatalyzed Direct α-thiocyanation of Cyclic β-ketoesters by N-Thiocyanatophthalimide Jiashen Qiu, Di Wu, Pran Gopal Karmaker, Hongquan Yin, and Fu-Xue Chen*
Supplementary Figure 1. (X-ray structures of 6p and 7f) O N. Br 6p
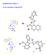 Supplementary Figure 1 (X-ray structures of 6p and 7f) Me Br 6p 6p Supplementary Figures 2-68 (MR Spectra) Supplementary Figure 2. 1 H MR of the 6a Supplementary Figure 3. 13 C MR of the 6a Supplementary
Supplementary Figure 1 (X-ray structures of 6p and 7f) Me Br 6p 6p Supplementary Figures 2-68 (MR Spectra) Supplementary Figure 2. 1 H MR of the 6a Supplementary Figure 3. 13 C MR of the 6a Supplementary
Sotto, 8; Perugia, Italia. Fax: ; Tel: ;
 ELECTRONIC SUPPORTING INFORMATION Ermal Ismalaj a, Giacomo Strappaveccia a, Eleonora Ballerini a, Fausto Elisei a, Oriana Piermatti a, Dmitri Gelman b, Luigi Vaccaro a a CEMIN - Dipartimento di Chi mica,
ELECTRONIC SUPPORTING INFORMATION Ermal Ismalaj a, Giacomo Strappaveccia a, Eleonora Ballerini a, Fausto Elisei a, Oriana Piermatti a, Dmitri Gelman b, Luigi Vaccaro a a CEMIN - Dipartimento di Chi mica,
Supporting Information
 Supporting Information rganocatalytic Enantioselective Formal [4+2] Cycloaddition of Enones with Cyclic N-Sulfonylies and Methylene Chromene for Chiral Spirocyclic Compounds Jie Fei, Qingqing Qian, Xiaohua
Supporting Information rganocatalytic Enantioselective Formal [4+2] Cycloaddition of Enones with Cyclic N-Sulfonylies and Methylene Chromene for Chiral Spirocyclic Compounds Jie Fei, Qingqing Qian, Xiaohua
Supporting Information
 Supporting Information Metal-catalyzed Stereoselective and Protecting-group-free Synthesis of 1,2-cis-Glycosides Using 4,6-Dimethoxy-1,3,5-triazin-2-yl Glycosides as Glycosyl Donors Tomonari Tanaka,* 1
Supporting Information Metal-catalyzed Stereoselective and Protecting-group-free Synthesis of 1,2-cis-Glycosides Using 4,6-Dimethoxy-1,3,5-triazin-2-yl Glycosides as Glycosyl Donors Tomonari Tanaka,* 1
Supporting Information for
 Supporting Information for A ovel Synthesis of luorinated Pyrazoles via Gold(I)-Catalyzed Tandem Aminofluorination of Alkynes in the Presence of Selectfluor Jianqiang Qian, Yunkui Liu,* Jie Zhu, Bo Jiang,
Supporting Information for A ovel Synthesis of luorinated Pyrazoles via Gold(I)-Catalyzed Tandem Aminofluorination of Alkynes in the Presence of Selectfluor Jianqiang Qian, Yunkui Liu,* Jie Zhu, Bo Jiang,
Supporting Information. Consecutive hydrazino-ugi-azide reactions: synthesis of acylhydrazines bearing 1,5- disubstituted tetrazoles
 Supporting Information for Consecutive hydrazino-ugi-azide reactions: synthesis of acylhydrazines bearing 1,5- disubstituted tetrazoles Angélica de Fátima S. Barreto*, Veronica Alves dos Santos, and Carlos
Supporting Information for Consecutive hydrazino-ugi-azide reactions: synthesis of acylhydrazines bearing 1,5- disubstituted tetrazoles Angélica de Fátima S. Barreto*, Veronica Alves dos Santos, and Carlos
Supporting Information
 Supporting Information An Approach to 3,6-Disubstituted 2,5-Dioxybenzoquinones via Two Sequential Suzuki Couplings. Three-step Synthesis of Leucomelone Xianwen Gan, Wei Jiang, Wei Wang,,,* Lihong Hu,,*
Supporting Information An Approach to 3,6-Disubstituted 2,5-Dioxybenzoquinones via Two Sequential Suzuki Couplings. Three-step Synthesis of Leucomelone Xianwen Gan, Wei Jiang, Wei Wang,,,* Lihong Hu,,*
