Supporting information for: Molecular models for the hydrogen age: Hydrogen, nitrogen, oxygen, argon and water
|
|
|
- Ευαδνη Αθανασιάδης
- 6 χρόνια πριν
- Προβολές:
Transcript
1 Supporting information for: Molecular models for the hydrogen age: Hydrogen, nitrogen, oxygen, argon and water Andreas Köster, Monika Thol, and Jadran Vrabec, Lehrstuhl für Thermodynamik und Energietechnik, Universität Paderborn, Paderborn, Germany Lehrstuhl für Thermodynamik, Ruhr-Universität Bochum, Bochum, Germany Phone: Fax: Abstract This supporting information contains predictive mixture data for H 2 + O 2, additional VLE data for mixtures discussed in the main manuscript, VLE results for H 2 mixtures from the GERG-2008 EOS, results from the PC-SAFT and the Peng- Robinson EOS for the Henry s law constant and all molecular simulation data with the associated statistical uncertainty in numerical form. The confidence level for the reported data is 68.3 % (±1 standard deviation). S1
2 Contents 1 Predictive mixture data for H 2 + O 2 S3 2 Additional VLE data S4 3 VLE results for H 2 mixtures from the GERG-2008 EOS S8 4 Henry s law constant from the PC-SAFT and the Peng-Robinson EOS S9 5 Numerical molecular simulation data S Binary mixtures S Hydrogen + nitrogen S Hydrogen + argon S Nitrogen + oxygen S Nitrogen + argon S Argon + oxygen S Nitrogen + water S Hydrogen + water S Solubility data of aqueous systems S Ternary mixtures S Nitrogen + oxygen + argon S Hydrogen + nitrogen + argon S Quaternary mixture S Nitrogen + oxygen + argon + water S29 References S31 S2
3 1 Predictive mixture data for H 2 + O 2 No experimental data were available for this mixture so that the results shown in Fig. S1 are strictly predictive (no binary interaction parameter was adjusted). Figure S1: Isothermal fluid phase diagram of the binary mixture H 2 + O 2 : (, ) Molecular simulation results obtained with the present H 2 force field or (, ) with the H 2 force field of Marx and Nielaba S1, ( ) Peng-Robinson EOS and (- -) PC-SAFT EOS. For comparison, ( ) and ( ) depict molecular simulation data with binary interaction parameters of the chemically similar mixture H 2 + N 2, cf. Table 4 in the main manuscript. Statistical uncertainties of the molecular simulation data are only shown if they exceed symbol size. S3
4 2 Additional VLE data Figure S2: Isothermal saturated densities ρ sat (top) and residual enthalpy of vaporization h res vap (bottom) of the binary mixture H 2 + Ar: ( ) Molecular simulation results obtained with the present H 2 force field or ( ) with the H 2 force field of Marx and Nielaba S1, ( ) Peng-Robinson EOS and (- -) PC-SAFT EOS. Statistical uncertainties of the molecular simulation data are only shown if they exceed symbol size. S4
5 Figure S3: Isothermal saturated densities ρ sat (top) and residual enthalpy of vaporization h res vap (bottom) of the binary mixture N 2 + Ar: ( ) Molecular simulation results, ( ) Peng- Robinson EOS, (- -) PC-SAFT EOS and ( ) EOS-CG. S5
6 Figure S4: Fluid phase diagram of the ternary system N 2 + O 2 + Ar at 120 K and 1.95 to 1.99 MPa: ( ) Molecular simulation results, ( ) Peng-Robinson EOS, (- -) PC-SAFT EOS, ( ) EOS-CG and (+) experimental literature data S2. S6
7 Figure S5: Fluid phase diagram of the ternary system H 2 + N 2 + Ar at 100 K and 2.04 to 2.09 MPa: ( ) Molecular simulation results, ( ) Peng-Robinson EOS, (- -) PC-SAFT EOS, ( ) GERG-2008 EOS and (+) experimental literature data S3. S7
8 3 VLE results for H 2 mixtures from the GERG-2008 EOS In Fig. S6 the GERG-2008 EOS was used to calculate the phase envelope of H 2 mixtures. It can be seen that in some cases a false liquid-liquid equilibrium is predicted (bottom). In other cases, the vapor pressure is strongly overestimated. However, cryogenic H 2 mixtures were not the main focus during the development of the GERG-2008 EOS and its normal range of validity in temperature is specified as 90 to 450 K S4, which means that the lowest isotherm is an extrapolation. Figure S6: Isothermal fluid phase diagram of the binary mixtures H 2 + N 2 (top) and H 2 + Ar (bottom): (+) Experimental literature data S3,S5 S11 and ( ) GERG-2008 EOS S4. S8
9 4 Henry s law constant from the PC-SAFT and the Peng-Robinson EOS As discussed in the main manuscript, both the PC-SAFT and the Peng-Robinson EOS fail to reproduce the Henry s law constant of H 2, N 2, O 2 and Ar in H 2 O qualitatively, cf. Fig. S7. A single temperature independent binary interaction parameter was used for these calculations, cf. Table 4 in the main manuscript. Figure S7: Henry s law constant of H 2, N 2, O 2 and Ar in H 2 O (from top to bottom): (, black) official IAPWS guideline S12,S13, (, red) Peng-Robinson EOS, (- -) PC-SAFT EOS, ( ) EOS-CG and (+) experimental literature data (H 2 S14,S15 ; N 2 S15 S18 ; O 2 S19 S22 ; Ar S23 S26 ). S9
10 5 Numerical molecular simulation data 5.1 Binary mixtures Hydrogen + nitrogen Table S1: Homogeneous density data from molecular simulation for the equimolar mixture H 2 + N 2 using the present as well as the H 2 force field of Marx and Nielaba S1. Statistical uncertainties are denoted by δ. T p ρ H2,present δρ H2,present ρ H2,Marx δρ H2,Marx K MPa mol/l mol/l mol/l mol/l S10
11 Table S2: Vapor-liquid equilibrium data from molecular simulation for H2 + N2 using the present H2 force field. Statistical uncertainties are denoted by δ. T p δp xh2 yh2 δyh2 ρliq δρliq ρvap δρvap h res v δ h res v K MPa MPa mol/mol mol/mol mol/mol mol/l mol/l mol/l mol/l kj/mol kj/mol S11
12 Table S3: Vapor-liquid equilibrium data from molecular simulation for H2 + N2 using the H2 force field of Marx and Nielaba S1. Statistical uncertainties are denoted by δ. T p δp xh2 yh2 δyh2 ρliq δρliq ρvap δρvap h res v δ h res v K MPa MPa mol/mol mol/mol mol/mol mol/l mol/l mol/l mol/l kj/mol kj/mol S12
13 5.1.2 Hydrogen + argon Table S4: Homogeneous density data from molecular simulation for the equimolar mixture H 2 + Ar using the present as well as the H 2 force field of Marx and Nielaba S1. Statistical uncertainties are denoted by δ. T p ρ H2,present δρ H2,present ρ H2,Marx δρ H2,Marx K MPa mol/l mol/l mol/l mol/l S13
14 Table S5: Vapor-liquid equilibrium data from molecular simulation for H2 + Ar using the present H2 force field. Statistical uncertainties are denoted by δ. T p δp xh2 yh2 δyh2 ρliq δρliq ρvap δρvap h res v δ h res v K MPa MPa mol/mol mol/mol mol/mol mol/l mol/l mol/l mol/l kj/mol kj/mol S14
15 Table S6: Vapor-liquid equilibrium data from molecular simulation for H2 + Ar using the H2 force field of Marx and Nielaba S1. Statistical uncertainties are denoted by δ. T p δp xh2 yh2 δyh2 ρliq δρliq ρvap δρvap h res v δ h res v K MPa MPa mol/mol mol/mol mol/mol mol/l mol/l mol/l mol/l kj/mol kj/mol S15
16 5.1.3 Nitrogen + oxygen Table S7: Homogeneous density data from molecular simulation for the equimolar mixture N 2 + O 2. Statistical uncertainties are denoted by δ. T p ρ δρ K MPa mol/l mol/l S16
17 Table S8: Vapor-liquid equilibrium data from molecular simulation for N2 + O2. Statistical uncertainties are denoted by δ. T p δp xn2 yn2 δyn2 ρliq δρliq ρvap δρvap h res v δ h res v K MPa MPa mol/mol mol/mol mol/mol mol/l mol/l mol/l mol/l kj/mol kj/mol S17
18 5.1.4 Nitrogen + argon Table S9: Homogeneous density data from molecular simulation for the equimolar mixture N 2 + Ar. Statistical uncertainties are denoted by δ. T p ρ δρ T p ρ δρ K MPa mol/l mol/l K MPa mol/l mol/l S18
19 Table S10: Vapor-liquid equilibrium data from molecular simulation for N2 + Ar. Statistical uncertainties are denoted by δ. T p δp xn2 yn2 δyn2 ρliq δρliq ρvap δρvap h res v δ h res v K MPa MPa mol/mol mol/mol mol/mol mol/l mol/l mol/l mol/l kj/mol kj/mol S19
20 5.1.5 Argon + oxygen Table S11: Homogeneous density data from molecular simulation for the equimolar mixture Ar + O 2. Statistical uncertainties are denoted by δ. T p ρ δρ K MPa mol/l mol/l S20
21 Table S12: Vapor-liquid equilibrium data from molecular simulation for Ar + O2. Statistical uncertainties are denoted by δ. T p δp xar yar δyar ρliq δρliq ρvap δρvap h res v δ h res v K MPa MPa mol/mol mol/mol mol/mol mol/l mol/l mol/l mol/l kj/mol kj/mol S21
22 5.1.6 Nitrogen + water Table S13: Vapor-liquid equilibrium data from molecular simulation for N2 + H2O. Statistical uncertainties are denoted by δ. T p δp xn2 yn2 δyn2 ρliq δρliq ρvap δρvap h res v δ h res v K MPa MPa mol/mol mol/mol mol/mol mol/l mol/l mol/l mol/l kj/mol kj/mol S22
23 5.1.7 Hydrogen + water Table S14: Vapor-liquid equilibrium data from molecular simulation for H2 + H2O using the present H2 force field. Statistical uncertainties are denoted by δ. T p δp xh2 yh2 δyh2 ρliq δρliq ρvap δρvap h res v δ h res v K MPa MPa mol/mol mol/mol mol/mol mol/l mol/l mol/l mol/l kj/mol kj/mol S23
24 Table S15: Vapor-liquid equilibrium data from molecular simulation for H2 + H2O using the H2 force field of Marx and Nielaba S1. Statistical uncertainties are denoted by δ. T p δp xh2 yh2 δyh2 ρliq δρliq ρvap δρvap h res v δ h res v K MPa MPa mol/mol mol/mol mol/mol mol/l mol/l mol/l mol/l kj/mol kj/mol S24
25 5.1.8 Solubility data of aqueous systems Table S16: Henry s law constant data from molecular simulation for H2 (present as well as Marx and Nielaba force field S1 ), N2, O2 and Ar in H2O. Statistical uncertainties are denoted by δ. T HH2,present δhh2,present HH2,Marx δhh2,marx HN2 δhn2 HO2 δho2 HAr δhar K GPa GPa GPa GPa GPa GPa GPa GPa GPa GPa S25
26 5.2 Ternary mixtures Nitrogen + oxygen + argon Table S17: Homogeneous density data from molecular simulation for N 2 + O 2 + Ar at the composition of ambient air x N2 = 0.781, x O2 = and x Ar = mol/mol. Statistical uncertainties are denoted by δ. T p ρ δρ T p ρ δρ K MPa mol/l mol/l K MPa mol/l mol/l S26
27 Table S18: Vapor-liquid equilibrium data from molecular simulation for N2 + O2 + Ar. Statistical uncertainties are denoted by δ. T p δp xn2 xo2 xar yn2 δyn2 yo2 δyo2 yar δyar K MPa MPa mol/mol mol/mol mol/mol mol/mol mol/mol mol/mol mol/mol mol/mol mol/mol S27
28 5.2.2 Hydrogen + nitrogen + argon Table S19: Vapor-liquid equilibrium data from molecular simulation for H2 + N2 + Ar. Statistical uncertainties are denoted by δ. T p δp xh2 xn2 xar yh2 δyh2 yn2 δyn2 yar δyar K MPa MPa mol/mol mol/mol mol/mol mol/mol mol/mol mol/mol mol/mol mol/mol mol/mol S28
29 5.3 Quaternary mixture Nitrogen + oxygen + argon + water Table S20: Homogeneous density data from molecular simulation for N 2 + O 2 + Ar + H 2 O at a composition of x N2 = 0.758, x O2 = 0.204, x Ar = and x H2O = mol/mol (humid air). Statistical uncertainties are denoted by δ. T p ρ δρ T p ρ δρ K MPa mol/l mol/l K MPa mol/l mol/l S29
30 Table S21: Homogeneous density data from molecular simulation for N 2 + O 2 + Ar + H 2 O at a composition of x N2 = 0.163, x O2 = 0.044, x Ar = and x H2O = mol/mol (supercritical H 2 O containing 0.2 mol/mol of air components). Statistical uncertainties are denoted by δ. T p ρ δρ K MPa mol/l mol/l S30
31 References (S1) Marx, D.; Nielaba, P. Path-integral Monte Carlo techniques for rotational motion in two dimensions: Quenched, annealed, and no-spin quantum-statistical averages. Phys. Rev. A 1992, 45, (S2) Fastovskii, V. G.; Petrovskii, Y. V. A study of the vapor-liquid equilibrium in the system oxygen-argon-nitrogen. Zh. Fiz. Khim. 1957, 31, (S3) Xiao, J.; Liu, K. Measurement and correlation of vapor - liquid equilibrium data in H2 - N2 - Ar system. J. Chem. Eng. (China) 1990, 8, (S4) Kunz, O.; Wagner, W. The GERG-2008 wide-range equation of state for natural gases and other mixtures: an expansion of GERG J. Chem. Eng. Data 2012, 57, (S5) Eubanks, L. S. Vapor-Liquid equilibria in the system Hydrogen-Nitrogen-Carbon monoxide. Ph.D. thesis, Rice University, (S6) Akers, W. W.; Eubanks, L. S. Vapor-Liquid Equilibria in the System Hydrogen- Nitrogen-Carbon Monoxide. Adv. Cryog. Eng. 1960, 3, (S7) Streett, W. B.; Calado, J. C. G. Liquid-vapour equilibrium for hydrogen + nitrogen at temperatures from 63 to 110 K and pressures to 57 MPa. J. Chem. Thermodyn. 1978, 10, (S8) Calado, J. C. G.; Streett, W. B. Liquid-vapor equilibrium in the system H 2 + Ar at temperatures from 83 to 141 K and pressures to 52 MPa. Fluid Phase Equilib. 1979, 2, (S9) Ostronov, M. G.; Shatskaya, L. V.; Finyagina, R. A.; Brodskaya, L. F.; Zhirnova, N. A. Gas-Liquid Phase Equilibrium in the Argon-Hydrogen System at Intermediate Pressures. Russ. J. Phys. Chem. 1977, 51, S31
32 (S10) Volk, H.; Halsey Jr., G. D. Solubility of hydrogen and deuterium in liquid argon. J. Chem. Phys. 1960, 33, (S11) Mullins, J. C.; Ziegler, W. T. Phase equilibria in the argon-helium and argon-hydrogen systems from 68 to 108 K and pressures to 120 atmospheres. Adv. Cryog. Eng. 1965, 10, (S12) International Association for the Properties of Water and Steam, Guideline on the Henry s Constant and Vapor-Liquid Distribution Constant for Gases in H 2 O and D 2 O at High Temperatures (S13) Fernández-Prini, R.; Alvarez, J. L.; Harvey, A. H. Henry s constants and vapor liquid distribution constants for gaseous solutes in H 2 O and D 2 O at high temperatures. J. Phys. Chem. Ref. Data 2003, 32, (S14) Morris, D. R.; Yang, L.; Giraudeau, F.; Sun, X.; Steward, F. R. Henry s law constant for hydrogen in natural water and deuterium in heavy water. Phys. Chem. Chem. Phys. 2001, 3, (S15) Alvarez, J.; Fernández-Prini, R. A semiempirical procedure to describe the thermodynamics of dissolution of non-polar gases in water. Fluid Phase Equilib. 1991, 66, (S16) Rettich, T. R.; Battino, R.; Wilhelm, E. Solubility of gases in liquids. XVI. Henry s law coefficients for nitrogen in water at 5 to 50 C. J. Solution Chem. 1984, 13, (S17) Cosgrove, B. A.; Walkley, J. Solubilities of gases in H 2 O and 2 H 2 O. J. Chromatogr. A 1981, 216, (S18) Alvarez, J.; Crovetto, R.; Fernández-Prini, R. The dissolution of N 2 and of H 2 in water from room temperature to 640 K. Ber. Bunsenges. Phys. Chem. 1988, 92, S32
33 (S19) Rettich, T. R.; Battino, R.; Wilhelm, E. Solubility of gases in liquids. 22. Highprecision determination of Henry s law constants of oxygen in liquid water from T= 274 K to T= 328 K. J. Chem. Thermodyn. 2000, 32, (S20) Benson, B. B.; Krause, D.; Peterson, M. A. The solubility and isotopic fractionation of gases in dilute aqueous solution. I. Oxygen. J. Solution Chem. 1979, 8, (S21) Rettich, T. R.; Handa, Y. P.; Battino, R.; Wilhelm, E. Solubility of gases in liquids. 13. High-precision determination of Henry s constants for methane and ethane in liquid water at 275 to 328 K. J. Phys. Chem. 1981, 85, (S22) Cramer, S. D. The solubility of oxygen in brines from 0 to 300 C. Ind. Eng. Chem. Process Des. Dev. 1980, 19, (S23) Rettich, T. R.; Battino, R.; Wilhelm, E. Solubility of gases in liquids. 18. Highprecision determination of Henry fugacities for argon in liquid water at 2 to 40 C. J. Solution Chem. 1992, 21, (S24) Krause, D.; Benson, B. B. The solubility and isotopic fractionation of gases in dilute aqueous solution. IIa. Solubilities of the noble gases. J. Solution Chem. 1989, 18, (S25) Potter, R. W.; Clynne, M. A. The solubility of the noble gases He, Ne, Ar, Kr, and Xe in water up to the critical point. J. Solution Chem. 1978, 7, (S26) Crovetto, R.; Fernández-Prini, R.; Japas, M. L. Solubilities of inert gases and methane in H 2 O and in D 2 O in the temperature range of 300 to 600 K. J. Chem. Phys. 1982, 76, S33
Technical Information T-9100 SI. Suva. refrigerants. Thermodynamic Properties of. Suva Refrigerant [R-410A (50/50)]
![Technical Information T-9100 SI. Suva. refrigerants. Thermodynamic Properties of. Suva Refrigerant [R-410A (50/50)] Technical Information T-9100 SI. Suva. refrigerants. Thermodynamic Properties of. Suva Refrigerant [R-410A (50/50)]](/thumbs/89/98928029.jpg) d Suva refrigerants Technical Information T-9100SI Thermodynamic Properties of Suva 9100 Refrigerant [R-410A (50/50)] Thermodynamic Properties of Suva 9100 Refrigerant SI Units New tables of the thermodynamic
d Suva refrigerants Technical Information T-9100SI Thermodynamic Properties of Suva 9100 Refrigerant [R-410A (50/50)] Thermodynamic Properties of Suva 9100 Refrigerant SI Units New tables of the thermodynamic
DuPont Suva 95 Refrigerant
 Technical Information T-95 ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Technical Information T-95 ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
DuPont Suva. DuPont. Thermodynamic Properties of. Refrigerant (R-410A) Technical Information. refrigerants T-410A ENG
 Technical Information T-410A ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 410A Refrigerant (R-410A) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Technical Information T-410A ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 410A Refrigerant (R-410A) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
DuPont Suva 95 Refrigerant
 Technical Information T-95 SI DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Technical Information T-95 SI DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Mean bond enthalpy Standard enthalpy of formation Bond N H N N N N H O O O
 Q1. (a) Explain the meaning of the terms mean bond enthalpy and standard enthalpy of formation. Mean bond enthalpy... Standard enthalpy of formation... (5) (b) Some mean bond enthalpies are given below.
Q1. (a) Explain the meaning of the terms mean bond enthalpy and standard enthalpy of formation. Mean bond enthalpy... Standard enthalpy of formation... (5) (b) Some mean bond enthalpies are given below.
ΙΕΥΘΥΝΤΗΣ: Καθηγητής Γ. ΧΡΥΣΟΛΟΥΡΗΣ Ι ΑΚΤΟΡΙΚΗ ΙΑΤΡΙΒΗ
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΠΟΛΥΤΕΧΝΙΚΗ ΣΧΟΛΗ ΤΜΗΜΑ ΜΗΧΑΝΟΛΟΓΩΝ ΚΑΙ ΑΕΡΟΝΑΥΠΗΓΩΝ ΜΗΧΑΝΙΚΩΝ ΕΡΓΑΣΤΗΡΙΟ ΣΥΣΤΗΜΑΤΩΝ ΠΑΡΑΓΩΓΗΣ & ΑΥΤΟΜΑΤΙΣΜΟΥ / ΥΝΑΜΙΚΗΣ & ΘΕΩΡΙΑΣ ΜΗΧΑΝΩΝ ΙΕΥΘΥΝΤΗΣ: Καθηγητής Γ. ΧΡΥΣΟΛΟΥΡΗΣ Ι ΑΚΤΟΡΙΚΗ
ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΠΟΛΥΤΕΧΝΙΚΗ ΣΧΟΛΗ ΤΜΗΜΑ ΜΗΧΑΝΟΛΟΓΩΝ ΚΑΙ ΑΕΡΟΝΑΥΠΗΓΩΝ ΜΗΧΑΝΙΚΩΝ ΕΡΓΑΣΤΗΡΙΟ ΣΥΣΤΗΜΑΤΩΝ ΠΑΡΑΓΩΓΗΣ & ΑΥΤΟΜΑΤΙΣΜΟΥ / ΥΝΑΜΙΚΗΣ & ΘΕΩΡΙΑΣ ΜΗΧΑΝΩΝ ΙΕΥΘΥΝΤΗΣ: Καθηγητής Γ. ΧΡΥΣΟΛΟΥΡΗΣ Ι ΑΚΤΟΡΙΚΗ
Selecting Critical Properties of Terpenes and Terpenoids through Group-Contribution Methods and Equations of State
 Supporting Information Selecting Critical Properties of Terpenes and Terpenoids through Group-Contribution Methods and Equations of State Mónia A. R. Martins, 1,2 Pedro J. Carvalho, 1 André M. Palma, 1
Supporting Information Selecting Critical Properties of Terpenes and Terpenoids through Group-Contribution Methods and Equations of State Mónia A. R. Martins, 1,2 Pedro J. Carvalho, 1 André M. Palma, 1
APPENDIX A. Summary of the English Engineering (EE) System of Units
 Appendixes A. Summary of the English Engineering (EE) System of Units B. Summary of the International System (SI) of Units C. Friction-Factor Chart D. Oblique-Shock Charts (γ = 1.4) (Two-Dimensional) E.
Appendixes A. Summary of the English Engineering (EE) System of Units B. Summary of the International System (SI) of Units C. Friction-Factor Chart D. Oblique-Shock Charts (γ = 1.4) (Two-Dimensional) E.
Figure 1 T / K Explain, in terms of molecules, why the first part of the graph in Figure 1 is a line that slopes up from the origin.
 Q1.(a) Figure 1 shows how the entropy of a molecular substance X varies with temperature. Figure 1 T / K (i) Explain, in terms of molecules, why the entropy is zero when the temperature is zero Kelvin.
Q1.(a) Figure 1 shows how the entropy of a molecular substance X varies with temperature. Figure 1 T / K (i) Explain, in terms of molecules, why the entropy is zero when the temperature is zero Kelvin.
Η ΕΠΙΔΡΑΣΗ ΤΗΣ ΑΙΘΑΝΟΛΗΣ,ΤΗΣ ΜΕΘΑΝΟΛΗΣ ΚΑΙ ΤΟΥ ΑΙΘΥΛΟΤΡΙΤΟΤΑΓΗ ΒΟΥΤΥΛΑΙΘΕΡΑ ΣΤΙΣ ΙΔΙΟΤΗΤΕΣ ΤΗΣ ΒΕΝΖΙΝΗΣ
 ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙΔΕΥΤΙΚΟ ΙΔΡΥΜΑ ΚΑΒΑΛΑΣ ΣΧΟΛΗ ΤΕΧΝΟΛΟΓΙΚΩΝ ΕΦΑΡΜΟΓΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ Η ΕΠΙΔΡΑΣΗ ΤΗΣ ΑΙΘΑΝΟΛΗΣ,ΤΗΣ ΜΕΘΑΝΟΛΗΣ ΚΑΙ ΤΟΥ ΑΙΘΥΛΟΤΡΙΤΟΤΑΓΗ ΒΟΥΤΥΛΑΙΘΕΡΑ ΣΤΙΣ ΙΔΙΟΤΗΤΕΣ ΤΗΣ ΒΕΝΖΙΝΗΣ ΟΝΟΜΑΤΕΠΩΝΥΜΟ
ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙΔΕΥΤΙΚΟ ΙΔΡΥΜΑ ΚΑΒΑΛΑΣ ΣΧΟΛΗ ΤΕΧΝΟΛΟΓΙΚΩΝ ΕΦΑΡΜΟΓΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ Η ΕΠΙΔΡΑΣΗ ΤΗΣ ΑΙΘΑΝΟΛΗΣ,ΤΗΣ ΜΕΘΑΝΟΛΗΣ ΚΑΙ ΤΟΥ ΑΙΘΥΛΟΤΡΙΤΟΤΑΓΗ ΒΟΥΤΥΛΑΙΘΕΡΑ ΣΤΙΣ ΙΔΙΟΤΗΤΕΣ ΤΗΣ ΒΕΝΖΙΝΗΣ ΟΝΟΜΑΤΕΠΩΝΥΜΟ
4.6 Autoregressive Moving Average Model ARMA(1,1)
 84 CHAPTER 4. STATIONARY TS MODELS 4.6 Autoregressive Moving Average Model ARMA(,) This section is an introduction to a wide class of models ARMA(p,q) which we will consider in more detail later in this
84 CHAPTER 4. STATIONARY TS MODELS 4.6 Autoregressive Moving Average Model ARMA(,) This section is an introduction to a wide class of models ARMA(p,q) which we will consider in more detail later in this
PROPERTIES OF ORDINARY WATER SUBSTANCE AT SATURATION FROF TME CRITICAL POINT DOWN TO 66 C EQUATIONS & TABLES
 PROPERTIES OF ORDINARY WATER SUBSTANCE AT SATURATION FROF TME CRITICAL POINT DOWN TO 66 C EQUATIONS & TABLES M. CONDE ENGINEERING, 2002 M. CONDE ENGINEERING, Zurich 2002 Properties of ordinary water substance
PROPERTIES OF ORDINARY WATER SUBSTANCE AT SATURATION FROF TME CRITICAL POINT DOWN TO 66 C EQUATIONS & TABLES M. CONDE ENGINEERING, 2002 M. CONDE ENGINEERING, Zurich 2002 Properties of ordinary water substance
ΚΥΠΡΙΑΚΗ ΕΤΑΙΡΕΙΑ ΠΛΗΡΟΦΟΡΙΚΗΣ CYPRUS COMPUTER SOCIETY ΠΑΓΚΥΠΡΙΟΣ ΜΑΘΗΤΙΚΟΣ ΔΙΑΓΩΝΙΣΜΟΣ ΠΛΗΡΟΦΟΡΙΚΗΣ 19/5/2007
 Οδηγίες: Να απαντηθούν όλες οι ερωτήσεις. Αν κάπου κάνετε κάποιες υποθέσεις να αναφερθούν στη σχετική ερώτηση. Όλα τα αρχεία που αναφέρονται στα προβλήματα βρίσκονται στον ίδιο φάκελο με το εκτελέσιμο
Οδηγίες: Να απαντηθούν όλες οι ερωτήσεις. Αν κάπου κάνετε κάποιες υποθέσεις να αναφερθούν στη σχετική ερώτηση. Όλα τα αρχεία που αναφέρονται στα προβλήματα βρίσκονται στον ίδιο φάκελο με το εκτελέσιμο
Higher Derivative Gravity Theories
 Higher Derivative Gravity Theories Black Holes in AdS space-times James Mashiyane Supervisor: Prof Kevin Goldstein University of the Witwatersrand Second Mandelstam, 20 January 2018 James Mashiyane WITS)
Higher Derivative Gravity Theories Black Holes in AdS space-times James Mashiyane Supervisor: Prof Kevin Goldstein University of the Witwatersrand Second Mandelstam, 20 January 2018 James Mashiyane WITS)
Supporting Information
 Supporting Information Aqueous Biphasic Systems Composed of a Water Stable Ionic Liquid + Carbohydrates and their Applications Mara G. Freire a,b*, Cláudia L. S. Louros a, Luís Paulo N. Rebelo b and João
Supporting Information Aqueous Biphasic Systems Composed of a Water Stable Ionic Liquid + Carbohydrates and their Applications Mara G. Freire a,b*, Cláudia L. S. Louros a, Luís Paulo N. Rebelo b and João
Optimizing Microwave-assisted Extraction Process for Paprika Red Pigments Using Response Surface Methodology
 2012 34 2 382-387 http / /xuebao. jxau. edu. cn Acta Agriculturae Universitatis Jiangxiensis E - mail ndxb7775@ sina. com 212018 105 W 42 2 min 0. 631 TS202. 3 A 1000-2286 2012 02-0382 - 06 Optimizing
2012 34 2 382-387 http / /xuebao. jxau. edu. cn Acta Agriculturae Universitatis Jiangxiensis E - mail ndxb7775@ sina. com 212018 105 W 42 2 min 0. 631 TS202. 3 A 1000-2286 2012 02-0382 - 06 Optimizing
Απόκριση σε Μοναδιαία Ωστική Δύναμη (Unit Impulse) Απόκριση σε Δυνάμεις Αυθαίρετα Μεταβαλλόμενες με το Χρόνο. Απόστολος Σ.
 Απόκριση σε Δυνάμεις Αυθαίρετα Μεταβαλλόμενες με το Χρόνο The time integral of a force is referred to as impulse, is determined by and is obtained from: Newton s 2 nd Law of motion states that the action
Απόκριση σε Δυνάμεις Αυθαίρετα Μεταβαλλόμενες με το Χρόνο The time integral of a force is referred to as impulse, is determined by and is obtained from: Newton s 2 nd Law of motion states that the action
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. Supplementary to Theoretical Study on the Gas Adsorption Capacity and
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics.
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics.
Jesse Maassen and Mark Lundstrom Purdue University November 25, 2013
 Notes on Average Scattering imes and Hall Factors Jesse Maassen and Mar Lundstrom Purdue University November 5, 13 I. Introduction 1 II. Solution of the BE 1 III. Exercises: Woring out average scattering
Notes on Average Scattering imes and Hall Factors Jesse Maassen and Mar Lundstrom Purdue University November 5, 13 I. Introduction 1 II. Solution of the BE 1 III. Exercises: Woring out average scattering
STEAM TABLES. Mollier Diagram
 STEAM TABLES and Mollier Diagram (S.I. Units) dharm \M-therm\C-steam.pm5 CONTENTS Table No. Page No. 1. Saturated Water and Steam (Temperature) Tables I (ii) 2. Saturated Water and Steam (Pressure) Tables
STEAM TABLES and Mollier Diagram (S.I. Units) dharm \M-therm\C-steam.pm5 CONTENTS Table No. Page No. 1. Saturated Water and Steam (Temperature) Tables I (ii) 2. Saturated Water and Steam (Pressure) Tables
2. Chemical Thermodynamics and Energetics - I
 . Chemical Thermodynamics and Energetics - I 1. Given : Initial Volume ( = 5L dm 3 Final Volume (V = 10L dm 3 ext = 304 cm of Hg Work done W = ext V ext = 304 cm of Hg = 304 atm [... 76cm of Hg = 1 atm]
. Chemical Thermodynamics and Energetics - I 1. Given : Initial Volume ( = 5L dm 3 Final Volume (V = 10L dm 3 ext = 304 cm of Hg Work done W = ext V ext = 304 cm of Hg = 304 atm [... 76cm of Hg = 1 atm]
6.1. Dirac Equation. Hamiltonian. Dirac Eq.
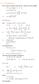 6.1. Dirac Equation Ref: M.Kaku, Quantum Field Theory, Oxford Univ Press (1993) η μν = η μν = diag(1, -1, -1, -1) p 0 = p 0 p = p i = -p i p μ p μ = p 0 p 0 + p i p i = E c 2 - p 2 = (m c) 2 H = c p 2
6.1. Dirac Equation Ref: M.Kaku, Quantum Field Theory, Oxford Univ Press (1993) η μν = η μν = diag(1, -1, -1, -1) p 0 = p 0 p = p i = -p i p μ p μ = p 0 p 0 + p i p i = E c 2 - p 2 = (m c) 2 H = c p 2
By R.L. Snyder (Revised March 24, 2005)
 Humidity Conversion By R.L. Snyder (Revised March 24, 2005) This Web page provides the equations used to make humidity conversions and tables o saturation vapor pressure. For a pd ile o this document,
Humidity Conversion By R.L. Snyder (Revised March 24, 2005) This Web page provides the equations used to make humidity conversions and tables o saturation vapor pressure. For a pd ile o this document,
Dr. D. Dinev, Department of Structural Mechanics, UACEG
 Lecture 4 Material behavior: Constitutive equations Field of the game Print version Lecture on Theory of lasticity and Plasticity of Dr. D. Dinev, Department of Structural Mechanics, UACG 4.1 Contents
Lecture 4 Material behavior: Constitutive equations Field of the game Print version Lecture on Theory of lasticity and Plasticity of Dr. D. Dinev, Department of Structural Mechanics, UACG 4.1 Contents
(1) Describe the process by which mercury atoms become excited in a fluorescent tube (3)
 Q1. (a) A fluorescent tube is filled with mercury vapour at low pressure. In order to emit electromagnetic radiation the mercury atoms must first be excited. (i) What is meant by an excited atom? (1) (ii)
Q1. (a) A fluorescent tube is filled with mercury vapour at low pressure. In order to emit electromagnetic radiation the mercury atoms must first be excited. (i) What is meant by an excited atom? (1) (ii)
Διπλωματική Εργασία. Μελέτη των μηχανικών ιδιοτήτων των stents που χρησιμοποιούνται στην Ιατρική. Αντωνίου Φάνης
 Διπλωματική Εργασία Μελέτη των μηχανικών ιδιοτήτων των stents που χρησιμοποιούνται στην Ιατρική Αντωνίου Φάνης Επιβλέπουσες: Θεοδώρα Παπαδοπούλου, Ομότιμη Καθηγήτρια ΕΜΠ Ζάννη-Βλαστού Ρόζα, Καθηγήτρια
Διπλωματική Εργασία Μελέτη των μηχανικών ιδιοτήτων των stents που χρησιμοποιούνται στην Ιατρική Αντωνίου Φάνης Επιβλέπουσες: Θεοδώρα Παπαδοπούλου, Ομότιμη Καθηγήτρια ΕΜΠ Ζάννη-Βλαστού Ρόζα, Καθηγήτρια
Copper-catalyzed formal O-H insertion reaction of α-diazo-1,3-dicarb- onyl compounds to carboxylic acids with the assistance of isocyanide
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Copper-catalyzed formal O-H insertion reaction of α-diazo-1,3-dicarb- onyl compounds to carboxylic
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Copper-catalyzed formal O-H insertion reaction of α-diazo-1,3-dicarb- onyl compounds to carboxylic
stability and aromaticity in the benzonitrile H 2 O complex with Na+ or Cl
 Electronic Supplementary Data A theoretical investigation on the cooperativity effect, reduced density gradient, stability and aromaticity in the benzonitrile H 2 O complex with Na+ or Cl Jiang-Bo Xie
Electronic Supplementary Data A theoretical investigation on the cooperativity effect, reduced density gradient, stability and aromaticity in the benzonitrile H 2 O complex with Na+ or Cl Jiang-Bo Xie
of the methanol-dimethylamine complex
 Electronic Supplementary Information for: Fundamental and overtone virational spectroscopy, enthalpy of hydrogen ond formation and equilirium constant determination of the methanol-dimethylamine complex
Electronic Supplementary Information for: Fundamental and overtone virational spectroscopy, enthalpy of hydrogen ond formation and equilirium constant determination of the methanol-dimethylamine complex
Aquinas College. Edexcel Mathematical formulae and statistics tables DO NOT WRITE ON THIS BOOKLET
 Aquinas College Edexcel Mathematical formulae and statistics tables DO NOT WRITE ON THIS BOOKLET Pearson Edexcel Level 3 Advanced Subsidiary and Advanced GCE in Mathematics and Further Mathematics Mathematical
Aquinas College Edexcel Mathematical formulae and statistics tables DO NOT WRITE ON THIS BOOKLET Pearson Edexcel Level 3 Advanced Subsidiary and Advanced GCE in Mathematics and Further Mathematics Mathematical
Reaction of a Platinum Electrode for the Measurement of Redox Potential of Paddy Soil
 J. Jpn. Soc. Soil Phys. No. +*0, p.- +*,**1 Eh * ** Reaction of a Platinum Electrode for the Measurement of Redox Potential of Paddy Soil Daisuke MURAKAMI* and Tatsuaki KASUBUCHI** * The United Graduate
J. Jpn. Soc. Soil Phys. No. +*0, p.- +*,**1 Eh * ** Reaction of a Platinum Electrode for the Measurement of Redox Potential of Paddy Soil Daisuke MURAKAMI* and Tatsuaki KASUBUCHI** * The United Graduate
Phys460.nb Solution for the t-dependent Schrodinger s equation How did we find the solution? (not required)
 Phys460.nb 81 ψ n (t) is still the (same) eigenstate of H But for tdependent H. The answer is NO. 5.5.5. Solution for the tdependent Schrodinger s equation If we assume that at time t 0, the electron starts
Phys460.nb 81 ψ n (t) is still the (same) eigenstate of H But for tdependent H. The answer is NO. 5.5.5. Solution for the tdependent Schrodinger s equation If we assume that at time t 0, the electron starts
VBA Microsoft Excel. J. Comput. Chem. Jpn., Vol. 5, No. 1, pp (2006)
 J. Comput. Chem. Jpn., Vol. 5, No. 1, pp. 29 38 (2006) Microsoft Excel, 184-8588 2-24-16 e-mail: yosimura@cc.tuat.ac.jp (Received: July 28, 2005; Accepted for publication: October 24, 2005; Published on
J. Comput. Chem. Jpn., Vol. 5, No. 1, pp. 29 38 (2006) Microsoft Excel, 184-8588 2-24-16 e-mail: yosimura@cc.tuat.ac.jp (Received: July 28, 2005; Accepted for publication: October 24, 2005; Published on
Assalamu `alaikum wr. wb.
 LUMP SUM Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. LUMP SUM Lump sum lump sum lump sum. lump sum fixed price lump sum lump
LUMP SUM Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. LUMP SUM Lump sum lump sum lump sum. lump sum fixed price lump sum lump
The Simply Typed Lambda Calculus
 Type Inference Instead of writing type annotations, can we use an algorithm to infer what the type annotations should be? That depends on the type system. For simple type systems the answer is yes, and
Type Inference Instead of writing type annotations, can we use an algorithm to infer what the type annotations should be? That depends on the type system. For simple type systems the answer is yes, and
 22 .5 Real consumption.5 Real residential investment.5.5.5 965 975 985 995 25.5 965 975 985 995 25.5 Real house prices.5 Real fixed investment.5.5.5 965 975 985 995 25.5 965 975 985 995 25.3 Inflation
22 .5 Real consumption.5 Real residential investment.5.5.5 965 975 985 995 25.5 965 975 985 995 25.5 Real house prices.5 Real fixed investment.5.5.5 965 975 985 995 25.5 965 975 985 995 25.3 Inflation
Table of Contents 1 Supplementary Data MCD
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2017 Supporting Information for Magnetic circular dichroism and density functional theory
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2017 Supporting Information for Magnetic circular dichroism and density functional theory
2 Composition. Invertible Mappings
 Arkansas Tech University MATH 4033: Elementary Modern Algebra Dr. Marcel B. Finan Composition. Invertible Mappings In this section we discuss two procedures for creating new mappings from old ones, namely,
Arkansas Tech University MATH 4033: Elementary Modern Algebra Dr. Marcel B. Finan Composition. Invertible Mappings In this section we discuss two procedures for creating new mappings from old ones, namely,
Κάθε γνήσιο αντίγραφο φέρει υπογραφή του συγγραφέα. / Each genuine copy is signed by the author.
 Κάθε γνήσιο αντίγραφο φέρει υπογραφή του συγγραφέα. / Each genuine copy is signed by the author. 2012, Γεράσιμος Χρ. Σιάσος / Gerasimos Siasos, All rights reserved. Στοιχεία επικοινωνίας συγγραφέα / Author
Κάθε γνήσιο αντίγραφο φέρει υπογραφή του συγγραφέα. / Each genuine copy is signed by the author. 2012, Γεράσιμος Χρ. Σιάσος / Gerasimos Siasos, All rights reserved. Στοιχεία επικοινωνίας συγγραφέα / Author
C H Activation of Cp* Ligand Coordinated to Ruthenium. Center: Synthesis and Reactivity of a Thiolate-Bridged
 Supporting Information C H Activation of Cp* Ligand Coordinated to Ruthenium Center: Synthesis and Reactivity of a Thiolate-Bridged Diruthenium Complex Featuring Fulvene-like Cp* Ligand Xiaoxiao Ji, Dawei
Supporting Information C H Activation of Cp* Ligand Coordinated to Ruthenium Center: Synthesis and Reactivity of a Thiolate-Bridged Diruthenium Complex Featuring Fulvene-like Cp* Ligand Xiaoxiao Ji, Dawei
Major Concepts. Multiphase Equilibrium Stability Applications to Phase Equilibrium. Two-Phase Coexistence
 Major Concepts Multiphase Equilibrium Stability Applications to Phase Equilibrium Phase Rule Clausius-Clapeyron Equation Special case of Gibbs-Duhem wo-phase Coexistence Criticality Metastability Spinodal
Major Concepts Multiphase Equilibrium Stability Applications to Phase Equilibrium Phase Rule Clausius-Clapeyron Equation Special case of Gibbs-Duhem wo-phase Coexistence Criticality Metastability Spinodal
Enthalpy data for the reacting species are given in the table below. The activation energy decreases when the temperature is increased.
 Q1.This question is about the reaction given below. O(g) + H 2O(g) O 2(g) + H 2(g) Enthalpy data for the reacting species are given in the table below. Substance O(g) H 2O(g) O 2(g) H 2(g) ΔH / kj mol
Q1.This question is about the reaction given below. O(g) + H 2O(g) O 2(g) + H 2(g) Enthalpy data for the reacting species are given in the table below. Substance O(g) H 2O(g) O 2(g) H 2(g) ΔH / kj mol
ΕΦΑΡΜΟΓΗ ΕΥΤΕΡΟΒΑΘΜΙΑ ΕΠΕΞΕΡΓΑΣΜΕΝΩΝ ΥΓΡΩΝ ΑΠΟΒΛΗΤΩΝ ΣΕ ΦΥΣΙΚΑ ΣΥΣΤΗΜΑΤΑ ΚΛΙΝΗΣ ΚΑΛΑΜΙΩΝ
 ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙ ΕΥΤΙΚΟ Ι ΡΥΜΑ ΚΡΗΤΗΣ ΤΜΗΜΑ ΦΥΣΙΚΩΝ ΠΟΡΩΝ ΚΑΙ ΠΕΡΙΒΑΛΛΟΝΤΟΣ ΕΦΑΡΜΟΓΗ ΕΥΤΕΡΟΒΑΘΜΙΑ ΕΠΕΞΕΡΓΑΣΜΕΝΩΝ ΥΓΡΩΝ ΑΠΟΒΛΗΤΩΝ ΣΕ ΦΥΣΙΚΑ ΣΥΣΤΗΜΑΤΑ ΚΛΙΝΗΣ ΚΑΛΑΜΙΩΝ ΕΠΙΜΕΛΕΙΑ: ΑΡΜΕΝΑΚΑΣ ΜΑΡΙΝΟΣ ΧΑΝΙΑ
ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙ ΕΥΤΙΚΟ Ι ΡΥΜΑ ΚΡΗΤΗΣ ΤΜΗΜΑ ΦΥΣΙΚΩΝ ΠΟΡΩΝ ΚΑΙ ΠΕΡΙΒΑΛΛΟΝΤΟΣ ΕΦΑΡΜΟΓΗ ΕΥΤΕΡΟΒΑΘΜΙΑ ΕΠΕΞΕΡΓΑΣΜΕΝΩΝ ΥΓΡΩΝ ΑΠΟΒΛΗΤΩΝ ΣΕ ΦΥΣΙΚΑ ΣΥΣΤΗΜΑΤΑ ΚΛΙΝΗΣ ΚΑΛΑΜΙΩΝ ΕΠΙΜΕΛΕΙΑ: ΑΡΜΕΝΑΚΑΣ ΜΑΡΙΝΟΣ ΧΑΝΙΑ
ST5224: Advanced Statistical Theory II
 ST5224: Advanced Statistical Theory II 2014/2015: Semester II Tutorial 7 1. Let X be a sample from a population P and consider testing hypotheses H 0 : P = P 0 versus H 1 : P = P 1, where P j is a known
ST5224: Advanced Statistical Theory II 2014/2015: Semester II Tutorial 7 1. Let X be a sample from a population P and consider testing hypotheses H 0 : P = P 0 versus H 1 : P = P 1, where P j is a known
 1 1 1 2 1 2 2 1 43 123 5 122 3 1 312 1 1 122 1 1 1 1 6 1 7 1 6 1 7 1 3 4 2 312 43 4 3 3 1 1 4 1 1 52 122 54 124 8 1 3 1 1 1 1 1 152 1 1 1 1 1 1 152 1 5 1 152 152 1 1 3 9 1 159 9 13 4 5 1 122 1 4 122 5
1 1 1 2 1 2 2 1 43 123 5 122 3 1 312 1 1 122 1 1 1 1 6 1 7 1 6 1 7 1 3 4 2 312 43 4 3 3 1 1 4 1 1 52 122 54 124 8 1 3 1 1 1 1 1 152 1 1 1 1 1 1 152 1 5 1 152 152 1 1 3 9 1 159 9 13 4 5 1 122 1 4 122 5
ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΤΜΗΜΑ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ ΥΠΟΛΟΓΙΣΤΩΝ ΤΟΜΕΑΣ ΣΥΣΤΗΜΑΤΩΝ ΗΛΕΚΤΡΙΚΗΣ ΕΝΕΡΓΕΙΑΣ
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΤΜΗΜΑ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ ΥΠΟΛΟΓΙΣΤΩΝ ΤΟΜΕΑΣ ΣΥΣΤΗΜΑΤΩΝ ΗΛΕΚΤΡΙΚΗΣ ΕΝΕΡΓΕΙΑΣ Διπλωματική Εργασία του φοιτητή του τμήματος Ηλεκτρολόγων Μηχανικών και Τεχνολογίας Ηλεκτρονικών
ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΤΜΗΜΑ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ ΥΠΟΛΟΓΙΣΤΩΝ ΤΟΜΕΑΣ ΣΥΣΤΗΜΑΤΩΝ ΗΛΕΚΤΡΙΚΗΣ ΕΝΕΡΓΕΙΑΣ Διπλωματική Εργασία του φοιτητή του τμήματος Ηλεκτρολόγων Μηχανικών και Τεχνολογίας Ηλεκτρονικών
DESIGN OF MACHINERY SOLUTION MANUAL h in h 4 0.
 DESIGN OF MACHINERY SOLUTION MANUAL -7-1! PROBLEM -7 Statement: Design a double-dwell cam to move a follower from to 25 6, dwell for 12, fall 25 and dwell for the remader The total cycle must take 4 sec
DESIGN OF MACHINERY SOLUTION MANUAL -7-1! PROBLEM -7 Statement: Design a double-dwell cam to move a follower from to 25 6, dwell for 12, fall 25 and dwell for the remader The total cycle must take 4 sec
Approximation of distance between locations on earth given by latitude and longitude
 Approximation of distance between locations on earth given by latitude and longitude Jan Behrens 2012-12-31 In this paper we shall provide a method to approximate distances between two points on earth
Approximation of distance between locations on earth given by latitude and longitude Jan Behrens 2012-12-31 In this paper we shall provide a method to approximate distances between two points on earth
MOTROL. COMMISSION OF MOTORIZATION AND ENERGETICS IN AGRICULTURE 2014, Vol. 16, No. 5,
 MOTROL. COMMISSION OF MOTORIZATION AND ENERGETICS IN AGRICULTURE 2014, Vol. 16, No. 5, 3 14 -, :., 83, 66404 e-mail: chupinvr@istu.irk.ru...,,., -,.,. :,,,,,, -, - [1].,.., [2, 3].,.,,,.,,, [4, 5].,..1.
MOTROL. COMMISSION OF MOTORIZATION AND ENERGETICS IN AGRICULTURE 2014, Vol. 16, No. 5, 3 14 -, :., 83, 66404 e-mail: chupinvr@istu.irk.ru...,,., -,.,. :,,,,,, -, - [1].,.., [2, 3].,.,,,.,,, [4, 5].,..1.
Lewis Acid Catalyzed Propargylation of Arenes with O-Propargyl Trichloroacetimidate: Synthesis of 1,3-Diarylpropynes
 Supporting Information for Lewis Acid Catalyzed Propargylation of Arenes with O-Propargyl Trichloroacetimidate: Synthesis of 1,3-Diarylpropynes Changkun Li and Jianbo Wang* Beijing National Laboratory
Supporting Information for Lewis Acid Catalyzed Propargylation of Arenes with O-Propargyl Trichloroacetimidate: Synthesis of 1,3-Diarylpropynes Changkun Li and Jianbo Wang* Beijing National Laboratory
Strain gauge and rosettes
 Strain gauge and rosettes Introduction A strain gauge is a device which is used to measure strain (deformation) on an object subjected to forces. Strain can be measured using various types of devices classified
Strain gauge and rosettes Introduction A strain gauge is a device which is used to measure strain (deformation) on an object subjected to forces. Strain can be measured using various types of devices classified
Supporting Information
 Supporting Information Wiley-VC 007 9 Weinheim, Germany ew ear Infrared Dyes and Fluorophores Based on Diketopyrrolopyrroles Dipl.-Chem. Georg M. Fischer, Dipl.-Chem. Andreas P. Ehlers, Prof. Dr. Andreas
Supporting Information Wiley-VC 007 9 Weinheim, Germany ew ear Infrared Dyes and Fluorophores Based on Diketopyrrolopyrroles Dipl.-Chem. Georg M. Fischer, Dipl.-Chem. Andreas P. Ehlers, Prof. Dr. Andreas
ΜΕΛΕΤΗ ΤΗΣ ΠΛΑΣΤΙΚΟΤΗΤΑΣ ΑΡΓΙΛΟΥΧΩΝ ΜΙΓΜΑΤΩΝ ΜΕ ΠΡΟΣΘΗΚΗ ΣΙΔΗΡΑΛΟΥΜΙΝΑΣ ΑΠΟ ΤΗ ΔΙΕΡΓΑΣΙΑ BAYER
 Πρακτικά 1ου Πανελληνίου Συνεδρίου για την Αξιοποίηση των Βιομηχανικών Παραπροϊόντων στη Δόμηση, ΕΒΙΠΑΡ, Θεσσαλονίκη, 24-26 Νοεμβρίου 2005 ΜΕΛΕΤΗ ΤΗΣ ΠΛΑΣΤΙΚΟΤΗΤΑΣ ΑΡΓΙΛΟΥΧΩΝ ΜΙΓΜΑΤΩΝ ΜΕ ΠΡΟΣΘΗΚΗ ΣΙΔΗΡΑΛΟΥΜΙΝΑΣ
Πρακτικά 1ου Πανελληνίου Συνεδρίου για την Αξιοποίηση των Βιομηχανικών Παραπροϊόντων στη Δόμηση, ΕΒΙΠΑΡ, Θεσσαλονίκη, 24-26 Νοεμβρίου 2005 ΜΕΛΕΤΗ ΤΗΣ ΠΛΑΣΤΙΚΟΤΗΤΑΣ ΑΡΓΙΛΟΥΧΩΝ ΜΙΓΜΑΤΩΝ ΜΕ ΠΡΟΣΘΗΚΗ ΣΙΔΗΡΑΛΟΥΜΙΝΑΣ
Ύγρανση και Αφύγρανση. Ψυχρομετρία. 21-Nov-16
 Ύγρανση και Αφύγρανση Ψυχρομετρία η μελέτη των ιδιοτήτων του υγρού αέρα δηλαδή του μίγματος αέρα-ατμού-νερού. Ένα βασικό πρόβλημα: Δεδομένων των P (bar) Πίεση T ( o C) Θερμοκρασία Υ (kg/kg ξβ) Υγρασία
Ύγρανση και Αφύγρανση Ψυχρομετρία η μελέτη των ιδιοτήτων του υγρού αέρα δηλαδή του μίγματος αέρα-ατμού-νερού. Ένα βασικό πρόβλημα: Δεδομένων των P (bar) Πίεση T ( o C) Θερμοκρασία Υ (kg/kg ξβ) Υγρασία
Problem Set 9 Solutions. θ + 1. θ 2 + cotθ ( ) sinθ e iφ is an eigenfunction of the ˆ L 2 operator. / θ 2. φ 2. sin 2 θ φ 2. ( ) = e iφ. = e iφ cosθ.
 Chemistry 362 Dr Jean M Standard Problem Set 9 Solutions The ˆ L 2 operator is defined as Verify that the angular wavefunction Y θ,φ) Also verify that the eigenvalue is given by 2! 2 & L ˆ 2! 2 2 θ 2 +
Chemistry 362 Dr Jean M Standard Problem Set 9 Solutions The ˆ L 2 operator is defined as Verify that the angular wavefunction Y θ,φ) Also verify that the eigenvalue is given by 2! 2 & L ˆ 2! 2 2 θ 2 +
Σπανό Ιωάννη Α.Μ. 148
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΔΙΑΤΜΗΜΑΤΙΚΟ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΙΣ ΠΕΡΙΒΑΛΛΟΝΤΙΚΕΣ ΕΠΙΣΤΗΜΕΣ Ηλεκτροχημική εναπόθεση και μελέτη των ιδιοτήτων, λεπτών υμενίων μεταβατικών μετάλλων, για παραγωγή H2 Διπλωματική
ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΔΙΑΤΜΗΜΑΤΙΚΟ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΙΣ ΠΕΡΙΒΑΛΛΟΝΤΙΚΕΣ ΕΠΙΣΤΗΜΕΣ Ηλεκτροχημική εναπόθεση και μελέτη των ιδιοτήτων, λεπτών υμενίων μεταβατικών μετάλλων, για παραγωγή H2 Διπλωματική
Συστήματα Βιομηχανικών Διεργασιών 6ο εξάμηνο
 Συστήματα Βιομηχανικών Διεργασιών 6ο εξάμηνο Μέρος 1 ο : Εισαγωγικά (διαστ., πυκν., θερμ., πίεση, κτλ.) Μέρος 2 ο : Ισοζύγια μάζας Μέρος 3 ο : 8 ο μάθημα Εκτός ύλης ΔΠΘ-ΜΠΔ Συστήματα Βιομηχανικών Διεργασιών
Συστήματα Βιομηχανικών Διεργασιών 6ο εξάμηνο Μέρος 1 ο : Εισαγωγικά (διαστ., πυκν., θερμ., πίεση, κτλ.) Μέρος 2 ο : Ισοζύγια μάζας Μέρος 3 ο : 8 ο μάθημα Εκτός ύλης ΔΠΘ-ΜΠΔ Συστήματα Βιομηχανικών Διεργασιών
Statistics 104: Quantitative Methods for Economics Formula and Theorem Review
 Harvard College Statistics 104: Quantitative Methods for Economics Formula and Theorem Review Tommy MacWilliam, 13 tmacwilliam@college.harvard.edu March 10, 2011 Contents 1 Introduction to Data 5 1.1 Sample
Harvard College Statistics 104: Quantitative Methods for Economics Formula and Theorem Review Tommy MacWilliam, 13 tmacwilliam@college.harvard.edu March 10, 2011 Contents 1 Introduction to Data 5 1.1 Sample
ΠΟΛΥΤΕΧΝΕΙΟ ΚΡΗΤΗΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΕΡΙΒΑΛΛΟΝΤΟΣ
 ΠΟΛΥΤΕΧΝΕΙΟ ΚΡΗΤΗΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΕΡΙΒΑΛΛΟΝΤΟΣ Τομέας Περιβαλλοντικής Υδραυλικής και Γεωπεριβαλλοντικής Μηχανικής (III) Εργαστήριο Γεωπεριβαλλοντικής Μηχανικής TECHNICAL UNIVERSITY OF CRETE SCHOOL of
ΠΟΛΥΤΕΧΝΕΙΟ ΚΡΗΤΗΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΕΡΙΒΑΛΛΟΝΤΟΣ Τομέας Περιβαλλοντικής Υδραυλικής και Γεωπεριβαλλοντικής Μηχανικής (III) Εργαστήριο Γεωπεριβαλλοντικής Μηχανικής TECHNICAL UNIVERSITY OF CRETE SCHOOL of
ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ
 ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΣΧΟΛΗ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΜΗΧΑΝΙΚΩΝ ΥΠΟΛΟΓΙΣΤΩΝ ΤΟΜΕΑΣ ΤΕΧΝΟΛΟΓΙΑΣ ΠΛΗΡΟΦΟΡΙΚΗΣ ΚΑΙ ΥΠΟΛΟΓΙΣΤΩΝ Σημασιολογική Συσταδοποίηση Αντικειμένων Με Χρήση Οντολογικών Περιγραφών.
ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΣΧΟΛΗ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΜΗΧΑΝΙΚΩΝ ΥΠΟΛΟΓΙΣΤΩΝ ΤΟΜΕΑΣ ΤΕΧΝΟΛΟΓΙΑΣ ΠΛΗΡΟΦΟΡΙΚΗΣ ΚΑΙ ΥΠΟΛΟΓΙΣΤΩΝ Σημασιολογική Συσταδοποίηση Αντικειμένων Με Χρήση Οντολογικών Περιγραφών.
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ ΥΓΕΙΑΣ
 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ ΥΓΕΙΑΣ Πτυχιακή Εργασία "Η ΣΗΜΑΝΤΙΚΟΤΗΤΑ ΤΟΥ ΜΗΤΡΙΚΟΥ ΘΗΛΑΣΜΟΥ ΣΤΗ ΠΡΟΛΗΨΗ ΤΗΣ ΠΑΙΔΙΚΗΣ ΠΑΧΥΣΑΡΚΙΑΣ" Ειρήνη Σωτηρίου Λεμεσός 2014 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ ΥΓΕΙΑΣ Πτυχιακή Εργασία "Η ΣΗΜΑΝΤΙΚΟΤΗΤΑ ΤΟΥ ΜΗΤΡΙΚΟΥ ΘΗΛΑΣΜΟΥ ΣΤΗ ΠΡΟΛΗΨΗ ΤΗΣ ΠΑΙΔΙΚΗΣ ΠΑΧΥΣΑΡΚΙΑΣ" Ειρήνη Σωτηρίου Λεμεσός 2014 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ
CH 3 CH 2 COOCH 2 CH 2 CH 3 + H 2 O
 Q1.An experiment was carried out to determine the equilibrium constant, K c, for the following reaction. CH 3 CH 2 COOH + CH 3 CH 2 CH 2 OH CH 3 CH 2 COOCH 2 CH 2 CH 3 + H 2 O A student added measured
Q1.An experiment was carried out to determine the equilibrium constant, K c, for the following reaction. CH 3 CH 2 COOH + CH 3 CH 2 CH 2 OH CH 3 CH 2 COOCH 2 CH 2 CH 3 + H 2 O A student added measured
Studies on the Binding Mechanism of Several Antibiotics and Human Serum Albumin
 2005 63 Vol. 63, 2005 23, 2169 2173 ACTA CHIMICA SINICA No. 23, 2169 2173 a,b a a a *,a ( a 130012) ( b 133002), 26 K A 1.98 10 4, 1.01 10 3, 1.38 10 3, 5.97 10 4 7.15 10 4 L mol 1, n 1.16, 0.86, 1.19,
2005 63 Vol. 63, 2005 23, 2169 2173 ACTA CHIMICA SINICA No. 23, 2169 2173 a,b a a a *,a ( a 130012) ( b 133002), 26 K A 1.98 10 4, 1.01 10 3, 1.38 10 3, 5.97 10 4 7.15 10 4 L mol 1, n 1.16, 0.86, 1.19,
Potential Dividers. 46 minutes. 46 marks. Page 1 of 11
 Potential Dividers 46 minutes 46 marks Page 1 of 11 Q1. In the circuit shown in the figure below, the battery, of negligible internal resistance, has an emf of 30 V. The pd across the lamp is 6.0 V and
Potential Dividers 46 minutes 46 marks Page 1 of 11 Q1. In the circuit shown in the figure below, the battery, of negligible internal resistance, has an emf of 30 V. The pd across the lamp is 6.0 V and
P É Ô Ô² 1,2,.. Ò± 1,.. ±μ 1,. ƒ. ±μ μ 1,.Š. ±μ μ 1, ˆ.. Ê Ò 1,.. Ê Ò 1 Œˆ ˆŸ. ² μ Ê ² μ Ì μ ÉÓ. É μ ±, Ì μé μ Ò É μ Ò ² μ Ö
 P11-2015-60. É Ô Ô² 1,2,.. Ò± 1,.. ±μ 1,. ƒ. ±μ μ 1,.Š. ±μ μ 1, ˆ.. Ê Ò 1,.. Ê Ò 1 Œ Œ ˆ Š Œ ˆ ˆ Œˆ ˆŸ ƒ Š ˆŒ Š ² μ Ê ² μ Ì μ ÉÓ. É μ ±, Ì μé μ Ò É μ Ò ² μ Ö 1 Ñ Ò É ÉÊÉ Ö ÒÌ ² μ, Ê 2 Œμ μ²ó ± μ Ê É Ò
P11-2015-60. É Ô Ô² 1,2,.. Ò± 1,.. ±μ 1,. ƒ. ±μ μ 1,.Š. ±μ μ 1, ˆ.. Ê Ò 1,.. Ê Ò 1 Œ Œ ˆ Š Œ ˆ ˆ Œˆ ˆŸ ƒ Š ˆŒ Š ² μ Ê ² μ Ì μ ÉÓ. É μ ±, Ì μé μ Ò É μ Ò ² μ Ö 1 Ñ Ò É ÉÊÉ Ö ÒÌ ² μ, Ê 2 Œμ μ²ó ± μ Ê É Ò
Finite Field Problems: Solutions
 Finite Field Problems: Solutions 1. Let f = x 2 +1 Z 11 [x] and let F = Z 11 [x]/(f), a field. Let Solution: F =11 2 = 121, so F = 121 1 = 120. The possible orders are the divisors of 120. Solution: The
Finite Field Problems: Solutions 1. Let f = x 2 +1 Z 11 [x] and let F = Z 11 [x]/(f), a field. Let Solution: F =11 2 = 121, so F = 121 1 = 120. The possible orders are the divisors of 120. Solution: The
Study of In-vehicle Sound Field Creation by Simultaneous Equation Method
 Study of In-vehicle Sound Field Creation by Simultaneous Equation Method Kensaku FUJII Isao WAKABAYASI Tadashi UJINO Shigeki KATO Abstract FUJITSU TEN Limited has developed "TOYOTA remium Sound System"
Study of In-vehicle Sound Field Creation by Simultaneous Equation Method Kensaku FUJII Isao WAKABAYASI Tadashi UJINO Shigeki KATO Abstract FUJITSU TEN Limited has developed "TOYOTA remium Sound System"
Other Test Constructions: Likelihood Ratio & Bayes Tests
 Other Test Constructions: Likelihood Ratio & Bayes Tests Side-Note: So far we have seen a few approaches for creating tests such as Neyman-Pearson Lemma ( most powerful tests of H 0 : θ = θ 0 vs H 1 :
Other Test Constructions: Likelihood Ratio & Bayes Tests Side-Note: So far we have seen a few approaches for creating tests such as Neyman-Pearson Lemma ( most powerful tests of H 0 : θ = θ 0 vs H 1 :
Supporting Information
 Supporting Information rigin of the Regio- and Stereoselectivity of Allylic Substitution of rganocopper Reagents Naohiko Yoshikai, Song-Lin Zhang, and Eiichi Nakamura* Department of Chemistry, The University
Supporting Information rigin of the Regio- and Stereoselectivity of Allylic Substitution of rganocopper Reagents Naohiko Yoshikai, Song-Lin Zhang, and Eiichi Nakamura* Department of Chemistry, The University
Supporting Information
 Supporting Information On the Ambiguity of 1,3,2-Benzodiazaboroles as Donor/Acceptor unctionalities in Luminescent Molecules Lothar Weber *[a], Johannes Halama [a], Kenny Hanke [a], Lena Böhling [a], Andreas
Supporting Information On the Ambiguity of 1,3,2-Benzodiazaboroles as Donor/Acceptor unctionalities in Luminescent Molecules Lothar Weber *[a], Johannes Halama [a], Kenny Hanke [a], Lena Böhling [a], Andreas
Supplementary!Information!
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015! Synthesis)and)characterisation)of)an)open1cage) fullerene)encapsulating)hydrogen)fluoride! Supplementary!Information!
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015! Synthesis)and)characterisation)of)an)open1cage) fullerene)encapsulating)hydrogen)fluoride! Supplementary!Information!
6.3 Forecasting ARMA processes
 122 CHAPTER 6. ARMA MODELS 6.3 Forecasting ARMA processes The purpose of forecasting is to predict future values of a TS based on the data collected to the present. In this section we will discuss a linear
122 CHAPTER 6. ARMA MODELS 6.3 Forecasting ARMA processes The purpose of forecasting is to predict future values of a TS based on the data collected to the present. In this section we will discuss a linear
Μηχανική Μάθηση Hypothesis Testing
 ΕΛΛΗΝΙΚΗ ΔΗΜΟΚΡΑΤΙΑ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΡΗΤΗΣ Μηχανική Μάθηση Hypothesis Testing Γιώργος Μπορμπουδάκης Τμήμα Επιστήμης Υπολογιστών Procedure 1. Form the null (H 0 ) and alternative (H 1 ) hypothesis 2. Consider
ΕΛΛΗΝΙΚΗ ΔΗΜΟΚΡΑΤΙΑ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΡΗΤΗΣ Μηχανική Μάθηση Hypothesis Testing Γιώργος Μπορμπουδάκης Τμήμα Επιστήμης Υπολογιστών Procedure 1. Form the null (H 0 ) and alternative (H 1 ) hypothesis 2. Consider
Μεταπτυχιακή διατριβή. Ανδρέας Παπαευσταθίου
 Σχολή Γεωτεχνικών Επιστημών και Διαχείρισης Περιβάλλοντος Μεταπτυχιακή διατριβή Κτίρια σχεδόν μηδενικής ενεργειακής κατανάλωσης :Αξιολόγηση συστημάτων θέρμανσης -ψύξης και ΑΠΕ σε οικιστικά κτίρια στην
Σχολή Γεωτεχνικών Επιστημών και Διαχείρισης Περιβάλλοντος Μεταπτυχιακή διατριβή Κτίρια σχεδόν μηδενικής ενεργειακής κατανάλωσης :Αξιολόγηση συστημάτων θέρμανσης -ψύξης και ΑΠΕ σε οικιστικά κτίρια στην
[1] P Q. Fig. 3.1
![[1] P Q. Fig. 3.1 [1] P Q. Fig. 3.1](/thumbs/79/80362156.jpg) 1 (a) Define resistance....... [1] (b) The smallest conductor within a computer processing chip can be represented as a rectangular block that is one atom high, four atoms wide and twenty atoms long. One
1 (a) Define resistance....... [1] (b) The smallest conductor within a computer processing chip can be represented as a rectangular block that is one atom high, four atoms wide and twenty atoms long. One
ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ
 ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΣΧΟΛΗ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΜΗΧΑΝΙΚΩΝ ΥΠΟΛΟΓΙΣΤΩΝ ΤΟΜΕΑΣ ΗΛΕΚΤΡΟΜΑΓΝΗΤΙΚΩΝ ΕΦΑΡΜΟΓΩΝ ΗΛΕΚΤΡΟΟΠΤΙΚΗΣ & ΗΛΕΚΤΡΟΝΙΚΩΝ ΥΛΙΚΩΝ Μελέτη Επίδρασης Υπεριώδους Ακτινοβολίας σε Λεπτά
ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΣΧΟΛΗ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΜΗΧΑΝΙΚΩΝ ΥΠΟΛΟΓΙΣΤΩΝ ΤΟΜΕΑΣ ΗΛΕΚΤΡΟΜΑΓΝΗΤΙΚΩΝ ΕΦΑΡΜΟΓΩΝ ΗΛΕΚΤΡΟΟΠΤΙΚΗΣ & ΗΛΕΚΤΡΟΝΙΚΩΝ ΥΛΙΚΩΝ Μελέτη Επίδρασης Υπεριώδους Ακτινοβολίας σε Λεπτά
: Monte Carlo EM 313, Louis (1982) EM, EM Newton-Raphson, /. EM, 2 Monte Carlo EM Newton-Raphson, Monte Carlo EM, Monte Carlo EM, /. 3, Monte Carlo EM
 2008 6 Chinese Journal of Applied Probability and Statistics Vol.24 No.3 Jun. 2008 Monte Carlo EM 1,2 ( 1,, 200241; 2,, 310018) EM, E,,. Monte Carlo EM, EM E Monte Carlo,. EM, Monte Carlo EM,,,,. Newton-Raphson.
2008 6 Chinese Journal of Applied Probability and Statistics Vol.24 No.3 Jun. 2008 Monte Carlo EM 1,2 ( 1,, 200241; 2,, 310018) EM, E,,. Monte Carlo EM, EM E Monte Carlo,. EM, Monte Carlo EM,,,,. Newton-Raphson.
ZLW Series. Single-stage Monoblock Centrifugal Pump ZL PUMP GROUP.,LTD
 ZLW Series Single-stage Monoblock Centrifugal Pump ZL PUMP GROUP.,LTD 1 Application Apply as the transportation of liquids in the fields of air condition, heating, sanitary water, water treatment cooling,
ZLW Series Single-stage Monoblock Centrifugal Pump ZL PUMP GROUP.,LTD 1 Application Apply as the transportation of liquids in the fields of air condition, heating, sanitary water, water treatment cooling,
ΠΕΡΙΕΧΟΜΕΝΑ. Κεφάλαιο 1: Κεφάλαιο 2: Κεφάλαιο 3:
 4 Πρόλογος Η παρούσα διπλωµατική εργασία µε τίτλο «ιερεύνηση χωρικής κατανοµής µετεωρολογικών µεταβλητών. Εφαρµογή στον ελληνικό χώρο», ανατέθηκε από το ιεπιστηµονικό ιατµηµατικό Πρόγραµµα Μεταπτυχιακών
4 Πρόλογος Η παρούσα διπλωµατική εργασία µε τίτλο «ιερεύνηση χωρικής κατανοµής µετεωρολογικών µεταβλητών. Εφαρµογή στον ελληνικό χώρο», ανατέθηκε από το ιεπιστηµονικό ιατµηµατικό Πρόγραµµα Μεταπτυχιακών
DETERMINATION OF THERMAL PERFORMANCE OF GLAZED LIQUID HEATING SOLAR COLLECTORS
 ΕΘΝΙΚΟ ΚΕΝΤΡΟ ΕΡΕΥΝΑΣ ΦΥΣΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΗΜΟΚΡΙΤΟΣ / DEMOKRITOS NATIONAL CENTER FOR SCIENTIFIC RESEARCH ΕΡΓΑΣΤΗΡΙΟ ΟΚΙΜΩΝ ΗΛΙΑΚΩΝ & ΑΛΛΩΝ ΕΝΕΡΓΕΙΑΚΩΝ ΣΥΣΤΗΜΑΤΩΝ LABORATORY OF TESTIN SOLAR & OTHER ENERY
ΕΘΝΙΚΟ ΚΕΝΤΡΟ ΕΡΕΥΝΑΣ ΦΥΣΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΗΜΟΚΡΙΤΟΣ / DEMOKRITOS NATIONAL CENTER FOR SCIENTIFIC RESEARCH ΕΡΓΑΣΤΗΡΙΟ ΟΚΙΜΩΝ ΗΛΙΑΚΩΝ & ΑΛΛΩΝ ΕΝΕΡΓΕΙΑΚΩΝ ΣΥΣΤΗΜΑΤΩΝ LABORATORY OF TESTIN SOLAR & OTHER ENERY
Lowara SPECIFICATIONS
 SH Series Centrifugal pumps entirely made of AISI 36 stainless steel according to EN 733 (ex DIN 24255). Designed to pump hot, cold and moderately aggressive liquids. Available versions: SHE Close-coupled
SH Series Centrifugal pumps entirely made of AISI 36 stainless steel according to EN 733 (ex DIN 24255). Designed to pump hot, cold and moderately aggressive liquids. Available versions: SHE Close-coupled
Supporting information. An unusual bifunctional Tb-MOF for highly sensing of Ba 2+ ions and remarkable selectivities of CO 2 /N 2 and CO 2 /CH 4
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Supporting information An unusual bifunctional Tb-MOF for highly sensing
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Supporting information An unusual bifunctional Tb-MOF for highly sensing
CHAPTER 25 SOLVING EQUATIONS BY ITERATIVE METHODS
 CHAPTER 5 SOLVING EQUATIONS BY ITERATIVE METHODS EXERCISE 104 Page 8 1. Find the positive root of the equation x + 3x 5 = 0, correct to 3 significant figures, using the method of bisection. Let f(x) =
CHAPTER 5 SOLVING EQUATIONS BY ITERATIVE METHODS EXERCISE 104 Page 8 1. Find the positive root of the equation x + 3x 5 = 0, correct to 3 significant figures, using the method of bisection. Let f(x) =
Supporting Information
 Supporting Information Prediction of Novel High-Pressure Structures of Magnesium Niobium Dihydride Chuanzhao Zhang,,, Guoliang Sun, Jingjing Wang, Cheng Lu,*,, Yuanyuan Jin, Xiaoyu Kuang,*, and Andreas
Supporting Information Prediction of Novel High-Pressure Structures of Magnesium Niobium Dihydride Chuanzhao Zhang,,, Guoliang Sun, Jingjing Wang, Cheng Lu,*,, Yuanyuan Jin, Xiaoyu Kuang,*, and Andreas
5.4 The Poisson Distribution.
 The worst thing you can do about a situation is nothing. Sr. O Shea Jackson 5.4 The Poisson Distribution. Description of the Poisson Distribution Discrete probability distribution. The random variable
The worst thing you can do about a situation is nothing. Sr. O Shea Jackson 5.4 The Poisson Distribution. Description of the Poisson Distribution Discrete probability distribution. The random variable
Math 6 SL Probability Distributions Practice Test Mark Scheme
 Math 6 SL Probability Distributions Practice Test Mark Scheme. (a) Note: Award A for vertical line to right of mean, A for shading to right of their vertical line. AA N (b) evidence of recognizing symmetry
Math 6 SL Probability Distributions Practice Test Mark Scheme. (a) Note: Award A for vertical line to right of mean, A for shading to right of their vertical line. AA N (b) evidence of recognizing symmetry
ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΣΧΟΛΗ ΑΓΡΟΝΟΜΩΝ & ΤΟΠΟΓΡΑΦΩΝ ΜΗΧΑΝΙΚΩΝ ΤΟΜΕΑΣ ΕΡΓΩΝ ΥΠΟΔΟΜΗΣ ΚΑΙ ΑΓΡΟΤΙΚΗΣ ΑΝΑΠΤΥΞΗΣ ΤΟΜΕΑΣ ΓΕΝΙΚΗΣ ΓΕΩΔΑΙΣΙΑΣ
 ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΣΧΟΛΗ ΑΓΡΟΝΟΜΩΝ & ΤΟΠΟΓΡΑΦΩΝ ΜΗΧΑΝΙΚΩΝ ΤΟΜΕΑΣ ΕΡΓΩΝ ΥΠΟΔΟΜΗΣ ΚΑΙ ΑΓΡΟΤΙΚΗΣ ΑΝΑΠΤΥΞΗΣ ΤΟΜΕΑΣ ΓΕΝΙΚΗΣ ΓΕΩΔΑΙΣΙΑΣ ΔΙΑΧΕΙΡΙΣΗ ΠΕΡΙΤΥΠΩΜΑΤΩΝ ΣΕ ΣΙΔΗΡΟΔΡΟΜΙΚΕΣ ΔΙΑΒΑΣΕΙΣ ΔΙΠΛΩΜΑΤΙΚΗ
ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΣΧΟΛΗ ΑΓΡΟΝΟΜΩΝ & ΤΟΠΟΓΡΑΦΩΝ ΜΗΧΑΝΙΚΩΝ ΤΟΜΕΑΣ ΕΡΓΩΝ ΥΠΟΔΟΜΗΣ ΚΑΙ ΑΓΡΟΤΙΚΗΣ ΑΝΑΠΤΥΞΗΣ ΤΟΜΕΑΣ ΓΕΝΙΚΗΣ ΓΕΩΔΑΙΣΙΑΣ ΔΙΑΧΕΙΡΙΣΗ ΠΕΡΙΤΥΠΩΜΑΤΩΝ ΣΕ ΣΙΔΗΡΟΔΡΟΜΙΚΕΣ ΔΙΑΒΑΣΕΙΣ ΔΙΠΛΩΜΑΤΙΚΗ
Supplementary Appendix
 Supplementary Appendix Measuring crisis risk using conditional copulas: An empirical analysis of the 2008 shipping crisis Sebastian Opitz, Henry Seidel and Alexander Szimayer Model specification Table
Supplementary Appendix Measuring crisis risk using conditional copulas: An empirical analysis of the 2008 shipping crisis Sebastian Opitz, Henry Seidel and Alexander Szimayer Model specification Table
UDZ Swirl diffuser. Product facts. Quick-selection. Swirl diffuser UDZ. Product code example:
 UDZ Swirl diffuser Swirl diffuser UDZ, which is intended for installation in a ventilation duct, can be used in premises with a large volume, for example factory premises, storage areas, superstores, halls,
UDZ Swirl diffuser Swirl diffuser UDZ, which is intended for installation in a ventilation duct, can be used in premises with a large volume, for example factory premises, storage areas, superstores, halls,
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ ΥΓΕΙΑΣ ΤΜΗΜΑ ΝΟΣΗΛΕΥΤΙΚΗΣ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΕΠΗΡΕΑΖΕΙ ΤΗΝ ΠΡΟΛΗΨΗ ΚΑΡΚΙΝΟΥ ΤΟΥ ΜΑΣΤΟΥ
 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ ΥΓΕΙΑΣ ΤΜΗΜΑ ΝΟΣΗΛΕΥΤΙΚΗΣ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΠΩΣ Η ΚΑΤΑΝΑΛΩΣΗ ΦΡΟΥΤΩΝ ΚΑΙ ΛΑΧΑΝΙΚΩΝ ΕΠΗΡΕΑΖΕΙ ΤΗΝ ΠΡΟΛΗΨΗ ΚΑΡΚΙΝΟΥ ΤΟΥ ΜΑΣΤΟΥ Όνομα φοιτήτριας ΚΑΛΑΠΟΔΑ ΜΑΡΚΕΛΛΑ
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ ΥΓΕΙΑΣ ΤΜΗΜΑ ΝΟΣΗΛΕΥΤΙΚΗΣ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΠΩΣ Η ΚΑΤΑΝΑΛΩΣΗ ΦΡΟΥΤΩΝ ΚΑΙ ΛΑΧΑΝΙΚΩΝ ΕΠΗΡΕΑΖΕΙ ΤΗΝ ΠΡΟΛΗΨΗ ΚΑΡΚΙΝΟΥ ΤΟΥ ΜΑΣΤΟΥ Όνομα φοιτήτριας ΚΑΛΑΠΟΔΑ ΜΑΡΚΕΛΛΑ
HOMEWORK 4 = G. In order to plot the stress versus the stretch we define a normalized stretch:
 HOMEWORK 4 Problem a For the fast loading case, we want to derive the relationship between P zz and λ z. We know that the nominal stress is expressed as: P zz = ψ λ z where λ z = λ λ z. Therefore, applying
HOMEWORK 4 Problem a For the fast loading case, we want to derive the relationship between P zz and λ z. We know that the nominal stress is expressed as: P zz = ψ λ z where λ z = λ λ z. Therefore, applying
ΠΣΤΥΙΑΚΗ ΔΡΓΑΙΑ. Μειέηε Υξόλνπ Απνζηείξσζεο Κνλζέξβαο κε Τπνινγηζηηθή Ρεπζηνδπλακηθή. Αζαλαζηάδνπ Βαξβάξα
 ΣΔΥΝΟΛΟΓΙΚΟ ΔΚΠΑΙΓΔΤΣΙΚΟ ΙΓΡΤΜΑ ΘΔΑΛΟΝΙΚΗ ΥΟΛΗ ΣΔΥΝΟΛΟΓΙΑ ΣΡΟΦΙΜΩΝ & ΓΙΑΣΡΟΦΗ ΣΜΗΜΑ ΣΔΥΝΟΛΟΓΙΑ ΣΡΟΦΙΜΩΝ ΠΣΤΥΙΑΚΗ ΔΡΓΑΙΑ Μειέηε Υξόλνπ Απνζηείξσζεο Κνλζέξβαο κε Τπνινγηζηηθή Ρεπζηνδπλακηθή Αζαλαζηάδνπ Βαξβάξα
ΣΔΥΝΟΛΟΓΙΚΟ ΔΚΠΑΙΓΔΤΣΙΚΟ ΙΓΡΤΜΑ ΘΔΑΛΟΝΙΚΗ ΥΟΛΗ ΣΔΥΝΟΛΟΓΙΑ ΣΡΟΦΙΜΩΝ & ΓΙΑΣΡΟΦΗ ΣΜΗΜΑ ΣΔΥΝΟΛΟΓΙΑ ΣΡΟΦΙΜΩΝ ΠΣΤΥΙΑΚΗ ΔΡΓΑΙΑ Μειέηε Υξόλνπ Απνζηείξσζεο Κνλζέξβαο κε Τπνινγηζηηθή Ρεπζηνδπλακηθή Αζαλαζηάδνπ Βαξβάξα
Electronic Supplementary Information DFT Characterization on the Mechanism of Water Splitting Catalyzed by Single-Ru-substituted Polyoxometalates
 Electronic Supplementary Information DFT Characterization on the Mechanism of Water Splitting Catalyzed by Single-Ru-substituted Polyoxometalates Zhong-Ling Lang, Guo-Chun Yang, Na-Na Ma, Shi-Zheng Wen,
Electronic Supplementary Information DFT Characterization on the Mechanism of Water Splitting Catalyzed by Single-Ru-substituted Polyoxometalates Zhong-Ling Lang, Guo-Chun Yang, Na-Na Ma, Shi-Zheng Wen,
An experimental and theoretical study of the gas phase kinetics of atomic chlorine reactions with CH 3 NH 2, (CH 3 ) 2 NH, and (CH 3 ) 3 N
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2015 An experimental and theoretical study of the gas phase kinetics of atomic chlorine
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2015 An experimental and theoretical study of the gas phase kinetics of atomic chlorine
ΔΙΕΡΕΥΝΗΣΗ ΤΗΣ ΣΕΞΟΥΑΛΙΚΗΣ ΔΡΑΣΤΗΡΙΟΤΗΤΑΣ ΤΩΝ ΓΥΝΑΙΚΩΝ ΚΑΤΑ ΤΗ ΔΙΑΡΚΕΙΑ ΤΗΣ ΕΓΚΥΜΟΣΥΝΗΣ ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ ΥΓΕΙΑΣ
 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ ΥΓΕΙΑΣ Πτυχιακή Εργασία ΔΙΕΡΕΥΝΗΣΗ ΤΗΣ ΣΕΞΟΥΑΛΙΚΗΣ ΔΡΑΣΤΗΡΙΟΤΗΤΑΣ ΤΩΝ ΓΥΝΑΙΚΩΝ ΚΑΤΑ ΤΗ ΔΙΑΡΚΕΙΑ ΤΗΣ ΕΓΚΥΜΟΣΥΝΗΣ ΑΝΔΡΕΟΥ ΣΤΕΦΑΝΙΑ Λεμεσός 2012 i ii ΤΕΧΝΟΛΟΓΙΚΟ
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ ΥΓΕΙΑΣ Πτυχιακή Εργασία ΔΙΕΡΕΥΝΗΣΗ ΤΗΣ ΣΕΞΟΥΑΛΙΚΗΣ ΔΡΑΣΤΗΡΙΟΤΗΤΑΣ ΤΩΝ ΓΥΝΑΙΚΩΝ ΚΑΤΑ ΤΗ ΔΙΑΡΚΕΙΑ ΤΗΣ ΕΓΚΥΜΟΣΥΝΗΣ ΑΝΔΡΕΟΥ ΣΤΕΦΑΝΙΑ Λεμεσός 2012 i ii ΤΕΧΝΟΛΟΓΙΚΟ
Zebra reaction or the recipe for heterodimeric zinc complexes synthesis
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 05 Supporting Information Zebra reaction or the recipe for heterodimeric zinc complexes synthesis
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 05 Supporting Information Zebra reaction or the recipe for heterodimeric zinc complexes synthesis
Stress Relaxation Test and Constitutive Equation of Saturated Soft Soil
 8 7 011 7 Journal of Highway and Transportation Research and Development Vol. 8 No. 7 Jul. 011 100-068 011 07-0014 - 05 1 1. 0009. 710064 k 0 Merchant 4 Merchant U416. 1 + 6 A Stress Relaxation Test and
8 7 011 7 Journal of Highway and Transportation Research and Development Vol. 8 No. 7 Jul. 011 100-068 011 07-0014 - 05 1 1. 0009. 710064 k 0 Merchant 4 Merchant U416. 1 + 6 A Stress Relaxation Test and
; +302 ; +313; +320,.
 1.,,*+, - +./ +/2 +, -. ; +, - +* cm : Key words: snow-water content, surface soil, snow type, water permeability, water retention +,**. +,,**/.. +30- +302 ; +302 ; +313; +320,. + *+, *2// + -.*, **. **+.,
1.,,*+, - +./ +/2 +, -. ; +, - +* cm : Key words: snow-water content, surface soil, snow type, water permeability, water retention +,**. +,,**/.. +30- +302 ; +302 ; +313; +320,. + *+, *2// + -.*, **. **+.,
EE512: Error Control Coding
 EE512: Error Control Coding Solution for Assignment on Finite Fields February 16, 2007 1. (a) Addition and Multiplication tables for GF (5) and GF (7) are shown in Tables 1 and 2. + 0 1 2 3 4 0 0 1 2 3
EE512: Error Control Coding Solution for Assignment on Finite Fields February 16, 2007 1. (a) Addition and Multiplication tables for GF (5) and GF (7) are shown in Tables 1 and 2. + 0 1 2 3 4 0 0 1 2 3
«ΑΝΑΠΣΤΞΖ ΓΠ ΚΑΗ ΥΩΡΗΚΖ ΑΝΑΛΤΖ ΜΔΣΔΩΡΟΛΟΓΗΚΩΝ ΓΔΓΟΜΔΝΩΝ ΣΟΝ ΔΛΛΑΓΗΚΟ ΥΩΡΟ»
 ΓΔΩΠΟΝΗΚΟ ΠΑΝΔΠΗΣΖΜΗΟ ΑΘΖΝΩΝ ΣΜΗΜΑ ΑΞΙΟΠΟΙΗΗ ΦΤΙΚΩΝ ΠΟΡΩΝ & ΓΕΩΡΓΙΚΗ ΜΗΥΑΝΙΚΗ ΣΟΜΕΑ ΕΔΑΦΟΛΟΓΙΑ ΚΑΙ ΓΕΩΡΓΙΚΗ ΥΗΜΕΙΑ ΕΙΔΙΚΕΤΗ: ΕΦΑΡΜΟΓΕ ΣΗ ΓΕΩΠΛΗΡΟΦΟΡΙΚΗ ΣΟΤ ΦΤΙΚΟΤ ΠΟΡΟΤ «ΑΝΑΠΣΤΞΖ ΓΠ ΚΑΗ ΥΩΡΗΚΖ ΑΝΑΛΤΖ ΜΔΣΔΩΡΟΛΟΓΗΚΩΝ
ΓΔΩΠΟΝΗΚΟ ΠΑΝΔΠΗΣΖΜΗΟ ΑΘΖΝΩΝ ΣΜΗΜΑ ΑΞΙΟΠΟΙΗΗ ΦΤΙΚΩΝ ΠΟΡΩΝ & ΓΕΩΡΓΙΚΗ ΜΗΥΑΝΙΚΗ ΣΟΜΕΑ ΕΔΑΦΟΛΟΓΙΑ ΚΑΙ ΓΕΩΡΓΙΚΗ ΥΗΜΕΙΑ ΕΙΔΙΚΕΤΗ: ΕΦΑΡΜΟΓΕ ΣΗ ΓΕΩΠΛΗΡΟΦΟΡΙΚΗ ΣΟΤ ΦΤΙΚΟΤ ΠΟΡΟΤ «ΑΝΑΠΣΤΞΖ ΓΠ ΚΑΗ ΥΩΡΗΚΖ ΑΝΑΛΤΖ ΜΔΣΔΩΡΟΛΟΓΗΚΩΝ
