Comparison of experimental and estimated polarizabilities for organic compounds using ThermoML Archive data
|
|
|
- Ἀγαυή Ταρσούλη
- 6 χρόνια πριν
- Προβολές:
Transcript
1 Comparison of experimental and estimated polarizabilities for organic compounds using ThermoML Archive data Axel Drefahl Internet: We compare values for the experimentally derived and estimated molecular polarizability for compounds that have not been used in the original design of the estimation method. The compounds along with mass density, ρ, and refractive index (Na-D line), n D, have been extracted from the ThermoML Archive (status: February, 2007). Experimental polarizability values are those calculated with the Lorentz- Lorenz (LL) equation from observed data for the liquid density, ρ, and the refractive index (Na-D line), n D : α LL = n2 D 1 M n 2 D + 2 ρ (1) Estimated polarizability values are those estimated the atom additivity (AA) approach of Bosque and Sales (J.Chem.Inf.Comput.Sci.2003,42, ): α AA = Σc A N A (2) where c A is the contribution for atom with element symbol A and N A is number of respective atoms per molecule. A can be C, H, O, S, N, P, F, Cl, Br, and I. The summation is over all element symbols Pearson s correlation coefficient values are those estimat R = Σ(d AAd LL ) (Σd 2 AA Σd2 LL )1/2 (3) where d AA = α AA,i ᾱ AA and d LL = α LL,i ᾱ LL. Each summation is done over all i pairs. The pairs (α AA, α LL ) are listed in the following for 20 and 25 C ordered by molecular formula. The result: R = for the data at 20 C and R = for the data 25 C. 1
2 Compounds with experimental polarizabilities at 20 C: C2H2Br4 1,1,2,2-tetrabromoethane [ ] jct/2006v38/i01/j.jct α AA = 16.85, α LL = C2H2Br4 1,1,2,2-tetrabromoethane [ ] jct/2006v38/i09/2006alhalb0 α AA = 16.85, α LL = C2H2Cl4 1,1,1,2-tetrachloroethane [ ] fpe/2006v249/i01/j.fluid α AA = 12.33, α LL = C2H3Cl3 trichloroethane [ ] fpe/2006v249/i01/j.fluid α AA = 10.34, α LL = C3H5Cl 3-chloro-1-propene [ ] jced/2003v48/i04/je034018b α AA = 7.88, α LL = 8.11 C5H10O 2-ethoxypropene [ ] jct/2006v38/i07/j.jct α AA = 10.18, α LL = C5H10O 3-methyl-2-butanone [ ] jced/2006v51/i05/je060025z.xml α AA = 10.18, α LL = C5H10O2 tetrahydrofurfuryl alcohol [ ] jced/2005v50/i02/je049605r α AA = 10.75, α LL = C5H12O 2-methyl-1-butanol [ ] jced/2004v49/i06/je030216r α AA = 10.53, α LL =
3 C6H10O4 dimethyl succinate [ ] jct/2007v39/i02/j.jct α AA = 13.40, α LL = C6H12O2 ethyl butanoate [ ] fpe/2005v234/i01/j.fluid α AA = 12.61, α LL = C7H12O4 dimethyl glutarate [ ] jct/2007v39/i02/j.jct xml α AA = 15.26, α LL = C7H14O 5-methyl-2-hexanone [ ] fpe/2006v250/i01/j.fluid α AA = 13.89, α LL = C7H16O3 ethyl orthoformate [ ] jct/2004v36/i11/j.jct α AA = 15.38, α LL = C8H14O2 cyclohexyl acetate [ ] jced/2006v51/i05/je060025z α AA = 15.98, α LL = C8H14O2 cyclohexyl acetate [ ] jct/2005v37/i02/j.jct α AA = 15.98, α LL = C8H14O2 cyclohexyl acetate [ ] jced/2005v50/i02/je049605r α AA = 15.98, α LL = C8H14O4 dimethyl adipate [ ] jct/2007v39/i02/j.jct α AA = 17.12, α LL =
4 C8H14O4 dimethyl adipate [ ] fpe/2005v230/i01/2005inc0 α AA = 17.12, α LL = C8H16O 6-methyl-3-heptanone [ ] fpe/2006v250/i01/j.fluid α AA = 15.75, α LL = C9H12 propylbenzene [ ] jced/2006v51/i01/je α AA = 16.00, α LL = C9H20O 1-nonanol [ ] jced/2006v51/i05/je060025z α AA = 17.96, α LL = C10H14 butylbenzene [ ] jced/2006v51/i01/je α AA = 17.85, α LL = C12H14O4 diethyl phthalate [ ] jct/2006v38/i11/2006xuliu0 α AA = 23.16, α LL = C12H14O4 diethyl phthalate [ ] jct/2005v37/i10/j.jct α AA = 23.16, α LL = C10H16.alpha.-pinene [ ] jced/2003v48/i05/je025631i α AA = 18.20, α LL = C10H16.alpha.-pinene [ ] jct/2005v37/i03/j.jct α AA = 18.20, α LL = C18H36O 4
5 (Z)-9-octadecen-1-ol [ ] jced/2006v51/i05/je060025z α AA = 34.33, α LL = C18H36O (Z)-9-octadecen-1-ol [ ] fpe/2006v250/i01/j.fluid α AA = 34.33, α LL = C24H51N trioctylamine [ ] jced/2006v51/i05/je060025z α AA = 46.46, α LL = C28H46O4 1,2-benzenedicarboxylic acid, diisodecyl ester [ ] jced/2005v50/i06/je050151n.xml α AA = 52.89, α LL = Compounds with experimental polarizabilities at 25 C: C4H9Cl 2-chlorobutane [ ] jct/2007v39/i01/j.jct α AA = 10.08, α LL = C4H9Cl isobutyl chloride [ ] jct/2007v39/i01/j.jct α AA = 10.08, α LL = C4H9Cl tert-butyl chloride [ ] jct/2007v39/i01/j.jct α AA = 10.08, α LL = C4H12N2O 2-((2-aminoethyl)amino)ethanol [ ] jced/2006v51/i04/je060032n α AA = 11.08, α LL = C5H8O2 vinyl propionate [ ] jced/2005v50/i02/je049909d.xml α AA = 10.40, α LL =
6 C5H10Cl2 1,5-dichloropentane [ ] jct/2005v37/i12/j.jct α AA = 13.93, α LL = C5H10O 3-methyl-2-butanone [ ] jced/2005v50/i04/je050062a α AA = 10.18, α LL = C5H10O2 butyl formate [ ] jct/2006v38/i09/2006ortmar0 α AA = 10.75, α LL = C5H10O3 ethyl lactate [ ] jced/2006v51/i04/je060052p α AA = 11.32, α LL = C5H12O 2-methyl-1-butanol [ ] jced/2006v51/i05/je060238o α AA = 10.53, α LL = C5H12O 2-methyl-1-butanol [ ] jced/2005v50/i05/je α AA = 10.53, α LL = C5H12O 2-methyl-1-butanol [ ] jced/2006v51/i02/je α AA = 10.53, α LL = C5H12O 2-methyl-1-butanol [ ] jced/2006v51/i01/je α AA = 10.53, α LL = C5H12O 3-methyl-2-butanol [ ] jced/2005v50/i04/je050062a α AA = 10.53, α LL =
7 C5H12O2 2,2-dimethoxypropane [ ] jced/2005v50/i06/je050013y α AA = 11.10, α LL = C6H5Cl2OP phenylphosphonic dichloride [ ] jced/2004v49/i03/je034263v α AA = 17.62, α LL = C6H5F fluorobenzene [ ] tca/2005v439/i01/j.tca α AA = 10.47, α LL = C6H10O4 diethyl oxalate [ ] jced/2003v48/i06/je α AA = 13.40, α LL = C6H10O4 diethyl oxalate [ ] jced/2005v50/i03/je049610v α AA = 13.40, α LL = C6H12Cl2 1,6-dichlorohexane [ ] jct/2005v37/i12/j.jct α AA = 15.79, α LL = C6H12O2 ethyl butanoate [ ] jct/2005v37/i12/j.jct α AA = 12.61, α LL = C6H12O2 propyl propanoate [ ] jced/2006v51/i01/je α AA = 12.61, α LL = C6H12O2 isobutyl ethanoate [ ] fpe/2005v232/i01/2005munmon0 α AA = 12.61, α LL = C6H12O2 7
8 tert-butyl ethanoate [ ] fpe/2005v227/i01/2005monmun0 α AA = 12.61, α LL = C6H12O3 paraldehyde [ ] jced/2006v51/i01/je050183a α AA = 13.18, α LL = C6H13Cl 1-chlorohexane [ ] jct/2006v38/i07/j.jct α AA = 13.80, α LL = C6H13Cl 1-chlorohexane [ ] tca/2005v426/i01/2005oswgar0 α AA = 13.80, α LL = C6H14 2-methylpentane [ ] jced/2005v50/i04/je050062a α AA = 11.81, α LL = C6H14O2 3,6-dioxaoctane [ ] tca/2006v447/i02/2006fracom0 α AA = 12.96, α LL = C7H12O4 dimethyl glutarate [ ] jced/2005v50/i05/je049571n α AA = 15.26, α LL = C7H14O2 butyl propanoate [ ] jced/2006v51/i06/je060314n α AA = 14.47, α LL = C7H14O2 butyl propanoate [ ] fpe/2005v238/i01/j.fluid α AA = 14.47, α LL = C7H14O2 butyl propanoate [ ] 8
9 jct/2006v38/i09/2006ortmar0 α AA = 14.47, α LL = C7H14O2 ethyl pentanoate [ ] jct/2005v37/i12/j.jct α AA = 14.47, α LL = C7H14O2 ethyl isovalerate [ ] jced/2006v51/i05/je060139a α AA = 14.47, α LL = C7H14O2 ethyl isovalerate [ ] α AA = 14.47, α LL = C7H16O butane, 2-ethoxy-2-methyl- [ ] fpe/2005v233/i01/j.fluid α AA = 14.24, α LL = C7H16O butane, 2-ethoxy-2-methyl- [ ] jced/2004v49/i03/je α AA = 14.24, α LL = C8H8O2 methyl benzoate [ ] jced/2006v51/i05/je060139a α AA = 14.93, α LL = C8H8O2 methyl benzoate [ ] α AA = 14.93, α LL = C8H11N DL-1-phenylethylamine [ ] jced/2005v50/i01/je α AA = 15.34, α LL = C8H16O2 methyl heptanoate [ ] jct/2007v39/i02/j.jct
10 α AA = 16.32, α LL = C8H16O2 ethyl caproate [ ] α AA = 16.32, α LL = C8H16O2 ethyl caproate [ ] jced/2006v51/i05/je α AA = 16.32, α LL = C8H16O2 butyl butanoate [ ] jct/2006v38/i09/2006ortmar0 α AA = 16.32, α LL = C8H18O butyl tert-butyl ether [ ] tca/2005v435/i02/2005arcarc0 α AA = 16.10, α LL = C8H18O4 triglyme [ ] jced/2004v49/i02/je α AA = 17.81, α LL = C9H10O2 benzyl acetate [ ] jced/2006v51/i05/je060139a α AA = 16.79, α LL = C9H10O2 benzyl acetate [ ] α AA = 16.79, α LL = C9H18O2 isopentyl butanoate [ ] α AA = 18.18, α LL = C9H18O2 isopentyl butanoate [ ] jced/2006v51/i05/je α AA = 18.18, α LL =
11 C10H12O2 benzyl propanoate [ ] jced/2006v51/i05/je060139a α AA = 18.65, α LL = C10H12O2 benzyl propanoate [ ] α AA = 18.65, α LL = C10H12O2 ethyl phenylethanoate [ ] α AA = 18.65, α LL = C10H12O2 ethyl phenylethanoate [ ] jced/2006v51/i05/je α AA = 18.65, α LL = C10H14 butylbenzene [ ] jct/2005v37/i09/j.jct α AA = 17.85, α LL = C10H18 trans-decalin [ ] fpe/2006v243/i01/j.fluid α AA = 18.55, α LL = C10H18O dl-2,6-dimethyl-2,7-octadien-6-ol [ ] fpe/2005v238/i01/j.fluid α AA = 19.12, α LL = C10H20O2 ethyl octanoate [ ] α AA = 20.04, α LL = C10H20O2 ethyl octanoate [ ] jced/2006v51/i05/je α AA = 20.04, α LL =
12 C10H22O5 dimethoxytetraglycol [ ] jced/2004v49/i02/je α AA = 22.10, α LL = C12H14O4 diethyl phthalate [ ] jced/2003v48/i06/je α AA = 23.16, α LL = C14H30 tetradecane [ ] jct/2006v38/i08/2006mutman0 α AA = 26.68, α LL = C14H30 tetradecane [ ] jced/2003v48/i06/je030147g α AA = 26.68, α LL = C24H38O4 dioctyl phthalate [ ] jced/2003v48/i06/je α AA = 45.45, α LL = C30H62 2,6,10,15,19,23-hexamethyltetracosane [ ] ijt/2005v26/i03/2005tri0 α AA = 56.41, α LL = This document is supplementing the paper entitled Extraction and Application of Environmentally Relevant Chemical Information from the ThermoML Archive ( submitted to the EnviroInfo 2007, Sept in Poland, Warsaw 2007 Axel Drefahl 12
Chemical Name Index. Chemical Name Index 459
 459 Primary chemical names used in the tables together with other (alternative and trade) names are ordered alphabetically in this index. Chemical Abstracts Service Registry Numbers (CASRN) are provided
459 Primary chemical names used in the tables together with other (alternative and trade) names are ordered alphabetically in this index. Chemical Abstracts Service Registry Numbers (CASRN) are provided
Universal Solvent GC Method Table
 Acetone 67-64-1 OVM-3500/3520 ORBO 91 (CT) Acetonitrile 75-05-8 OVM-3500/3520 Acrylonitrile 107-13-1 OVM-3500/3520 Allyl Alcohol 107-18-6 OVM-3500/3520 6μg Allyl Chloride 107-05-1 226-09 Amyl Acetate (mixed
Acetone 67-64-1 OVM-3500/3520 ORBO 91 (CT) Acetonitrile 75-05-8 OVM-3500/3520 Acrylonitrile 107-13-1 OVM-3500/3520 Allyl Alcohol 107-18-6 OVM-3500/3520 6μg Allyl Chloride 107-05-1 226-09 Amyl Acetate (mixed
Technical Information T-9100 SI. Suva. refrigerants. Thermodynamic Properties of. Suva Refrigerant [R-410A (50/50)]
![Technical Information T-9100 SI. Suva. refrigerants. Thermodynamic Properties of. Suva Refrigerant [R-410A (50/50)] Technical Information T-9100 SI. Suva. refrigerants. Thermodynamic Properties of. Suva Refrigerant [R-410A (50/50)]](/thumbs/89/98928029.jpg) d Suva refrigerants Technical Information T-9100SI Thermodynamic Properties of Suva 9100 Refrigerant [R-410A (50/50)] Thermodynamic Properties of Suva 9100 Refrigerant SI Units New tables of the thermodynamic
d Suva refrigerants Technical Information T-9100SI Thermodynamic Properties of Suva 9100 Refrigerant [R-410A (50/50)] Thermodynamic Properties of Suva 9100 Refrigerant SI Units New tables of the thermodynamic
Supplementary Material for Statistical Field Theory for Polar Fluids
 Supplementary Material for Statistical Field Theory for Polar Fluids Bilin Zhuang 1, 2 and Zhen-Gang Wang 1, 1 Division of Chemistry and Chemical Engineering, California Institute of Technology, Pasadena,
Supplementary Material for Statistical Field Theory for Polar Fluids Bilin Zhuang 1, 2 and Zhen-Gang Wang 1, 1 Division of Chemistry and Chemical Engineering, California Institute of Technology, Pasadena,
DuPont Suva. DuPont. Thermodynamic Properties of. Refrigerant (R-410A) Technical Information. refrigerants T-410A ENG
 Technical Information T-410A ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 410A Refrigerant (R-410A) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Technical Information T-410A ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 410A Refrigerant (R-410A) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
DuPont Suva 95 Refrigerant
 Technical Information T-95 SI DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Technical Information T-95 SI DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
DuPont Suva 95 Refrigerant
 Technical Information T-95 ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Technical Information T-95 ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Chemical Name Index 443
 Chemical Name Index Primary chemical names used in the tables together with other (alternative and trade) names are ordered alphabetically in this index. Chemical Abstracts Service Registry Numbers (CASRN)
Chemical Name Index Primary chemical names used in the tables together with other (alternative and trade) names are ordered alphabetically in this index. Chemical Abstracts Service Registry Numbers (CASRN)
Selecting Critical Properties of Terpenes and Terpenoids through Group-Contribution Methods and Equations of State
 Supporting Information Selecting Critical Properties of Terpenes and Terpenoids through Group-Contribution Methods and Equations of State Mónia A. R. Martins, 1,2 Pedro J. Carvalho, 1 André M. Palma, 1
Supporting Information Selecting Critical Properties of Terpenes and Terpenoids through Group-Contribution Methods and Equations of State Mónia A. R. Martins, 1,2 Pedro J. Carvalho, 1 André M. Palma, 1
ELECTRONIC SUPPLEMENTARY MATERIAL. Figure 1S.- Chemical structure of: a) alizarin; b) purpurin; c) pseudopurpurin; d) xanthopurpurin; e) quinizarin.
 ELECTRNIC SUPPLEMENTARY MATERIAL Figure 1S.- Chemical structure of: a) alizarin; b) purpurin; c) pseudopurpurin; d) xanthopurpurin; e) quinizarin. R 1 R 2 R 3 R 4 Name R 1 R 2 R 3 R 4 a Alizarin H H H
ELECTRNIC SUPPLEMENTARY MATERIAL Figure 1S.- Chemical structure of: a) alizarin; b) purpurin; c) pseudopurpurin; d) xanthopurpurin; e) quinizarin. R 1 R 2 R 3 R 4 Name R 1 R 2 R 3 R 4 a Alizarin H H H
PROPERTIES OF COMMON LABORATORY SOLVENTS
 PROPERTIES OF COMMON LABORATORY SOLVENTS This table give properties of 200 organic solvents which are frequently used in laboratory and industrial applications. Compounds are listed in alphabetical order
PROPERTIES OF COMMON LABORATORY SOLVENTS This table give properties of 200 organic solvents which are frequently used in laboratory and industrial applications. Compounds are listed in alphabetical order
Copper-catalyzed formal O-H insertion reaction of α-diazo-1,3-dicarb- onyl compounds to carboxylic acids with the assistance of isocyanide
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Copper-catalyzed formal O-H insertion reaction of α-diazo-1,3-dicarb- onyl compounds to carboxylic
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Copper-catalyzed formal O-H insertion reaction of α-diazo-1,3-dicarb- onyl compounds to carboxylic
Mean bond enthalpy Standard enthalpy of formation Bond N H N N N N H O O O
 Q1. (a) Explain the meaning of the terms mean bond enthalpy and standard enthalpy of formation. Mean bond enthalpy... Standard enthalpy of formation... (5) (b) Some mean bond enthalpies are given below.
Q1. (a) Explain the meaning of the terms mean bond enthalpy and standard enthalpy of formation. Mean bond enthalpy... Standard enthalpy of formation... (5) (b) Some mean bond enthalpies are given below.
CONCENTRATIVE PROPERTIES OF AQUEOUS SOLUTIONS: DENSITY, REFRACTIVE INDEX, FREEZING POINT DEPRESSION, AND VISCOSITY
 DENSITY, REFRACTIVE INDEX, FREEZING POINT DEPRESSION, AND VISCOSITY This table gives properties of aqueous solutions of 66 substances as a function of concentration. All data refer to a temperature of
DENSITY, REFRACTIVE INDEX, FREEZING POINT DEPRESSION, AND VISCOSITY This table gives properties of aqueous solutions of 66 substances as a function of concentration. All data refer to a temperature of
derivation of the Laplacian from rectangular to spherical coordinates
 derivation of the Laplacian from rectangular to spherical coordinates swapnizzle 03-03- :5:43 We begin by recognizing the familiar conversion from rectangular to spherical coordinates (note that φ is used
derivation of the Laplacian from rectangular to spherical coordinates swapnizzle 03-03- :5:43 We begin by recognizing the familiar conversion from rectangular to spherical coordinates (note that φ is used
GC-MS 84.22% % Aroma components analysis in blackcurrant fruit and fruit wine by GC-MS
 2014 33 3 265 141 GC-MS 1 1 * 2 2 1. 730070 2. 100029 - HS-SPME - GC-MS 1399 50 84.22% 2- -3- -2-4- -1-48 92.65% 3- -1- - - TS207.3 doi 10.3969/j.issn.0254-5071.2014.03.034 A 0254-50712014 03-0141-06 Aroma
2014 33 3 265 141 GC-MS 1 1 * 2 2 1. 730070 2. 100029 - HS-SPME - GC-MS 1399 50 84.22% 2- -3- -2-4- -1-48 92.65% 3- -1- - - TS207.3 doi 10.3969/j.issn.0254-5071.2014.03.034 A 0254-50712014 03-0141-06 Aroma
Approximation of distance between locations on earth given by latitude and longitude
 Approximation of distance between locations on earth given by latitude and longitude Jan Behrens 2012-12-31 In this paper we shall provide a method to approximate distances between two points on earth
Approximation of distance between locations on earth given by latitude and longitude Jan Behrens 2012-12-31 In this paper we shall provide a method to approximate distances between two points on earth
difluoroboranyls derived from amides carrying donor group Supporting Information
 The influence of the π-conjugated spacer on photophysical properties of difluoroboranyls derived from amides carrying donor group Supporting Information Anna Maria Grabarz a Adèle D. Laurent b, Beata Jędrzejewska
The influence of the π-conjugated spacer on photophysical properties of difluoroboranyls derived from amides carrying donor group Supporting Information Anna Maria Grabarz a Adèle D. Laurent b, Beata Jędrzejewska
Supporting Information
 Supporting Information for AgOTf-catalyzed one-pot reactions of 2-alkynylbenzaldoximes with α,β-unsaturated carbonyl compounds Qiuping Ding 1, Dan Wang 1, Puying Luo* 2, Meiling Liu 1, Shouzhi Pu* 3 and
Supporting Information for AgOTf-catalyzed one-pot reactions of 2-alkynylbenzaldoximes with α,β-unsaturated carbonyl compounds Qiuping Ding 1, Dan Wang 1, Puying Luo* 2, Meiling Liu 1, Shouzhi Pu* 3 and
Divergent synthesis of various iminocyclitols from D-ribose
 Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 205 Divergent synthesis of various iminocyclitols from D-ribose Ramu Petakamsetty,
Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 205 Divergent synthesis of various iminocyclitols from D-ribose Ramu Petakamsetty,
Molecular Basis of Olfactory Chemoreception in the Common Bed Bug, Cimex lectularius. Feng Liu, Zhou Chen, Nannan Liu
 Molecular Basis of Olfactory Chemoreception in the Common Bed Bug, Cimex lectularius Feng Liu, Zhou Chen, Nannan Liu Supplement Information Figure legend Figure S1. Representative response traces of OR-expressed
Molecular Basis of Olfactory Chemoreception in the Common Bed Bug, Cimex lectularius Feng Liu, Zhou Chen, Nannan Liu Supplement Information Figure legend Figure S1. Representative response traces of OR-expressed
Enantioselective Organocatalytic Michael Addition of Isorhodanines. to α, β-unsaturated Aldehydes
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2016 Enantioselective Organocatalytic Michael Addition of Isorhodanines to α,
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2016 Enantioselective Organocatalytic Michael Addition of Isorhodanines to α,
A facile and general route to 3-((trifluoromethyl)thio)benzofurans and 3-((trifluoromethyl)thio)benzothiophenes
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 A facile and general route to 3-((trifluoromethyl)thio)benzofurans and 3-((trifluoromethyl)thio)benzothiophenes
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 A facile and general route to 3-((trifluoromethyl)thio)benzofurans and 3-((trifluoromethyl)thio)benzothiophenes
Si + Al Mg Fe + Mn +Ni Ca rim Ca p.f.u
 .6.5. y = -.4x +.8 R =.9574 y = - x +.14 R =.9788 y = -.4 x +.7 R =.9896 Si + Al Fe + Mn +Ni y =.55 x.36 R =.9988.149.148.147.146.145..88 core rim.144 4 =.6 ±.6 4 =.6 ±.18.84.88 p.f.u..86.76 y = -3.9 x
.6.5. y = -.4x +.8 R =.9574 y = - x +.14 R =.9788 y = -.4 x +.7 R =.9896 Si + Al Fe + Mn +Ni y =.55 x.36 R =.9988.149.148.147.146.145..88 core rim.144 4 =.6 ±.6 4 =.6 ±.18.84.88 p.f.u..86.76 y = -3.9 x
Patrycja Miszczyk, Dorota Wieczorek, Joanna Gałęzowska, Błażej Dziuk, Joanna Wietrzyk and Ewa Chmielewska. 1. Spectroscopic Data.
 ; doi:10.3390/molecules22020254 S1 of S23 Supplementary Materials: Reaction of 3-Amino-1,2,4-Triazole with Diethyl Phosphite and Triethyl Orthoformate: Acid-Base Properties and Antiosteoporotic Activities
; doi:10.3390/molecules22020254 S1 of S23 Supplementary Materials: Reaction of 3-Amino-1,2,4-Triazole with Diethyl Phosphite and Triethyl Orthoformate: Acid-Base Properties and Antiosteoporotic Activities
Supplemental Materials
 Supplemental Materials Fuel Specific Influences on the Composition of Reaction Intermediates in Premixed Flames of Three C 5 H 10 O 2 Ester Isomers Bin Yang, Charles K. Westbrook, Terrill A. Cool, Nils
Supplemental Materials Fuel Specific Influences on the Composition of Reaction Intermediates in Premixed Flames of Three C 5 H 10 O 2 Ester Isomers Bin Yang, Charles K. Westbrook, Terrill A. Cool, Nils
The Free Internet Journal for Organic Chemistry
 The Free Internet Journal for Organic Chemistry Paper Archive for Organic Chemistry Arkivoc 2018, part iii, S1-S6 Synthesis of dihydropyranones and dihydropyrano[2,3- d][1,3]dioxine-diones by cyclization
The Free Internet Journal for Organic Chemistry Paper Archive for Organic Chemistry Arkivoc 2018, part iii, S1-S6 Synthesis of dihydropyranones and dihydropyrano[2,3- d][1,3]dioxine-diones by cyclization
The effect of curcumin on the stability of Aβ. dimers
 The effect of curcumin on the stability of Aβ dimers Li Na Zhao, See-Wing Chiu, Jérôme Benoit, Lock Yue Chew,, and Yuguang Mu, School of Physical and Mathematical Sciences, Nanyang Technological University,
The effect of curcumin on the stability of Aβ dimers Li Na Zhao, See-Wing Chiu, Jérôme Benoit, Lock Yue Chew,, and Yuguang Mu, School of Physical and Mathematical Sciences, Nanyang Technological University,
Η ΕΠΙΔΡΑΣΗ ΤΗΣ ΑΙΘΑΝΟΛΗΣ,ΤΗΣ ΜΕΘΑΝΟΛΗΣ ΚΑΙ ΤΟΥ ΑΙΘΥΛΟΤΡΙΤΟΤΑΓΗ ΒΟΥΤΥΛΑΙΘΕΡΑ ΣΤΙΣ ΙΔΙΟΤΗΤΕΣ ΤΗΣ ΒΕΝΖΙΝΗΣ
 ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙΔΕΥΤΙΚΟ ΙΔΡΥΜΑ ΚΑΒΑΛΑΣ ΣΧΟΛΗ ΤΕΧΝΟΛΟΓΙΚΩΝ ΕΦΑΡΜΟΓΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ Η ΕΠΙΔΡΑΣΗ ΤΗΣ ΑΙΘΑΝΟΛΗΣ,ΤΗΣ ΜΕΘΑΝΟΛΗΣ ΚΑΙ ΤΟΥ ΑΙΘΥΛΟΤΡΙΤΟΤΑΓΗ ΒΟΥΤΥΛΑΙΘΕΡΑ ΣΤΙΣ ΙΔΙΟΤΗΤΕΣ ΤΗΣ ΒΕΝΖΙΝΗΣ ΟΝΟΜΑΤΕΠΩΝΥΜΟ
ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙΔΕΥΤΙΚΟ ΙΔΡΥΜΑ ΚΑΒΑΛΑΣ ΣΧΟΛΗ ΤΕΧΝΟΛΟΓΙΚΩΝ ΕΦΑΡΜΟΓΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ Η ΕΠΙΔΡΑΣΗ ΤΗΣ ΑΙΘΑΝΟΛΗΣ,ΤΗΣ ΜΕΘΑΝΟΛΗΣ ΚΑΙ ΤΟΥ ΑΙΘΥΛΟΤΡΙΤΟΤΑΓΗ ΒΟΥΤΥΛΑΙΘΕΡΑ ΣΤΙΣ ΙΔΙΟΤΗΤΕΣ ΤΗΣ ΒΕΝΖΙΝΗΣ ΟΝΟΜΑΤΕΠΩΝΥΜΟ
6.1. Dirac Equation. Hamiltonian. Dirac Eq.
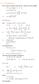 6.1. Dirac Equation Ref: M.Kaku, Quantum Field Theory, Oxford Univ Press (1993) η μν = η μν = diag(1, -1, -1, -1) p 0 = p 0 p = p i = -p i p μ p μ = p 0 p 0 + p i p i = E c 2 - p 2 = (m c) 2 H = c p 2
6.1. Dirac Equation Ref: M.Kaku, Quantum Field Theory, Oxford Univ Press (1993) η μν = η μν = diag(1, -1, -1, -1) p 0 = p 0 p = p i = -p i p μ p μ = p 0 p 0 + p i p i = E c 2 - p 2 = (m c) 2 H = c p 2
Direct Transformation of Ethylarenes into Primary Aromatic Amides with N-Bromosuccinimide and I 2 -aq NH 3
 Supporting Information Direct Transformation of Ethylarenes into Primary Aromatic Amides with N-Bromosuccinimide and I 2 -aq NH 3 Shohei Shimokawa, Yuhsuke Kawagoe, Katsuhiko Moriyama, Hideo Togo* Graduate
Supporting Information Direct Transformation of Ethylarenes into Primary Aromatic Amides with N-Bromosuccinimide and I 2 -aq NH 3 Shohei Shimokawa, Yuhsuke Kawagoe, Katsuhiko Moriyama, Hideo Togo* Graduate
Supporting Information. Experimental section
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Experimental section General. Proton nuclear magnetic resonance ( 1
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Experimental section General. Proton nuclear magnetic resonance ( 1
Metal-free Oxidative Coupling of Amines with Sodium Sulfinates: A Mild Access to Sulfonamides
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting information for Metal-free Oxidative Coupling of Amines with Sodium Sulfinates:
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting information for Metal-free Oxidative Coupling of Amines with Sodium Sulfinates:
1 h, , CaCl 2. pelamis) 58.1%, (Headspace solid -phase microextraction and gas chromatography -mass spectrometry,hs -SPME - Vol. 15 No.
 2 0 1 5 7 Journal of Chinese Institute of Food Science and Technology Vol. 15 No. 7 Jul. 2 0 1 5 1,2 1 1 1 1* ( 1 315211 2 315502) : ( ) : (E-Nose), - : GC-MS, 20.98% 2.5%, 53.15% 11.2%, 0. 51% 71.86%
2 0 1 5 7 Journal of Chinese Institute of Food Science and Technology Vol. 15 No. 7 Jul. 2 0 1 5 1,2 1 1 1 1* ( 1 315211 2 315502) : ( ) : (E-Nose), - : GC-MS, 20.98% 2.5%, 53.15% 11.2%, 0. 51% 71.86%
Facile construction of the functionalized 4H-chromene via tandem. benzylation and cyclization. Jinmin Fan and Zhiyong Wang*
 Facile construction of the functionalized 4H-chromene via tandem benzylation and cyclization Jinmin Fan and Zhiyong Wang* Hefei National Laboratory for Physical Science at Microscale, Joint- Lab of Green
Facile construction of the functionalized 4H-chromene via tandem benzylation and cyclization Jinmin Fan and Zhiyong Wang* Hefei National Laboratory for Physical Science at Microscale, Joint- Lab of Green
Supporting Information-B. A Facile Iterative Synthesis of 2,5-Terpyrimidinylenes as Non-peptidic α-helical Mimics
 Supporting Information-B A Facile Iterative Synthesis of 2,5-Terpyrimidinylenes as on-peptidic α-helical Mimics Laura Anderson, 1,2 Mingzhou Zhou, 1,2 Vasudha Sharma, 2 Jillian M. McLaughlin, 2 Daniel.
Supporting Information-B A Facile Iterative Synthesis of 2,5-Terpyrimidinylenes as on-peptidic α-helical Mimics Laura Anderson, 1,2 Mingzhou Zhou, 1,2 Vasudha Sharma, 2 Jillian M. McLaughlin, 2 Daniel.
The Simply Typed Lambda Calculus
 Type Inference Instead of writing type annotations, can we use an algorithm to infer what the type annotations should be? That depends on the type system. For simple type systems the answer is yes, and
Type Inference Instead of writing type annotations, can we use an algorithm to infer what the type annotations should be? That depends on the type system. For simple type systems the answer is yes, and
DESIGN OF MACHINERY SOLUTION MANUAL h in h 4 0.
 DESIGN OF MACHINERY SOLUTION MANUAL -7-1! PROBLEM -7 Statement: Design a double-dwell cam to move a follower from to 25 6, dwell for 12, fall 25 and dwell for the remader The total cycle must take 4 sec
DESIGN OF MACHINERY SOLUTION MANUAL -7-1! PROBLEM -7 Statement: Design a double-dwell cam to move a follower from to 25 6, dwell for 12, fall 25 and dwell for the remader The total cycle must take 4 sec
Supplementary Information 1.
 Supplementary Information 1. Fig. S1. Correlations between litter-derived-c and N (percent of initial input) and Al-/Fe- (hydr)oxides dissolved by ammonium oxalate (AO); a) 0 10 cm; b) 10 20 cm; c) 20
Supplementary Information 1. Fig. S1. Correlations between litter-derived-c and N (percent of initial input) and Al-/Fe- (hydr)oxides dissolved by ammonium oxalate (AO); a) 0 10 cm; b) 10 20 cm; c) 20
Electronic Supplementary Information
 Electronic Supplementary Information Unprecedented Carbon-Carbon Bond Cleavage in Nucleophilic Aziridine Ring Opening Reaction, Efficient Ring Transformation of Aziridines to Imidazolidin-4-ones Jin-Yuan
Electronic Supplementary Information Unprecedented Carbon-Carbon Bond Cleavage in Nucleophilic Aziridine Ring Opening Reaction, Efficient Ring Transformation of Aziridines to Imidazolidin-4-ones Jin-Yuan
Lewis Acid Catalyzed Propargylation of Arenes with O-Propargyl Trichloroacetimidate: Synthesis of 1,3-Diarylpropynes
 Supporting Information for Lewis Acid Catalyzed Propargylation of Arenes with O-Propargyl Trichloroacetimidate: Synthesis of 1,3-Diarylpropynes Changkun Li and Jianbo Wang* Beijing National Laboratory
Supporting Information for Lewis Acid Catalyzed Propargylation of Arenes with O-Propargyl Trichloroacetimidate: Synthesis of 1,3-Diarylpropynes Changkun Li and Jianbo Wang* Beijing National Laboratory
Math 6 SL Probability Distributions Practice Test Mark Scheme
 Math 6 SL Probability Distributions Practice Test Mark Scheme. (a) Note: Award A for vertical line to right of mean, A for shading to right of their vertical line. AA N (b) evidence of recognizing symmetry
Math 6 SL Probability Distributions Practice Test Mark Scheme. (a) Note: Award A for vertical line to right of mean, A for shading to right of their vertical line. AA N (b) evidence of recognizing symmetry
An experimental and theoretical study of the gas phase kinetics of atomic chlorine reactions with CH 3 NH 2, (CH 3 ) 2 NH, and (CH 3 ) 3 N
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2015 An experimental and theoretical study of the gas phase kinetics of atomic chlorine
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2015 An experimental and theoretical study of the gas phase kinetics of atomic chlorine
Srednicki Chapter 55
 Srednicki Chapter 55 QFT Problems & Solutions A. George August 3, 03 Srednicki 55.. Use equations 55.3-55.0 and A i, A j ] = Π i, Π j ] = 0 (at equal times) to verify equations 55.-55.3. This is our third
Srednicki Chapter 55 QFT Problems & Solutions A. George August 3, 03 Srednicki 55.. Use equations 55.3-55.0 and A i, A j ] = Π i, Π j ] = 0 (at equal times) to verify equations 55.-55.3. This is our third
Matrices and Determinants
 Matrices and Determinants SUBJECTIVE PROBLEMS: Q 1. For what value of k do the following system of equations possess a non-trivial (i.e., not all zero) solution over the set of rationals Q? x + ky + 3z
Matrices and Determinants SUBJECTIVE PROBLEMS: Q 1. For what value of k do the following system of equations possess a non-trivial (i.e., not all zero) solution over the set of rationals Q? x + ky + 3z
Supporting Information
 Supporting Information On the Ambiguity of 1,3,2-Benzodiazaboroles as Donor/Acceptor unctionalities in Luminescent Molecules Lothar Weber *[a], Johannes Halama [a], Kenny Hanke [a], Lena Böhling [a], Andreas
Supporting Information On the Ambiguity of 1,3,2-Benzodiazaboroles as Donor/Acceptor unctionalities in Luminescent Molecules Lothar Weber *[a], Johannes Halama [a], Kenny Hanke [a], Lena Böhling [a], Andreas
Aquinas College. Edexcel Mathematical formulae and statistics tables DO NOT WRITE ON THIS BOOKLET
 Aquinas College Edexcel Mathematical formulae and statistics tables DO NOT WRITE ON THIS BOOKLET Pearson Edexcel Level 3 Advanced Subsidiary and Advanced GCE in Mathematics and Further Mathematics Mathematical
Aquinas College Edexcel Mathematical formulae and statistics tables DO NOT WRITE ON THIS BOOKLET Pearson Edexcel Level 3 Advanced Subsidiary and Advanced GCE in Mathematics and Further Mathematics Mathematical
Lyotropic Liquid Crystals in Amino acid derived Protic Ionic Liquids: Physicochemical Properties and Behaviour as Amphiphile Self-Assembly Media
 Lyotropic Liquid Crystals in Amino acid derived Protic Ionic Liquids: Physicochemical Properties and Behaviour as Amphiphile Self-Assembly Media Jiayi Wang, [a,b] Tamar L. Greaves, [b] Danielle F. Kennedy,
Lyotropic Liquid Crystals in Amino acid derived Protic Ionic Liquids: Physicochemical Properties and Behaviour as Amphiphile Self-Assembly Media Jiayi Wang, [a,b] Tamar L. Greaves, [b] Danielle F. Kennedy,
Copper-Catalyzed Oxidative Dehydrogenative N-N Bond. Formation for the Synthesis of N,N -Diarylindazol-3-ones
 Electronic Supplementary Material (ESI) for Organic Chemistry Frontiers. This journal is the Partner Organisations 2016 Supporting information Copper-Catalyzed Oxidative Dehydrogenative - Bond Formation
Electronic Supplementary Material (ESI) for Organic Chemistry Frontiers. This journal is the Partner Organisations 2016 Supporting information Copper-Catalyzed Oxidative Dehydrogenative - Bond Formation
Exercises to Statistics of Material Fatigue No. 5
 Prof. Dr. Christine Müller Dipl.-Math. Christoph Kustosz Eercises to Statistics of Material Fatigue No. 5 E. 9 (5 a Show, that a Fisher information matri for a two dimensional parameter θ (θ,θ 2 R 2, can
Prof. Dr. Christine Müller Dipl.-Math. Christoph Kustosz Eercises to Statistics of Material Fatigue No. 5 E. 9 (5 a Show, that a Fisher information matri for a two dimensional parameter θ (θ,θ 2 R 2, can
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΓΕΩΠΟΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΒΙΟΤΕΧΝΟΛΟΓΙΑΣ ΚΑΙ ΕΠΙΣΤΗΜΗΣ ΤΡΟΦΙΜΩΝ. Πτυχιακή εργασία
 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΓΕΩΠΟΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΒΙΟΤΕΧΝΟΛΟΓΙΑΣ ΚΑΙ ΕΠΙΣΤΗΜΗΣ ΤΡΟΦΙΜΩΝ Πτυχιακή εργασία ΜΕΛΕΤΗ ΠΟΛΥΦΑΙΝΟΛΩΝ ΚΑΙ ΑΝΤΙΟΞΕΙΔΩΤΙΚΗΣ ΙΚΑΝΟΤΗΤΑΣ ΣΟΚΟΛΑΤΑΣ Αναστασία Σιάντωνα Λεμεσός
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΓΕΩΠΟΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΒΙΟΤΕΧΝΟΛΟΓΙΑΣ ΚΑΙ ΕΠΙΣΤΗΜΗΣ ΤΡΟΦΙΜΩΝ Πτυχιακή εργασία ΜΕΛΕΤΗ ΠΟΛΥΦΑΙΝΟΛΩΝ ΚΑΙ ΑΝΤΙΟΞΕΙΔΩΤΙΚΗΣ ΙΚΑΝΟΤΗΤΑΣ ΣΟΚΟΛΑΤΑΣ Αναστασία Σιάντωνα Λεμεσός
Other Test Constructions: Likelihood Ratio & Bayes Tests
 Other Test Constructions: Likelihood Ratio & Bayes Tests Side-Note: So far we have seen a few approaches for creating tests such as Neyman-Pearson Lemma ( most powerful tests of H 0 : θ = θ 0 vs H 1 :
Other Test Constructions: Likelihood Ratio & Bayes Tests Side-Note: So far we have seen a few approaches for creating tests such as Neyman-Pearson Lemma ( most powerful tests of H 0 : θ = θ 0 vs H 1 :
Solutions to the Schrodinger equation atomic orbitals. Ψ 1 s Ψ 2 s Ψ 2 px Ψ 2 py Ψ 2 pz
 Solutions to the Schrodinger equation atomic orbitals Ψ 1 s Ψ 2 s Ψ 2 px Ψ 2 py Ψ 2 pz ybridization Valence Bond Approach to bonding sp 3 (Ψ 2 s + Ψ 2 px + Ψ 2 py + Ψ 2 pz) sp 2 (Ψ 2 s + Ψ 2 px + Ψ 2 py)
Solutions to the Schrodinger equation atomic orbitals Ψ 1 s Ψ 2 s Ψ 2 px Ψ 2 py Ψ 2 pz ybridization Valence Bond Approach to bonding sp 3 (Ψ 2 s + Ψ 2 px + Ψ 2 py + Ψ 2 pz) sp 2 (Ψ 2 s + Ψ 2 px + Ψ 2 py)
Env-A 1400 Regulated Toxic Air Pollutant (RTAP) Analysis - STEP 1 - De Minimus Evaluation
 gal/yr lb/gal lb/day lb/yr HAP lb/day lb/yr conc. % to air % lb/day lb/yr lb/day lb/yr Fail free formaldehyde, max. 000050-00-0 VOC, HAP 0.000066 0.0165 100 100 0.00 0.02 0.01 3.8 pass ethanol 000064-17-5
gal/yr lb/gal lb/day lb/yr HAP lb/day lb/yr conc. % to air % lb/day lb/yr lb/day lb/yr Fail free formaldehyde, max. 000050-00-0 VOC, HAP 0.000066 0.0165 100 100 0.00 0.02 0.01 3.8 pass ethanol 000064-17-5
CE 530 Molecular Simulation
 C 53 olecular Siulation Lecture Histogra Reweighting ethods David. Kofke Departent of Cheical ngineering SUNY uffalo kofke@eng.buffalo.edu Histogra Reweighting ethod to cobine results taken at different
C 53 olecular Siulation Lecture Histogra Reweighting ethods David. Kofke Departent of Cheical ngineering SUNY uffalo kofke@eng.buffalo.edu Histogra Reweighting ethod to cobine results taken at different
ΠΕΡΙΕΧΟΜΕΝΑ. Κεφάλαιο 1: Κεφάλαιο 2: Κεφάλαιο 3:
 4 Πρόλογος Η παρούσα διπλωµατική εργασία µε τίτλο «ιερεύνηση χωρικής κατανοµής µετεωρολογικών µεταβλητών. Εφαρµογή στον ελληνικό χώρο», ανατέθηκε από το ιεπιστηµονικό ιατµηµατικό Πρόγραµµα Μεταπτυχιακών
4 Πρόλογος Η παρούσα διπλωµατική εργασία µε τίτλο «ιερεύνηση χωρικής κατανοµής µετεωρολογικών µεταβλητών. Εφαρµογή στον ελληνικό χώρο», ανατέθηκε από το ιεπιστηµονικό ιατµηµατικό Πρόγραµµα Μεταπτυχιακών
Partial Trace and Partial Transpose
 Partial Trace and Partial Transpose by José Luis Gómez-Muñoz http://homepage.cem.itesm.mx/lgomez/quantum/ jose.luis.gomez@itesm.mx This document is based on suggestions by Anirban Das Introduction This
Partial Trace and Partial Transpose by José Luis Gómez-Muñoz http://homepage.cem.itesm.mx/lgomez/quantum/ jose.luis.gomez@itesm.mx This document is based on suggestions by Anirban Das Introduction This
Supplementary Materials
 Supplementary Materials Figure S1. Chemical structures of phloracetophenone intermediates (2a, 2c 2g). 2a 2c 2d 2e 2f 2g Figure S2. Chemical structures of geranylacetophenone analogues (3a, 3c, 3e g).
Supplementary Materials Figure S1. Chemical structures of phloracetophenone intermediates (2a, 2c 2g). 2a 2c 2d 2e 2f 2g Figure S2. Chemical structures of geranylacetophenone analogues (3a, 3c, 3e g).
Finite Field Problems: Solutions
 Finite Field Problems: Solutions 1. Let f = x 2 +1 Z 11 [x] and let F = Z 11 [x]/(f), a field. Let Solution: F =11 2 = 121, so F = 121 1 = 120. The possible orders are the divisors of 120. Solution: The
Finite Field Problems: Solutions 1. Let f = x 2 +1 Z 11 [x] and let F = Z 11 [x]/(f), a field. Let Solution: F =11 2 = 121, so F = 121 1 = 120. The possible orders are the divisors of 120. Solution: The
EE512: Error Control Coding
 EE512: Error Control Coding Solution for Assignment on Finite Fields February 16, 2007 1. (a) Addition and Multiplication tables for GF (5) and GF (7) are shown in Tables 1 and 2. + 0 1 2 3 4 0 0 1 2 3
EE512: Error Control Coding Solution for Assignment on Finite Fields February 16, 2007 1. (a) Addition and Multiplication tables for GF (5) and GF (7) are shown in Tables 1 and 2. + 0 1 2 3 4 0 0 1 2 3
Mandelamide-Zinc Catalyzed Alkyne Addition to Heteroaromatic Aldehydes
 1 Mandelamide-Zinc Catalyzed Alkyne Addition to Heteroaromatic Aldehydes Gonzalo Blay, Isabel Fernández, Alícia Marco-Aleixandre, and José R. Pedro Departament de Química Orgànica, Facultat de Química,
1 Mandelamide-Zinc Catalyzed Alkyne Addition to Heteroaromatic Aldehydes Gonzalo Blay, Isabel Fernández, Alícia Marco-Aleixandre, and José R. Pedro Departament de Química Orgànica, Facultat de Química,
EPL 603 TOPICS IN SOFTWARE ENGINEERING. Lab 5: Component Adaptation Environment (COPE)
 EPL 603 TOPICS IN SOFTWARE ENGINEERING Lab 5: Component Adaptation Environment (COPE) Performing Static Analysis 1 Class Name: The fully qualified name of the specific class Type: The type of the class
EPL 603 TOPICS IN SOFTWARE ENGINEERING Lab 5: Component Adaptation Environment (COPE) Performing Static Analysis 1 Class Name: The fully qualified name of the specific class Type: The type of the class
Section 8.2 Graphs of Polar Equations
 Section 8. Graphs of Polar Equations Graphing Polar Equations The graph of a polar equation r = f(θ), or more generally F(r,θ) = 0, consists of all points P that have at least one polar representation
Section 8. Graphs of Polar Equations Graphing Polar Equations The graph of a polar equation r = f(θ), or more generally F(r,θ) = 0, consists of all points P that have at least one polar representation
athanasiadis@rhodes.aegean.gr , -.
 παιδαγωγικά ρεύµατα στο Αιγαίο Προσκήνιο 88 - * athanasiadis@rhodes.aegean.gr -., -.. Abstract The aim of this survey is to show how students of the three last school classes of the Primary School evaluated
παιδαγωγικά ρεύµατα στο Αιγαίο Προσκήνιο 88 - * athanasiadis@rhodes.aegean.gr -., -.. Abstract The aim of this survey is to show how students of the three last school classes of the Primary School evaluated
Lecture 2. Soundness and completeness of propositional logic
 Lecture 2 Soundness and completeness of propositional logic February 9, 2004 1 Overview Review of natural deduction. Soundness and completeness. Semantics of propositional formulas. Soundness proof. Completeness
Lecture 2 Soundness and completeness of propositional logic February 9, 2004 1 Overview Review of natural deduction. Soundness and completeness. Semantics of propositional formulas. Soundness proof. Completeness
Regioselectivity in the Stille coupling reactions of 3,5- dibromo-2-pyrone.
 Regioselectivity in the Stille coupling reactions of 3,5- dibromo-2-pyrone. Won-Suk Kim, Hyung-Jin Kim and Cheon-Gyu Cho Department of Chemistry, Hanyang University, Seoul 133-791, Korea Experimental Section
Regioselectivity in the Stille coupling reactions of 3,5- dibromo-2-pyrone. Won-Suk Kim, Hyung-Jin Kim and Cheon-Gyu Cho Department of Chemistry, Hanyang University, Seoul 133-791, Korea Experimental Section
Supporting Information
 Supporting Information Wiley-VC 007 9 Weinheim, Germany ew ear Infrared Dyes and Fluorophores Based on Diketopyrrolopyrroles Dipl.-Chem. Georg M. Fischer, Dipl.-Chem. Andreas P. Ehlers, Prof. Dr. Andreas
Supporting Information Wiley-VC 007 9 Weinheim, Germany ew ear Infrared Dyes and Fluorophores Based on Diketopyrrolopyrroles Dipl.-Chem. Georg M. Fischer, Dipl.-Chem. Andreas P. Ehlers, Prof. Dr. Andreas
Sulfonated graphene as highly efficient and reusable acid carbocatalyst for the synthesis of ester plasticizers
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 204 Supporting information for publication Sulfonated graphene as highly efficient and reusable
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 204 Supporting information for publication Sulfonated graphene as highly efficient and reusable
Section 9.2 Polar Equations and Graphs
 180 Section 9. Polar Equations and Graphs In this section, we will be graphing polar equations on a polar grid. In the first few examples, we will write the polar equation in rectangular form to help identify
180 Section 9. Polar Equations and Graphs In this section, we will be graphing polar equations on a polar grid. In the first few examples, we will write the polar equation in rectangular form to help identify
ΚΥΠΡΙΑΚΟΣ ΣΥΝΔΕΣΜΟΣ ΠΛΗΡΟΦΟΡΙΚΗΣ CYPRUS COMPUTER SOCIETY 21 ος ΠΑΓΚΥΠΡΙΟΣ ΜΑΘΗΤΙΚΟΣ ΔΙΑΓΩΝΙΣΜΟΣ ΠΛΗΡΟΦΟΡΙΚΗΣ Δεύτερος Γύρος - 30 Μαρτίου 2011
 Διάρκεια Διαγωνισμού: 3 ώρες Απαντήστε όλες τις ερωτήσεις Μέγιστο Βάρος (20 Μονάδες) Δίνεται ένα σύνολο από N σφαιρίδια τα οποία δεν έχουν όλα το ίδιο βάρος μεταξύ τους και ένα κουτί που αντέχει μέχρι
Διάρκεια Διαγωνισμού: 3 ώρες Απαντήστε όλες τις ερωτήσεις Μέγιστο Βάρος (20 Μονάδες) Δίνεται ένα σύνολο από N σφαιρίδια τα οποία δεν έχουν όλα το ίδιο βάρος μεταξύ τους και ένα κουτί που αντέχει μέχρι
Partial Differential Equations in Biology The boundary element method. March 26, 2013
 The boundary element method March 26, 203 Introduction and notation The problem: u = f in D R d u = ϕ in Γ D u n = g on Γ N, where D = Γ D Γ N, Γ D Γ N = (possibly, Γ D = [Neumann problem] or Γ N = [Dirichlet
The boundary element method March 26, 203 Introduction and notation The problem: u = f in D R d u = ϕ in Γ D u n = g on Γ N, where D = Γ D Γ N, Γ D Γ N = (possibly, Γ D = [Neumann problem] or Γ N = [Dirichlet
Nickel and Platinum PCP Pincer Complexes Incorporating an Acyclic Diaminoalkyl Central Moiety Connecting Imidazole or Pyrazole Rings
 ickel and Platinum PCP Pincer Complexes Incorporating an Acyclic Diaminoalkyl Central Moiety Connecting Imidazole or Pyrazole Rings Braulio M. Puerta Lombardi, Rudy M. Braun, Chris Gendy, Chia Yun Chang,
ickel and Platinum PCP Pincer Complexes Incorporating an Acyclic Diaminoalkyl Central Moiety Connecting Imidazole or Pyrazole Rings Braulio M. Puerta Lombardi, Rudy M. Braun, Chris Gendy, Chia Yun Chang,
CRITICAL CONSTANTS. References
 CRITICAL CONSTANTS The parameters of the liquid gas critical point are important constants in determining the behavior of fluids. This table lists the critical temperature, pressure, and molar volume,
CRITICAL CONSTANTS The parameters of the liquid gas critical point are important constants in determining the behavior of fluids. This table lists the critical temperature, pressure, and molar volume,
Supplementary Materials for. Kinetic and Computational Studies on Pd(I) Dimer- Mediated Halogen Exchange of Aryl Iodides
 Supplementary Materials for Kinetic and Computational Studies on Pd(I) Dimer- Mediated Halogen Exchange of Aryl Iodides Indrek Kalvet, a Karl J. Bonney, a and Franziska Schoenebeck a * a Institute of Organic
Supplementary Materials for Kinetic and Computational Studies on Pd(I) Dimer- Mediated Halogen Exchange of Aryl Iodides Indrek Kalvet, a Karl J. Bonney, a and Franziska Schoenebeck a * a Institute of Organic
Derivation of Optical-Bloch Equations
 Appendix C Derivation of Optical-Bloch Equations In this appendix the optical-bloch equations that give the populations and coherences for an idealized three-level Λ system, Fig. 3. on page 47, will be
Appendix C Derivation of Optical-Bloch Equations In this appendix the optical-bloch equations that give the populations and coherences for an idealized three-level Λ system, Fig. 3. on page 47, will be
Global Reference List of Chemically Defined Substances
 Visit www.iofi.org for the latest version of the IOFI Global Reference List! 1 Table of contents: Global Reference List of Chemically Defined Substances - p3 Global Reference List of Chemically Defined
Visit www.iofi.org for the latest version of the IOFI Global Reference List! 1 Table of contents: Global Reference List of Chemically Defined Substances - p3 Global Reference List of Chemically Defined
ΠΟΛΥΤΕΧΝΕΙΟ ΚΡΗΤΗΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΕΡΙΒΑΛΛΟΝΤΟΣ
 ΠΟΛΥΤΕΧΝΕΙΟ ΚΡΗΤΗΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΕΡΙΒΑΛΛΟΝΤΟΣ Τομέας Περιβαλλοντικής Υδραυλικής και Γεωπεριβαλλοντικής Μηχανικής (III) Εργαστήριο Γεωπεριβαλλοντικής Μηχανικής TECHNICAL UNIVERSITY OF CRETE SCHOOL of
ΠΟΛΥΤΕΧΝΕΙΟ ΚΡΗΤΗΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΕΡΙΒΑΛΛΟΝΤΟΣ Τομέας Περιβαλλοντικής Υδραυλικής και Γεωπεριβαλλοντικής Μηχανικής (III) Εργαστήριο Γεωπεριβαλλοντικής Μηχανικής TECHNICAL UNIVERSITY OF CRETE SCHOOL of
Απόκριση σε Μοναδιαία Ωστική Δύναμη (Unit Impulse) Απόκριση σε Δυνάμεις Αυθαίρετα Μεταβαλλόμενες με το Χρόνο. Απόστολος Σ.
 Απόκριση σε Δυνάμεις Αυθαίρετα Μεταβαλλόμενες με το Χρόνο The time integral of a force is referred to as impulse, is determined by and is obtained from: Newton s 2 nd Law of motion states that the action
Απόκριση σε Δυνάμεις Αυθαίρετα Μεταβαλλόμενες με το Χρόνο The time integral of a force is referred to as impulse, is determined by and is obtained from: Newton s 2 nd Law of motion states that the action
Br, is used in the production of polymers.
 1 Allyl bromide, =CH Br, is used in the production of polymers. (a) Part of the C=C double bond in allyl bromide is called a π-bond. Draw a labelled diagram to show the formation of the π-bond. [2] (b)
1 Allyl bromide, =CH Br, is used in the production of polymers. (a) Part of the C=C double bond in allyl bromide is called a π-bond. Draw a labelled diagram to show the formation of the π-bond. [2] (b)
Spherical Coordinates
 Spherical Coordinates MATH 311, Calculus III J. Robert Buchanan Department of Mathematics Fall 2011 Spherical Coordinates Another means of locating points in three-dimensional space is known as the spherical
Spherical Coordinates MATH 311, Calculus III J. Robert Buchanan Department of Mathematics Fall 2011 Spherical Coordinates Another means of locating points in three-dimensional space is known as the spherical
Areas and Lengths in Polar Coordinates
 Kiryl Tsishchanka Areas and Lengths in Polar Coordinates In this section we develop the formula for the area of a region whose boundary is given by a polar equation. We need to use the formula for the
Kiryl Tsishchanka Areas and Lengths in Polar Coordinates In this section we develop the formula for the area of a region whose boundary is given by a polar equation. We need to use the formula for the
Solutions to Exercise Sheet 5
 Solutions to Eercise Sheet 5 jacques@ucsd.edu. Let X and Y be random variables with joint pdf f(, y) = 3y( + y) where and y. Determine each of the following probabilities. Solutions. a. P (X ). b. P (X
Solutions to Eercise Sheet 5 jacques@ucsd.edu. Let X and Y be random variables with joint pdf f(, y) = 3y( + y) where and y. Determine each of the following probabilities. Solutions. a. P (X ). b. P (X
Solution Series 9. i=1 x i and i=1 x i.
 Lecturer: Prof. Dr. Mete SONER Coordinator: Yilin WANG Solution Series 9 Q1. Let α, β >, the p.d.f. of a beta distribution with parameters α and β is { Γ(α+β) Γ(α)Γ(β) f(x α, β) xα 1 (1 x) β 1 for < x
Lecturer: Prof. Dr. Mete SONER Coordinator: Yilin WANG Solution Series 9 Q1. Let α, β >, the p.d.f. of a beta distribution with parameters α and β is { Γ(α+β) Γ(α)Γ(β) f(x α, β) xα 1 (1 x) β 1 for < x
Photo-Induced Self-Assembly of Pt(II)-Linked Rings and Cages via the Photolabilization of a Pt(II) Pyridine Bond
 Photo-Induced Self-Assembly of Pt(II)-Linked Rings and Cages via the Photolabilization of a Pt(II) Pyridine Bond Ken-ichi Yamashita, Kei-ichi Sato, Masaki Kawano and Makoto Fujita* Contents; Figure S1.
Photo-Induced Self-Assembly of Pt(II)-Linked Rings and Cages via the Photolabilization of a Pt(II) Pyridine Bond Ken-ichi Yamashita, Kei-ichi Sato, Masaki Kawano and Makoto Fujita* Contents; Figure S1.
Numerical Analysis FMN011
 Numerical Analysis FMN011 Carmen Arévalo Lund University carmen@maths.lth.se Lecture 12 Periodic data A function g has period P if g(x + P ) = g(x) Model: Trigonometric polynomial of order M T M (x) =
Numerical Analysis FMN011 Carmen Arévalo Lund University carmen@maths.lth.se Lecture 12 Periodic data A function g has period P if g(x + P ) = g(x) Model: Trigonometric polynomial of order M T M (x) =
Table S1. Summary of data collections and structure refinements for crystals 1Rb-1h, 1Rb-2h, and 1Rb-4h.
 Supporting Information [NH 3 CH 3 ] [In SbS 9 SH]: A novel methylamine-directed indium thioantimonate with Rb + ion-exchange property Kai-Yao Wang a,b, Mei-Ling Feng a, Jian-Rong Li a and Xiao-Ying Huang
Supporting Information [NH 3 CH 3 ] [In SbS 9 SH]: A novel methylamine-directed indium thioantimonate with Rb + ion-exchange property Kai-Yao Wang a,b, Mei-Ling Feng a, Jian-Rong Li a and Xiao-Ying Huang
Supporting Information. Palladium Complexes with Bulky Diphosphine. Synthesis of (Bio-) Adipic Acid from Pentenoic. Acid Mixtures.
 Supporting Information Palladium Complexes with Bulky Diphosphine Ligands as Highly Selective Catalysts for the Synthesis of (Bio-) Adipic Acid from Pentenoic Acid Mixtures. Choon Heng Low, James D. Nobbs,*
Supporting Information Palladium Complexes with Bulky Diphosphine Ligands as Highly Selective Catalysts for the Synthesis of (Bio-) Adipic Acid from Pentenoic Acid Mixtures. Choon Heng Low, James D. Nobbs,*
«ΑΓΡΟΤΟΥΡΙΣΜΟΣ ΚΑΙ ΤΟΠΙΚΗ ΑΝΑΠΤΥΞΗ: Ο ΡΟΛΟΣ ΤΩΝ ΝΕΩΝ ΤΕΧΝΟΛΟΓΙΩΝ ΣΤΗΝ ΠΡΟΩΘΗΣΗ ΤΩΝ ΓΥΝΑΙΚΕΙΩΝ ΣΥΝΕΤΑΙΡΙΣΜΩΝ»
 I ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΣΧΟΛΗ ΝΟΜΙΚΩΝ ΟΙΚΟΝΟΜΙΚΩΝ ΚΑΙ ΠΟΛΙΤΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΤΜΗΜΑ ΟΙΚΟΝΟΜΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΗΝ «ΔΙΟΙΚΗΣΗ ΚΑΙ ΟΙΚΟΝΟΜΙΑ» ΚΑΤΕΥΘΥΝΣΗ: ΟΙΚΟΝΟΜΙΚΗ
I ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΣΧΟΛΗ ΝΟΜΙΚΩΝ ΟΙΚΟΝΟΜΙΚΩΝ ΚΑΙ ΠΟΛΙΤΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΤΜΗΜΑ ΟΙΚΟΝΟΜΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΗΝ «ΔΙΟΙΚΗΣΗ ΚΑΙ ΟΙΚΟΝΟΜΙΑ» ΚΑΤΕΥΘΥΝΣΗ: ΟΙΚΟΝΟΜΙΚΗ
DETERMINATION OF DYNAMIC CHARACTERISTICS OF A 2DOF SYSTEM. by Zoran VARGA, Ms.C.E.
 DETERMINATION OF DYNAMIC CHARACTERISTICS OF A 2DOF SYSTEM by Zoran VARGA, Ms.C.E. Euro-Apex B.V. 1990-2012 All Rights Reserved. The 2 DOF System Symbols m 1 =3m [kg] m 2 =8m m=10 [kg] l=2 [m] E=210000
DETERMINATION OF DYNAMIC CHARACTERISTICS OF A 2DOF SYSTEM by Zoran VARGA, Ms.C.E. Euro-Apex B.V. 1990-2012 All Rights Reserved. The 2 DOF System Symbols m 1 =3m [kg] m 2 =8m m=10 [kg] l=2 [m] E=210000
Site-Selective Suzuki-Miyaura Cross-Coupling Reactions of 2,3,4,5-Tetrabromofuran
 1 Site-Selective Suzuki-Miyaura Cross-Coupling Reactions of 2,3,4,5-Tetrabromofuran Munawar Hussain, a Rasheed Ahmad Khera, a Nguyen Thai Hung, a Peter Langer* a,b a Institut für Chemie, Universität Rostock,
1 Site-Selective Suzuki-Miyaura Cross-Coupling Reactions of 2,3,4,5-Tetrabromofuran Munawar Hussain, a Rasheed Ahmad Khera, a Nguyen Thai Hung, a Peter Langer* a,b a Institut für Chemie, Universität Rostock,
Iodine-catalyzed synthesis of sulfur-bridged enaminones and chromones via double C(sp 2 )-H thiolation
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2017 Iodine-catalyzed synthesis of sulfur-bridged enaminones and chromones via
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2017 Iodine-catalyzed synthesis of sulfur-bridged enaminones and chromones via
ΔΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ ΕΠΑΝΑΣΧΕΔΙΑΣΜΟΣ ΓΡΑΜΜΗΣ ΣΥΝΑΡΜΟΛΟΓΗΣΗΣ ΜΕ ΧΡΗΣΗ ΕΡΓΑΛΕΙΩΝ ΛΙΤΗΣ ΠΑΡΑΓΩΓΗΣ REDESIGNING AN ASSEMBLY LINE WITH LEAN PRODUCTION TOOLS
 ΔΙΑΤΜΗΜΑΤΙΚΟ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΗ ΔΙΟΙΚΗΣΗ ΤΩΝ ΕΠΙΧΕΙΡΗΣΕΩΝ ΔΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ ΕΠΑΝΑΣΧΕΔΙΑΣΜΟΣ ΓΡΑΜΜΗΣ ΣΥΝΑΡΜΟΛΟΓΗΣΗΣ ΜΕ ΧΡΗΣΗ ΕΡΓΑΛΕΙΩΝ ΛΙΤΗΣ ΠΑΡΑΓΩΓΗΣ REDESIGNING AN ASSEMBLY LINE WITH
ΔΙΑΤΜΗΜΑΤΙΚΟ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΗ ΔΙΟΙΚΗΣΗ ΤΩΝ ΕΠΙΧΕΙΡΗΣΕΩΝ ΔΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ ΕΠΑΝΑΣΧΕΔΙΑΣΜΟΣ ΓΡΑΜΜΗΣ ΣΥΝΑΡΜΟΛΟΓΗΣΗΣ ΜΕ ΧΡΗΣΗ ΕΡΓΑΛΕΙΩΝ ΛΙΤΗΣ ΠΑΡΑΓΩΓΗΣ REDESIGNING AN ASSEMBLY LINE WITH
Homework 3 Solutions
 Homework 3 Solutions Igor Yanovsky (Math 151A TA) Problem 1: Compute the absolute error and relative error in approximations of p by p. (Use calculator!) a) p π, p 22/7; b) p π, p 3.141. Solution: For
Homework 3 Solutions Igor Yanovsky (Math 151A TA) Problem 1: Compute the absolute error and relative error in approximations of p by p. (Use calculator!) a) p π, p 22/7; b) p π, p 3.141. Solution: For
Supporting Information. Consecutive hydrazino-ugi-azide reactions: synthesis of acylhydrazines bearing 1,5- disubstituted tetrazoles
 Supporting Information for Consecutive hydrazino-ugi-azide reactions: synthesis of acylhydrazines bearing 1,5- disubstituted tetrazoles Angélica de Fátima S. Barreto*, Veronica Alves dos Santos, and Carlos
Supporting Information for Consecutive hydrazino-ugi-azide reactions: synthesis of acylhydrazines bearing 1,5- disubstituted tetrazoles Angélica de Fátima S. Barreto*, Veronica Alves dos Santos, and Carlos
Math221: HW# 1 solutions
 Math: HW# solutions Andy Royston October, 5 7.5.7, 3 rd Ed. We have a n = b n = a = fxdx = xdx =, x cos nxdx = x sin nx n sin nxdx n = cos nx n = n n, x sin nxdx = x cos nx n + cos nxdx n cos n = + sin
Math: HW# solutions Andy Royston October, 5 7.5.7, 3 rd Ed. We have a n = b n = a = fxdx = xdx =, x cos nxdx = x sin nx n sin nxdx n = cos nx n = n n, x sin nxdx = x cos nx n + cos nxdx n cos n = + sin
SCHOOL OF MATHEMATICAL SCIENCES G11LMA Linear Mathematics Examination Solutions
 SCHOOL OF MATHEMATICAL SCIENCES GLMA Linear Mathematics 00- Examination Solutions. (a) i. ( + 5i)( i) = (6 + 5) + (5 )i = + i. Real part is, imaginary part is. (b) ii. + 5i i ( + 5i)( + i) = ( i)( + i)
SCHOOL OF MATHEMATICAL SCIENCES GLMA Linear Mathematics 00- Examination Solutions. (a) i. ( + 5i)( i) = (6 + 5) + (5 )i = + i. Real part is, imaginary part is. (b) ii. + 5i i ( + 5i)( + i) = ( i)( + i)
Aluminium triflate as a Lewis acid catalyst for the ring opening of epoxides in alcohols
 Aluminium triflate as a Lewis acid catalyst for the ring opening of epoxides in alcohols D. Bradley G. Williams* and Michelle Lawton a Department of Chemistry, University of Johannesburg, P.O. Box 524,
Aluminium triflate as a Lewis acid catalyst for the ring opening of epoxides in alcohols D. Bradley G. Williams* and Michelle Lawton a Department of Chemistry, University of Johannesburg, P.O. Box 524,
If we restrict the domain of y = sin x to [ π, π ], the restrict function. y = sin x, π 2 x π 2
![If we restrict the domain of y = sin x to [ π, π ], the restrict function. y = sin x, π 2 x π 2 If we restrict the domain of y = sin x to [ π, π ], the restrict function. y = sin x, π 2 x π 2](/thumbs/53/31933086.jpg) Chapter 3. Analytic Trigonometry 3.1 The inverse sine, cosine, and tangent functions 1. Review: Inverse function (1) f 1 (f(x)) = x for every x in the domain of f and f(f 1 (x)) = x for every x in the
Chapter 3. Analytic Trigonometry 3.1 The inverse sine, cosine, and tangent functions 1. Review: Inverse function (1) f 1 (f(x)) = x for every x in the domain of f and f(f 1 (x)) = x for every x in the
3.4 SUM AND DIFFERENCE FORMULAS. NOTE: cos(α+β) cos α + cos β cos(α-β) cos α -cos β
 3.4 SUM AND DIFFERENCE FORMULAS Page Theorem cos(αβ cos α cos β -sin α cos(α-β cos α cos β sin α NOTE: cos(αβ cos α cos β cos(α-β cos α -cos β Proof of cos(α-β cos α cos β sin α Let s use a unit circle
3.4 SUM AND DIFFERENCE FORMULAS Page Theorem cos(αβ cos α cos β -sin α cos(α-β cos α cos β sin α NOTE: cos(αβ cos α cos β cos(α-β cos α -cos β Proof of cos(α-β cos α cos β sin α Let s use a unit circle
1 String with massive end-points
 1 String with massive end-points Πρόβλημα 5.11:Θεωρείστε μια χορδή μήκους, τάσης T, με δύο σημειακά σωματίδια στα άκρα της, το ένα μάζας m, και το άλλο μάζας m. α) Μελετώντας την κίνηση των άκρων βρείτε
1 String with massive end-points Πρόβλημα 5.11:Θεωρείστε μια χορδή μήκους, τάσης T, με δύο σημειακά σωματίδια στα άκρα της, το ένα μάζας m, και το άλλο μάζας m. α) Μελετώντας την κίνηση των άκρων βρείτε
