THERMODYNAMICS. For an ideal gas, Pv = RT or PV = mrt, and P 1
|
|
|
- Νέφθυς Σαμαράς
- 8 χρόνια πριν
- Προβολές:
Transcript
1 HERMODYNAMICS PROPERIES OF SINGLE-COMPONEN SYSEMS Nomenclature. Intense propertes are ndependent of mass.. Extense propertes are proportonal to mass. State Functons (propertes Absolute Pressure, P (lbf/n or Pa Absolute emperature, ( R or K Volume, V (ft 3 or m 3 V m (ft 3 /lbm or m 3 /kg Internal Energy, U (Btu or kj u U m (usually n Btu/lbm or kj/kg Entalpy, H (Btu or KJ Entalpy, u + P H/m (usually n Btu/lbm or kj/kg Entropy, S (Btu/ R or kj/k s S/m [Btu/(lbm- R or kj/ Gbbs Free Energy, g s (usually n Btu/lbm or kj/kg Helmolz Free Energy, a u s (usually n Btu/lbm or kj/kg Heat Capacty at Constant Pressure, c p b l Heat Capacty at Constant Volume, c b u l Qualty x (apples to lqud-apor systems at saturaton s x m g /(m g + m f m g m f mass of apor, and mass of lqud. can be wrtten: x g + ( x f or f + x fg f g fg g f Smlar expressons exst for u,, and s: u xu g + ( x u f or u u f + xu fg x g + ( x f or f + x fg s xs g + ( x s f or s s f + xs fg P For an deal gas, P R or PV mr, and P / P / P pressure, m mass of gas, R gas constant, and absolute temperature. V olume R s but can be found from R R ^ mol. wt R te unersal gas constant,545 ft-lbf/(lbmol- R 8,34 J/(kmol K. For deal gases, c p c R Also, for deal gases: b P u l b l For cold ar standard, eat capactes are assumed to be constant at ter room temperature alues. In tat case, te followng are true: Δu c Δ; Δ c p Δ Δs c p ln ( / R ln (P /P ; and Δs c ln ( / + R ln ( /. For eat capactes tat are temperature dependent, te alue to be used n te aboe equatons for Δ s known as te mean eat capacty `c p j and s gen by c p # cd p - Also, for constant entropy processes: k k - P P k ; P d n d n P k -, d n were k cp c For real gases, seeral equatons of state are aalable; one suc equaton s te an der Waals equaton wt constants based on te crtcal pont: P a c + m ^ - b R a 7 Rc Rc were c m f p, b 64 Pc 8Pc were P c and c are te pressure and temperature at te crtcal pont, respectely, and HERMODYNAMICS 73
2 FIRS LAW OF HERMODYNAMICS e Frst Law of ermodynamcs s a statement of conseraton of energy n a termodynamc system. e net energy crossng te system boundary s equal to te cange n energy nsde te system. Heat Q s energy transferred due to temperature dfference and s consdered poste f t s nward or added to te system. Closed ermodynamc System No mass crosses system boundary Q W ΔU + ΔKE + ΔPE were ΔKE cange n knetc energy, and ΔPE cange n potental energy. Energy can cross te boundary only n te form of eat or work. Work can be boundary work, w b, or oter work forms (electrcal work, etc. Work W bw W l m s consdered poste f t s outward or work done by te system. Reersble boundary work s gen by w b P d. Specal Cases of Closed Systems Constant Pressure ( : w b PΔ (deal gas / constant Constant Volume: w b (deal gas /P constant Isentropc (deal gas: P k constant w (P P /( k R( /( k Constant emperature ( : (deal gas P constant w b Rln ( / Rln (P /P Polytropc (deal gas: P n constant w (P P /( n Open ermodynamc System Mass crosses te system boundary P done by mass enterng te system. w re dp + Δke + Δpe Frst Law apples weter or not processes are reersble. FIRS LAW (energy balance Rmo 8 + V / + gzb- Rmo e8e + Ve / + gzeb + Qo - Wo d_ mu / dt, were n net s s Wo net rate of net or saft work transfer, ms us Qo rate of eat transfer (neglectng knetc and potental energy of te system. Specal Cases of Open Systems Constant Volume: w re (P P Constant Pressure: w re Constant emperature: (deal gas P constant w re R ln ( / R ln (P /P Isentropc (deal gas: P k constant w re k (P P /( k kr ( /( k w re ^k - / k k k P R > - d n H - P Polytropc: P n constant w re n (P P /( n Steady-State Systems e system does not cange state wt tme. s assumpton s ald for steady operaton of turbnes, pumps, compressors, trottlng ales, nozzles, and eat excangers, ncludng bolers and condensers. Rm o ` + V / + gzj- Rmo e`e + Ve / + gzej + Qo n - Wo out and Rmo Rmo were e mo and e refer to nlet and ext states of system, g acceleraton of graty, Z eleaton, V elocty, and Wo rate of work. Specal Cases of Steady-Flow Energy Equaton Nozzles, Dffusers: eleaton cange, no eat transfer, and no work. Sngle mass stream. + V / + V / e e Ve - V _ - es entalpy at sentropc ext state. urbnes, Pumps, Compressors: Often consdered adabatc (no eat transfer. Velocty terms usually can be gnored. e + w es 74 HERMODYNAMICS
3 - - e es es e - - rottlng Vales and rottlng Processes: No work, no eat transfer, and sngle-mass stream. Velocty terms are often e Bolers, Condensers, Eaporators, One Sde n a Heat Excanger: mass stream, te followng apples: + q e Heat Excangers: mo and mo : mo _ - mo _ - e e See MECHANICAL ENGINEERING secton. Mxers, Separators, Rm o Rm o e e and Rmo Rmo e BASIC CYCLES Heat engnes take n eat Q H at a g temperature H, produce a net amount of work W, and reject eat Q L at a low temperature L η of a eat engne s gen by: η W/Q H (Q H Q L /Q H Carnot Cycle. Its η c ( H L / H H and L absolute temperatures (Keln or Rankne. e followng eat-engne cycles are plotted on P- and -s dagrams (see later n ts capter: Carnot, Otto, Rankne Refrgeraton cycles are te reerse of eat-engne cycles. Heat s moed from low to g temperature requrng work, W. Cycles can be used eter for refrgeraton or as eat pumps. COP Q H /W for eat pumps, and as COP Q L /W for refrgerators and ar condtoners. Upper lmt of COP s based on reersed Carnot Cycle: COP c H /( H L for eat pumps and COP c L /( H L for refrgeraton. ton refrgeraton, Btu/r 3,56 W IDEAL GAS MIXURES,,, n consttuents. Eac consttuent s an deal gas. Mole Fracton: x N /N; N Σ N ; Σ x were N number of moles of component. Mass Fracton: y m /m; m Σ m ; Σ y Molecular Wegt: M m/n Σ x M Gas Constant: R R/ M o conert mole fractons x to mass fractons y : xm y R_ xm o conert mass fractons to mole fractons: y M x R_ y M mr Partal Pressures: P RP; P V mr Partal Volumes: V! V; V P P, V, te pressure, olume, and temperature of te mxture. x P /P V /V Oter Propertes: u Σ (y u ; Σ (y ; s Σ (y s u and are ealuated at, and s s ealuated at and P. PSYCHROMERICS We deal ere wt a mxture of dry ar (subscrpt a and water apor (subscrpt : P P a + P (absolute umdty, umdty rato ω: ω m /m a m mass of water apor and m a mass of dry ar. ω.6p /P a.6p /(P P Relate Humdty (r φ: φ P /P g P g saturaton pressure at. Entalpy : a + ω Dew-Pont emperature dp : dp sat at P g P HERMODYNAMICS 75
4 Wet-bulb temperature wb s te temperature ndcated by a termometer coered by a wck saturated wt lqud water and n contact wt mong ar. Humd Volume: Volume of most ar/mass of dry ar. Psycrometrc Cart temperature plotted for a alue of atmosperc pressure. (See cart at end of secton. PHASE RELAIONS Clapeyron Equaton for Pase ranstons: b d dp l sat fg s fg fg fg, were fg entalpy cange for pase transtons, fg olume cange, s fg entropy cange, absolute temperature, and (dp/d sat slope of pase transton (e.g.,apor-lqud saturaton lne. Clausus-Clapeyron Equaton s equaton results f t s assumed tat ( te olume cange ( fg can be replaced wt te apor olume ( g, ( te latter can be replaced wt P R from te deal gas law, and (3 fg s ndependent of te temperature (. P fg - lne d n P : R Gbbs Pase Rule (non-reactng systems P + F C + P number of pases makng up a system F degrees of freedom, and C number of components n a system COMBUSION PROCESSES Frst, te combuston equaton sould be wrtten and balanced. For example, for te stocometrc combuston of metane n oxygen: CH 4 + O CO + H O Combuston n Ar For eac mole of oxygen, tere wll be 3.76 moles of ntrogen. For stocometrc combuston of metane n ar: CH 4 + O + (3.76 N CO + H O N Combuston n Excess Ar e excess oxygen appears as oxygen on te rgt sde of te combuston equaton. Incomplete Combuston Some carbon s burned to create carbon monoxde (CO. Ar-Fuel Rato (A/F: A/F mass of ar mass of fuel Stocometrc (teoretcal ar-fuel rato s te ar-fuel rato calculated from te stocometrc combuston equaton. _ A F actual Percent eoretcal Ar # _ A F stocometrc _ A F - _ A F Percent Excess Ar _ A F actual stocometrc # stocometrc SECOND LAW OF HERMODYNAMICS ermal Energy Reserors ΔS reseror Q/ reseror Q s measured wt respect to te reseror. Keln-Planck Statement of Second Law No eat engne can operate n a cycle wle transferrng eat wt a sngle eat reseror. COROLLARY to Keln-Planck: No eat engne can ae a same reserors. Clausus Statement of Second Law No refrgeraton or eat pump cycle can operate wtout a net work nput. COROLLARY: No refrgerator or eat pump can ae a ger COP tan a Carnot Cycle refrgerator or eat pump. VAPOR-LIQUID MIXURES Henry s Law at Constant emperature At equlbrum, te partal pressure of a gas s proportonal to ts concentraton n a lqud. Henry s Law s ald for low concentratons;.e., x. P Py x Henry s Law constant, P partal pressure of a gas n contact wt a lqud, x mol fracton of te gas n te lqud, y mol fracton of te gas n te apor, and P total pressure. Raoult s Law for Vapor-Lqud Equlbrum Vald for concentratons near ;.e., x. P x P * P partal pressure of component, x mol fracton of component n te lqud, and P * apor pressure of pure component at te temperature of te mxture. 76 HERMODYNAMICS
5 ENROPY ds _ dqre s - s # _ dq Inequalty of Clausus # _ dq # re # re _ dq # s - s Isotermal, Reersble Process Δs s s Q/ Isentropc Process Δs ; ds A reersble adabatc process s sentropc. Adabatc Process δq ; Δs Increase of Entropy Prncple Dstotal Dssystem + Dssurroundngs $ Dso Rmo s - Rmo s - R_ Qo / total out out n n external external $ EXERGY Exergy s te porton of total energy aalable to do work. Closed-System Exergy (Aalablty (no cemcal reactons φ (u u o o (s s o + p o ( o were te subscrpt o desgnates enronmental condtons w reersble φ φ Open-System Exergy (Aalablty ψ ( o o (s s o + V / + gz w reersble ψ ψ Gbbs Free Energy, ΔG Energy released or absorbed n a reacton occurrng reersbly at constant pressure and temperature. Helmoltz Free Energy, ΔA Energy released or absorbed n a reacton occurrng reersbly at constant olume and temperature. emperature-entropy (-s Dagram Q re ds AREA HEA s Entropy Cange for Solds and Lquds ds c (d/ s s c (d/ c mean ln ( /, were c equals te eat capacty of te sold or lqud. Irreersblty I w re w actual HERMODYNAMICS 77
6 COMMON HERMODYNAMIC CYCLES Carnot Cycle Reersed Carnot Otto Cycle (Gasolne Engne η r k q r / Rankne Cycle Refrgeraton (Reersed Rankne Cycle w q q w c q q p p 3 p p 3 η ( ( COP ref 4 COP HP 3 78 HERMODYNAMICS
7 emp. o C Press. kpa p sat MPa Specfc Volume m 3 /kg lqud f apor g SEAM ABLES Saturated Water - emperature able Internal Energy Entalpy kj/kg kj/kg Eap. lqud apor lqud Eap. u f u fg u g f fg apor g lqud s f Entropy kj/(kg K Eap. s fg apor s g HERMODYNAMICS 79
8 Supereated Water ables u s u emp. m 3 /kg kj/kg kj/kg kj/(kg K m 3 /kg kj/kg kj/kg o C p. MPa (45.8 o C p.5 MPa (8.33 o C s kj/(kg K p. MPa (99.63 o C p. MPa (.3 o C p.4 MPa (43.63 o C p.6 MPa (58.85 o C p.8 MPa (7.43 o C p. MPa (79.9 o C HERMODYNAMICS
9 P- DIAGRAM FOR REFRIGERAN HFC-34a HERMODYNAMICS 8
10 ASHRAE PSYCHROMERIC CHAR NO WE BULB EMPERAURE - C ENHALPY - KJ PER KILOGRAM OF DRY AIR ENHALPY - KJ PER KILOGRAM OF DRY AIR SAURAION EMPERAURE - C DRY BULB EMPERAURE - C HUMIDIY RAIO - KILOGRAMS MOISURE PER KILOGRAM DRY AIR % 5 8% 5 7% 6% 5% VOLUME - CUBIC MEER PER KG DRY AIR ASHRAE PSYCHROMERIC CHAR NO. NORMAL EMPERAURE BAROMERIC PRESSURE:.35 kpa Copyrgt 99 R R AMERICAN SOCIEY OF HEAING, REFRIGERAING AND AIR-CONDIIONING ENGINEERS, INC. SEA LEVEL SENSIBLE HEA Qs OAL HEA Qt ENHALPY HUMIDIY RAIO W.84 4% 5 %.8 %.8 % RELAIVE HUMIDIY.78 8 HERMODYNAMICS
11 HERMAL AND PHYSICAL PROPERY ABLES GASES Substance Mol wt c p c kj/(kg K Btu/(lbm- R kj/(kg K Btu/(lbm- R k R kj/(kg K Gases Ar Argon Butane Carbon doxde Carbon monoxde Etane Helum Hydrogen Metane Neon Ntrogen Octane apor Oxygen Propane Steam SELECED LIQUIDS AND SOLIDS Substance c p Densty kj/(kg K Btu/(lbm- R kg/m 3 lbm/ft 3 Lquds Ammona Mercury Water , Solds Alumnum Copper Ice ( C; 3 F Iron Lead ,7 8,9 97 7,84, HERMODYNAMICS 83
DuPont Suva 95 Refrigerant
 Technical Information T-95 ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Technical Information T-95 ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Classical Theory (3): Thermostatics of Continuous Systems with External Forces
 Insttute of Flu- & Thermoynamcs Unersty of Segen Classcal Theory (3): Thermostatcs of Contnuous Systems wth External Forces 3/ Σ: Equlbrum State? Isolaton, Inhomogenety External Forces F ϕ Components:...
Insttute of Flu- & Thermoynamcs Unersty of Segen Classcal Theory (3): Thermostatcs of Contnuous Systems wth External Forces 3/ Σ: Equlbrum State? Isolaton, Inhomogenety External Forces F ϕ Components:...
DuPont Suva. DuPont. Thermodynamic Properties of. Refrigerant (R-410A) Technical Information. refrigerants T-410A ENG
 Technical Information T-410A ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 410A Refrigerant (R-410A) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Technical Information T-410A ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 410A Refrigerant (R-410A) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
2. Chemical Thermodynamics and Energetics - I
 . Chemical Thermodynamics and Energetics - I 1. Given : Initial Volume ( = 5L dm 3 Final Volume (V = 10L dm 3 ext = 304 cm of Hg Work done W = ext V ext = 304 cm of Hg = 304 atm [... 76cm of Hg = 1 atm]
. Chemical Thermodynamics and Energetics - I 1. Given : Initial Volume ( = 5L dm 3 Final Volume (V = 10L dm 3 ext = 304 cm of Hg Work done W = ext V ext = 304 cm of Hg = 304 atm [... 76cm of Hg = 1 atm]
APPENDIX A. Summary of the English Engineering (EE) System of Units
 Appendixes A. Summary of the English Engineering (EE) System of Units B. Summary of the International System (SI) of Units C. Friction-Factor Chart D. Oblique-Shock Charts (γ = 1.4) (Two-Dimensional) E.
Appendixes A. Summary of the English Engineering (EE) System of Units B. Summary of the International System (SI) of Units C. Friction-Factor Chart D. Oblique-Shock Charts (γ = 1.4) (Two-Dimensional) E.
Technical Information T-9100 SI. Suva. refrigerants. Thermodynamic Properties of. Suva Refrigerant [R-410A (50/50)]
![Technical Information T-9100 SI. Suva. refrigerants. Thermodynamic Properties of. Suva Refrigerant [R-410A (50/50)] Technical Information T-9100 SI. Suva. refrigerants. Thermodynamic Properties of. Suva Refrigerant [R-410A (50/50)]](/thumbs/89/98928029.jpg) d Suva refrigerants Technical Information T-9100SI Thermodynamic Properties of Suva 9100 Refrigerant [R-410A (50/50)] Thermodynamic Properties of Suva 9100 Refrigerant SI Units New tables of the thermodynamic
d Suva refrigerants Technical Information T-9100SI Thermodynamic Properties of Suva 9100 Refrigerant [R-410A (50/50)] Thermodynamic Properties of Suva 9100 Refrigerant SI Units New tables of the thermodynamic
DuPont Suva 95 Refrigerant
 Technical Information T-95 SI DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Technical Information T-95 SI DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Mean bond enthalpy Standard enthalpy of formation Bond N H N N N N H O O O
 Q1. (a) Explain the meaning of the terms mean bond enthalpy and standard enthalpy of formation. Mean bond enthalpy... Standard enthalpy of formation... (5) (b) Some mean bond enthalpies are given below.
Q1. (a) Explain the meaning of the terms mean bond enthalpy and standard enthalpy of formation. Mean bond enthalpy... Standard enthalpy of formation... (5) (b) Some mean bond enthalpies are given below.
Chapter 22 - Heat Engines, Entropy, and the Second Law of Thermodynamics
 apter - Heat Engines, Entropy, and te Seond Law o ermodynamis.1 (a).0 J e 0.069 4 or 6.94% 60 J (b) 60 J.0 J J. e eat to melt 1.0 g o Hg is 4 ml 1 10 kg 1.18 10 J kg 177 J e energy absorbed to reeze 1.00
apter - Heat Engines, Entropy, and te Seond Law o ermodynamis.1 (a).0 J e 0.069 4 or 6.94% 60 J (b) 60 J.0 J J. e eat to melt 1.0 g o Hg is 4 ml 1 10 kg 1.18 10 J kg 177 J e energy absorbed to reeze 1.00
Major Concepts. Multiphase Equilibrium Stability Applications to Phase Equilibrium. Two-Phase Coexistence
 Major Concepts Multiphase Equilibrium Stability Applications to Phase Equilibrium Phase Rule Clausius-Clapeyron Equation Special case of Gibbs-Duhem wo-phase Coexistence Criticality Metastability Spinodal
Major Concepts Multiphase Equilibrium Stability Applications to Phase Equilibrium Phase Rule Clausius-Clapeyron Equation Special case of Gibbs-Duhem wo-phase Coexistence Criticality Metastability Spinodal
One and two particle density matrices for single determinant HF wavefunctions. (1) = φ 2. )β(1) ( ) ) + β(1)β * β. (1)ρ RHF
 One and two partcle densty matrces for sngle determnant HF wavefunctons One partcle densty matrx Gven the Hartree-Fock wavefuncton ψ (,,3,!, = Âϕ (ϕ (ϕ (3!ϕ ( 3 The electronc energy s ψ H ψ = ϕ ( f ( ϕ
One and two partcle densty matrces for sngle determnant HF wavefunctons One partcle densty matrx Gven the Hartree-Fock wavefuncton ψ (,,3,!, = Âϕ (ϕ (ϕ (3!ϕ ( 3 The electronc energy s ψ H ψ = ϕ ( f ( ϕ
STEAM TABLES. Mollier Diagram
 STEAM TABLES and Mollier Diagram (S.I. Units) dharm \M-therm\C-steam.pm5 CONTENTS Table No. Page No. 1. Saturated Water and Steam (Temperature) Tables I (ii) 2. Saturated Water and Steam (Pressure) Tables
STEAM TABLES and Mollier Diagram (S.I. Units) dharm \M-therm\C-steam.pm5 CONTENTS Table No. Page No. 1. Saturated Water and Steam (Temperature) Tables I (ii) 2. Saturated Water and Steam (Pressure) Tables
938 Thermodynamics TABLE A 18. Ideal-gas properties of nitrogen, N 2 _ _ T
 cen84959c18-ap01.qxd 8/11/06 1:21 PM Page 938 938 ermodynamics ABLE A 18 Ideal-gas properties of nitrogen, N 2 0 0 0 0 600 17,563 12,574 212.066 220 6,391 4,562 182.639 610 17,864 12,792 212.564 230 6,683
cen84959c18-ap01.qxd 8/11/06 1:21 PM Page 938 938 ermodynamics ABLE A 18 Ideal-gas properties of nitrogen, N 2 0 0 0 0 600 17,563 12,574 212.066 220 6,391 4,562 182.639 610 17,864 12,792 212.564 230 6,683
Figure 1 T / K Explain, in terms of molecules, why the first part of the graph in Figure 1 is a line that slopes up from the origin.
 Q1.(a) Figure 1 shows how the entropy of a molecular substance X varies with temperature. Figure 1 T / K (i) Explain, in terms of molecules, why the entropy is zero when the temperature is zero Kelvin.
Q1.(a) Figure 1 shows how the entropy of a molecular substance X varies with temperature. Figure 1 T / K (i) Explain, in terms of molecules, why the entropy is zero when the temperature is zero Kelvin.
α & β spatial orbitals in
 The atrx Hartree-Fock equatons The most common method of solvng the Hartree-Fock equatons f the spatal btals s to expand them n terms of known functons, { χ µ } µ= consder the spn-unrestrcted case. We
The atrx Hartree-Fock equatons The most common method of solvng the Hartree-Fock equatons f the spatal btals s to expand them n terms of known functons, { χ µ } µ= consder the spn-unrestrcted case. We
Contents of Appendix
 Contents of Appendix A SI UNITS: SINGLE-STATE PROPERTIES 755 Table A.1 Conversion Factors, 755 Table A.2 Critical Constants, 758 Table A.3 Properties of Selected Solids at 25 C, 759 Table A.4 Properties
Contents of Appendix A SI UNITS: SINGLE-STATE PROPERTIES 755 Table A.1 Conversion Factors, 755 Table A.2 Critical Constants, 758 Table A.3 Properties of Selected Solids at 25 C, 759 Table A.4 Properties
Calculation of ODH classification for Nevis LHe e-bubble Chamber Cryostat
 Calculaton of ODH classfcaton for Nevs LHe e-bubble Chamber Cryostat. Physcal parameters Thermal propertes of Helum and Ntrogen Yongln Ju Nevs Laboratores, Columba Unversty, NY 533 T 3 [K] Pa 76 [mmhg]
Calculaton of ODH classfcaton for Nevs LHe e-bubble Chamber Cryostat. Physcal parameters Thermal propertes of Helum and Ntrogen Yongln Ju Nevs Laboratores, Columba Unversty, NY 533 T 3 [K] Pa 76 [mmhg]
HOMEWORK 4 = G. In order to plot the stress versus the stretch we define a normalized stretch:
 HOMEWORK 4 Problem a For the fast loading case, we want to derive the relationship between P zz and λ z. We know that the nominal stress is expressed as: P zz = ψ λ z where λ z = λ λ z. Therefore, applying
HOMEWORK 4 Problem a For the fast loading case, we want to derive the relationship between P zz and λ z. We know that the nominal stress is expressed as: P zz = ψ λ z where λ z = λ λ z. Therefore, applying
Pof moist air and uses these properties to analyze conditions and
 CHAPTER 6 PSYCHROMETRICS Composition of Dry and Moist Air... 6.1 United States Standard Atmosphere... 6.1 Thermodynamic Properties of Moist Air... 6.2 Thermodynamic Properties of Water at Saturation...
CHAPTER 6 PSYCHROMETRICS Composition of Dry and Moist Air... 6.1 United States Standard Atmosphere... 6.1 Thermodynamic Properties of Moist Air... 6.2 Thermodynamic Properties of Water at Saturation...
Phasor Diagram of an RC Circuit V R
 ESE Lecture 3 Phasor Dagram of an rcut VtV m snt V t V o t urrent s a reference n seres crcut KVL: V m V + V V ϕ I m V V m ESE Lecture 3 Phasor Dagram of an L rcut VtV m snt V t V t L V o t KVL: V m V
ESE Lecture 3 Phasor Dagram of an rcut VtV m snt V t V o t urrent s a reference n seres crcut KVL: V m V + V V ϕ I m V V m ESE Lecture 3 Phasor Dagram of an L rcut VtV m snt V t V t L V o t KVL: V m V
By R.L. Snyder (Revised March 24, 2005)
 Humidity Conversion By R.L. Snyder (Revised March 24, 2005) This Web page provides the equations used to make humidity conversions and tables o saturation vapor pressure. For a pd ile o this document,
Humidity Conversion By R.L. Snyder (Revised March 24, 2005) This Web page provides the equations used to make humidity conversions and tables o saturation vapor pressure. For a pd ile o this document,
Multi-dimensional Central Limit Theorem
 Mult-dmensonal Central Lmt heorem Outlne () () () t as () + () + + () () () Consder a sequence of ndependent random proceses t, t, dentcal to some ( t). Assume t 0. Defne the sum process t t t t () t tme
Mult-dmensonal Central Lmt heorem Outlne () () () t as () + () + + () () () Consder a sequence of ndependent random proceses t, t, dentcal to some ( t). Assume t 0. Defne the sum process t t t t () t tme
the total number of electrons passing through the lamp.
 1. A 12 V 36 W lamp is lit to normal brightness using a 12 V car battery of negligible internal resistance. The lamp is switched on for one hour (3600 s). For the time of 1 hour, calculate (i) the energy
1. A 12 V 36 W lamp is lit to normal brightness using a 12 V car battery of negligible internal resistance. The lamp is switched on for one hour (3600 s). For the time of 1 hour, calculate (i) the energy
Multi-dimensional Central Limit Theorem
 Mult-dmensonal Central Lmt heorem Outlne () () () t as () + () + + () () () Consder a sequence of ndependent random proceses t, t, dentcal to some ( t). Assume t 0. Defne the sum process t t t t () t ();
Mult-dmensonal Central Lmt heorem Outlne () () () t as () + () + + () () () Consder a sequence of ndependent random proceses t, t, dentcal to some ( t). Assume t 0. Defne the sum process t t t t () t ();
CH 3 CH 2 COOCH 2 CH 2 CH 3 + H 2 O
 Q1.An experiment was carried out to determine the equilibrium constant, K c, for the following reaction. CH 3 CH 2 COOH + CH 3 CH 2 CH 2 OH CH 3 CH 2 COOCH 2 CH 2 CH 3 + H 2 O A student added measured
Q1.An experiment was carried out to determine the equilibrium constant, K c, for the following reaction. CH 3 CH 2 COOH + CH 3 CH 2 CH 2 OH CH 3 CH 2 COOCH 2 CH 2 CH 3 + H 2 O A student added measured
PROPERTIES OF ORDINARY WATER SUBSTANCE AT SATURATION FROF TME CRITICAL POINT DOWN TO 66 C EQUATIONS & TABLES
 PROPERTIES OF ORDINARY WATER SUBSTANCE AT SATURATION FROF TME CRITICAL POINT DOWN TO 66 C EQUATIONS & TABLES M. CONDE ENGINEERING, 2002 M. CONDE ENGINEERING, Zurich 2002 Properties of ordinary water substance
PROPERTIES OF ORDINARY WATER SUBSTANCE AT SATURATION FROF TME CRITICAL POINT DOWN TO 66 C EQUATIONS & TABLES M. CONDE ENGINEERING, 2002 M. CONDE ENGINEERING, Zurich 2002 Properties of ordinary water substance
ΕΛΛΗΝΙΚΗ ΔΗΜΟΚΡΑΤΙΑ Ανώτατο Εκπαιδευτικό Ίδρυμα Πειραιά Τεχνολογικού Τομέα. Ξενόγλωσση Τεχνική Ορολογία
 ΕΛΛΗΝΙΚΗ ΔΗΜΟΚΡΑΤΙΑ Ανώτατο Εκπαιδευτικό Ίδρυμα Πειραιά Τεχνολογικού Τομέα Ξενόγλωσση Τεχνική Ορολογία Ενότητα: Principles of an Internal Combustion Engine Παναγιώτης Τσατσαρός Τμήμα Μηχανολόγων Μηχανικών
ΕΛΛΗΝΙΚΗ ΔΗΜΟΚΡΑΤΙΑ Ανώτατο Εκπαιδευτικό Ίδρυμα Πειραιά Τεχνολογικού Τομέα Ξενόγλωσση Τεχνική Ορολογία Ενότητα: Principles of an Internal Combustion Engine Παναγιώτης Τσατσαρός Τμήμα Μηχανολόγων Μηχανικών
Thi=Τ1. Thο=Τ2. Tci=Τ3. Tco=Τ4. Thm=Τ5. Tcm=Τ6
 1 Τ.Ε.Ι. ΑΘΗΝΑΣ / Σ.ΤΕ.Φ. ΤΜΗΜΑ ΕΝΕΡΓΕΙΑΚΗΣ ΤΕΧΝΟΛΟΓΙΑΣ ΕΡΓΑΣΤΗΡΙΟ ΜΕΤΑΔΟΣΗΣ ΘΕΡΜΟΤΗΤΟΣ Οδός Αγ.Σπυρίδωνος,12210 Αιγάλεω,Αθήνα Τηλ.: 2105385355, email: ptsiling@teiath.gr H ΕΠΙΔΡΑΣΗ ΠΑΡΟΧΗΣ ΟΓΚΟΥ ΣΤΗΝ
1 Τ.Ε.Ι. ΑΘΗΝΑΣ / Σ.ΤΕ.Φ. ΤΜΗΜΑ ΕΝΕΡΓΕΙΑΚΗΣ ΤΕΧΝΟΛΟΓΙΑΣ ΕΡΓΑΣΤΗΡΙΟ ΜΕΤΑΔΟΣΗΣ ΘΕΡΜΟΤΗΤΟΣ Οδός Αγ.Σπυρίδωνος,12210 Αιγάλεω,Αθήνα Τηλ.: 2105385355, email: ptsiling@teiath.gr H ΕΠΙΔΡΑΣΗ ΠΑΡΟΧΗΣ ΟΓΚΟΥ ΣΤΗΝ
Neutralino contributions to Dark Matter, LHC and future Linear Collider searches
 Neutralno contrbutons to Dark Matter, LHC and future Lnear Collder searches G.J. Gounars Unversty of Thessalonk, Collaboraton wth J. Layssac, P.I. Porfyrads, F.M. Renard and wth Th. Dakonds for the γz
Neutralno contrbutons to Dark Matter, LHC and future Lnear Collder searches G.J. Gounars Unversty of Thessalonk, Collaboraton wth J. Layssac, P.I. Porfyrads, F.M. Renard and wth Th. Dakonds for the γz
Thermodynamic Tables to accompany Modern Engineering Thermodynamics
 Thermodynamic Tables to accompany Modern Engineering Thermodynamics This page intentionally left blank Thermodynamic Tables to accompany Modern Engineering Thermodynamics Robert T. Balmer AMSTERDAM BOSTON
Thermodynamic Tables to accompany Modern Engineering Thermodynamics This page intentionally left blank Thermodynamic Tables to accompany Modern Engineering Thermodynamics Robert T. Balmer AMSTERDAM BOSTON
Ύγρανση και Αφύγρανση. Ψυχρομετρία. 21-Nov-16
 Ύγρανση και Αφύγρανση Ψυχρομετρία η μελέτη των ιδιοτήτων του υγρού αέρα δηλαδή του μίγματος αέρα-ατμού-νερού. Ένα βασικό πρόβλημα: Δεδομένων των P (bar) Πίεση T ( o C) Θερμοκρασία Υ (kg/kg ξβ) Υγρασία
Ύγρανση και Αφύγρανση Ψυχρομετρία η μελέτη των ιδιοτήτων του υγρού αέρα δηλαδή του μίγματος αέρα-ατμού-νερού. Ένα βασικό πρόβλημα: Δεδομένων των P (bar) Πίεση T ( o C) Θερμοκρασία Υ (kg/kg ξβ) Υγρασία
2 nd Law: m.s in + Q in /T in + S gen = ds/dt + m.s out + Q out /T out. S gen /ṁ = -(Q in /m)/t + Δs,
 P 444 5 xercises + f 4 6 Jan + Jan 5. kg ice at 6 K kg ater at 9 K. Heat Q at = is supplie y the surrunings. Specific heat ice : c i = 4 kj/kgk; ater c = 48 kj/kgk melting heat Δ m H = 4 kj/kg n La: m.s
P 444 5 xercises + f 4 6 Jan + Jan 5. kg ice at 6 K kg ater at 9 K. Heat Q at = is supplie y the surrunings. Specific heat ice : c i = 4 kj/kgk; ater c = 48 kj/kgk melting heat Δ m H = 4 kj/kg n La: m.s
Phys460.nb Solution for the t-dependent Schrodinger s equation How did we find the solution? (not required)
 Phys460.nb 81 ψ n (t) is still the (same) eigenstate of H But for tdependent H. The answer is NO. 5.5.5. Solution for the tdependent Schrodinger s equation If we assume that at time t 0, the electron starts
Phys460.nb 81 ψ n (t) is still the (same) eigenstate of H But for tdependent H. The answer is NO. 5.5.5. Solution for the tdependent Schrodinger s equation If we assume that at time t 0, the electron starts
Solar Neutrinos: Fluxes
 Solar Neutrinos: Fluxes pp chain Sun shines by : 4 p 4 He + e + + ν e + γ Solar Standard Model Fluxes CNO cycle e + N 13 =0.707MeV He 4 C 1 C 13 p p p p N 15 N 14 He 4 O 15 O 16 e + =0.997MeV O17
Solar Neutrinos: Fluxes pp chain Sun shines by : 4 p 4 He + e + + ν e + γ Solar Standard Model Fluxes CNO cycle e + N 13 =0.707MeV He 4 C 1 C 13 p p p p N 15 N 14 He 4 O 15 O 16 e + =0.997MeV O17
CHAPTER 25 SOLVING EQUATIONS BY ITERATIVE METHODS
 CHAPTER 5 SOLVING EQUATIONS BY ITERATIVE METHODS EXERCISE 104 Page 8 1. Find the positive root of the equation x + 3x 5 = 0, correct to 3 significant figures, using the method of bisection. Let f(x) =
CHAPTER 5 SOLVING EQUATIONS BY ITERATIVE METHODS EXERCISE 104 Page 8 1. Find the positive root of the equation x + 3x 5 = 0, correct to 3 significant figures, using the method of bisection. Let f(x) =
6.1. Dirac Equation. Hamiltonian. Dirac Eq.
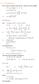 6.1. Dirac Equation Ref: M.Kaku, Quantum Field Theory, Oxford Univ Press (1993) η μν = η μν = diag(1, -1, -1, -1) p 0 = p 0 p = p i = -p i p μ p μ = p 0 p 0 + p i p i = E c 2 - p 2 = (m c) 2 H = c p 2
6.1. Dirac Equation Ref: M.Kaku, Quantum Field Theory, Oxford Univ Press (1993) η μν = η μν = diag(1, -1, -1, -1) p 0 = p 0 p = p i = -p i p μ p μ = p 0 p 0 + p i p i = E c 2 - p 2 = (m c) 2 H = c p 2
titanium laser beam volume to be heated joint titanium Fig. 5.1
 1 Lasers are often used to form precision-welded joints in titanium. To form one such joint it is first necessary to increase the temperature of the titanium to its melting point. Fig. 5.1 shows the joint
1 Lasers are often used to form precision-welded joints in titanium. To form one such joint it is first necessary to increase the temperature of the titanium to its melting point. Fig. 5.1 shows the joint
Mock Exam 7. 1 Hong Kong Educational Publishing Company. Section A 1. Reference: HKDSE Math M Q2 (a) (1 + kx) n 1M + 1A = (1) =
 Mock Eam 7 Mock Eam 7 Section A. Reference: HKDSE Math M 0 Q (a) ( + k) n nn ( )( k) + nk ( ) + + nn ( ) k + nk + + + A nk... () nn ( ) k... () From (), k...() n Substituting () into (), nn ( ) n 76n 76n
Mock Eam 7 Mock Eam 7 Section A. Reference: HKDSE Math M 0 Q (a) ( + k) n nn ( )( k) + nk ( ) + + nn ( ) k + nk + + + A nk... () nn ( ) k... () From (), k...() n Substituting () into (), nn ( ) n 76n 76n
MECHANICAL PROPERTIES OF MATERIALS
 MECHANICAL PROPERTIES OF MATERIALS! Simple Tension Test! The Stress-Strain Diagram! Stress-Strain Behavior of Ductile and Brittle Materials! Hooke s Law! Strain Energy! Poisson s Ratio! The Shear Stress-Strain
MECHANICAL PROPERTIES OF MATERIALS! Simple Tension Test! The Stress-Strain Diagram! Stress-Strain Behavior of Ductile and Brittle Materials! Hooke s Law! Strain Energy! Poisson s Ratio! The Shear Stress-Strain
DERIVATION OF MILES EQUATION FOR AN APPLIED FORCE Revision C
 DERIVATION OF MILES EQUATION FOR AN APPLIED FORCE Revision C By Tom Irvine Email: tomirvine@aol.com August 6, 8 Introduction The obective is to derive a Miles equation which gives the overall response
DERIVATION OF MILES EQUATION FOR AN APPLIED FORCE Revision C By Tom Irvine Email: tomirvine@aol.com August 6, 8 Introduction The obective is to derive a Miles equation which gives the overall response
Strain gauge and rosettes
 Strain gauge and rosettes Introduction A strain gauge is a device which is used to measure strain (deformation) on an object subjected to forces. Strain can be measured using various types of devices classified
Strain gauge and rosettes Introduction A strain gauge is a device which is used to measure strain (deformation) on an object subjected to forces. Strain can be measured using various types of devices classified
Model: MTZ22. Data. Cylinder count: 1 Displacement [m³/h]: 6,63 Cylinder capacity [cm³]: 38,1 RPM [min -1 ]: 2900 Weight [kg]: 21 Oil charge [dm³]: 1
![Model: MTZ22. Data. Cylinder count: 1 Displacement [m³/h]: 6,63 Cylinder capacity [cm³]: 38,1 RPM [min -1 ]: 2900 Weight [kg]: 21 Oil charge [dm³]: 1 Model: MTZ22. Data. Cylinder count: 1 Displacement [m³/h]: 6,63 Cylinder capacity [cm³]: 38,1 RPM [min -1 ]: 2900 Weight [kg]: 21 Oil charge [dm³]: 1](/thumbs/90/102819400.jpg) Technical data Data Cylinder count: 1 Displacement [m³/h]: 6,63 Cylinder capacity [cm³]: 38,1 RPM [min -1 ]: 2900 Weight [kg]: 21 Oil charge [dm³]: 1 Oil type: 160PZ Crankcase heater type: PTC 35 W Maximum
Technical data Data Cylinder count: 1 Displacement [m³/h]: 6,63 Cylinder capacity [cm³]: 38,1 RPM [min -1 ]: 2900 Weight [kg]: 21 Oil charge [dm³]: 1 Oil type: 160PZ Crankcase heater type: PTC 35 W Maximum
Démographie spatiale/spatial Demography
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΙΑΣ Démographie spatiale/spatial Demography Session 1: Introduction to spatial demography Basic concepts Michail Agorastakis Department of Planning & Regional Development Άδειες Χρήσης
ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΙΑΣ Démographie spatiale/spatial Demography Session 1: Introduction to spatial demography Basic concepts Michail Agorastakis Department of Planning & Regional Development Άδειες Χρήσης
UDZ Swirl diffuser. Product facts. Quick-selection. Swirl diffuser UDZ. Product code example:
 UDZ Swirl diffuser Swirl diffuser UDZ, which is intended for installation in a ventilation duct, can be used in premises with a large volume, for example factory premises, storage areas, superstores, halls,
UDZ Swirl diffuser Swirl diffuser UDZ, which is intended for installation in a ventilation duct, can be used in premises with a large volume, for example factory premises, storage areas, superstores, halls,
Variance of Trait in an Inbred Population. Variance of Trait in an Inbred Population
 Varance of Trat n an Inbred Populaton Varance of Trat n an Inbred Populaton Varance of Trat n an Inbred Populaton Revew of Mean Trat Value n Inbred Populatons We showed n the last lecture that for a populaton
Varance of Trat n an Inbred Populaton Varance of Trat n an Inbred Populaton Varance of Trat n an Inbred Populaton Revew of Mean Trat Value n Inbred Populatons We showed n the last lecture that for a populaton
Partial Differential Equations in Biology The boundary element method. March 26, 2013
 The boundary element method March 26, 203 Introduction and notation The problem: u = f in D R d u = ϕ in Γ D u n = g on Γ N, where D = Γ D Γ N, Γ D Γ N = (possibly, Γ D = [Neumann problem] or Γ N = [Dirichlet
The boundary element method March 26, 203 Introduction and notation The problem: u = f in D R d u = ϕ in Γ D u n = g on Γ N, where D = Γ D Γ N, Γ D Γ N = (possibly, Γ D = [Neumann problem] or Γ N = [Dirichlet
TSOKOS CHAPTER 6 TEST REVIEW
 PHYSICS I HONORS/PRE-IB PHYSICS Name: DEVIL PHYSICS Period: Date: # Marks: XX Raw Score: IB Curve: BADDEST CLASS ON CAMPUS TSOKOS CHAPTER 6 TEST REVIEW S1. Thermal energy is transferred through the glass
PHYSICS I HONORS/PRE-IB PHYSICS Name: DEVIL PHYSICS Period: Date: # Marks: XX Raw Score: IB Curve: BADDEST CLASS ON CAMPUS TSOKOS CHAPTER 6 TEST REVIEW S1. Thermal energy is transferred through the glass
RAC. Technical Data Book. RAC(Quantum) for North America (R410A, 60Hz, HP) Model : AR09/12/18/24KSFPDWQNCV AR09/12/18/24KSFPDWQXCV
 RAC Technical Data Book RAC(Quantum) for North America (R410A, 60Hz, HP) Model : AR09/12/18/24KSFPDWQNCV AR09/12/18/24KSFPDWQXCV History Version Modification Date Remark Ver 1.0 Release RAC (Quantum) TDB
RAC Technical Data Book RAC(Quantum) for North America (R410A, 60Hz, HP) Model : AR09/12/18/24KSFPDWQNCV AR09/12/18/24KSFPDWQXCV History Version Modification Date Remark Ver 1.0 Release RAC (Quantum) TDB
Symplecticity of the Störmer-Verlet algorithm for coupling between the shallow water equations and horizontal vehicle motion
 Symplectcty of the Störmer-Verlet algorthm for couplng between the shallow water equatons and horzontal vehcle moton by H. Alem Ardakan & T. J. Brdges Department of Mathematcs, Unversty of Surrey, Guldford
Symplectcty of the Störmer-Verlet algorthm for couplng between the shallow water equatons and horzontal vehcle moton by H. Alem Ardakan & T. J. Brdges Department of Mathematcs, Unversty of Surrey, Guldford
Heat exchanger. Type WT. For the reheating of airflows in rectangular ducting PD WT 1. 03/2017 DE/en
 X X testregistrierung Heat exchanger Type For the reheating of airflows in rectangular ducting Rectangular hot water heat exchanger for the reheating of airflows, suitable for VAV terminal units Type TVR,
X X testregistrierung Heat exchanger Type For the reheating of airflows in rectangular ducting Rectangular hot water heat exchanger for the reheating of airflows, suitable for VAV terminal units Type TVR,
Constant Elasticity of Substitution in Applied General Equilibrium
 Constant Elastct of Substtuton n Appled General Equlbru The choce of nput levels that nze the cost of producton for an set of nput prces and a fed level of producton can be epressed as n sty.. f Ltng for
Constant Elastct of Substtuton n Appled General Equlbru The choce of nput levels that nze the cost of producton for an set of nput prces and a fed level of producton can be epressed as n sty.. f Ltng for
ΑΕΡΙΑ ΚΑΤ ΚΑ Α Τ ΣΤ ΑΣΗ
 ΑΕΡΙΑ ΚΑΤΑΣΤΑΣΗ H αποστολή Pthfid Pathfinder το 1997 με το μικρό όχημα της ("Sojourner") ") ήταν δραστήρια στην Αρειανή επιφάνεια για αρκετούς μήνες, επιστρέφοντας μια μεγάλη συλλογή στοιχείων για το Αρειανό
ΑΕΡΙΑ ΚΑΤΑΣΤΑΣΗ H αποστολή Pthfid Pathfinder το 1997 με το μικρό όχημα της ("Sojourner") ") ήταν δραστήρια στην Αρειανή επιφάνεια για αρκετούς μήνες, επιστρέφοντας μια μεγάλη συλλογή στοιχείων για το Αρειανό
[1] P Q. Fig. 3.1
![[1] P Q. Fig. 3.1 [1] P Q. Fig. 3.1](/thumbs/79/80362156.jpg) 1 (a) Define resistance....... [1] (b) The smallest conductor within a computer processing chip can be represented as a rectangular block that is one atom high, four atoms wide and twenty atoms long. One
1 (a) Define resistance....... [1] (b) The smallest conductor within a computer processing chip can be represented as a rectangular block that is one atom high, four atoms wide and twenty atoms long. One
Enthalpy data for the reacting species are given in the table below. The activation energy decreases when the temperature is increased.
 Q1.This question is about the reaction given below. O(g) + H 2O(g) O 2(g) + H 2(g) Enthalpy data for the reacting species are given in the table below. Substance O(g) H 2O(g) O 2(g) H 2(g) ΔH / kj mol
Q1.This question is about the reaction given below. O(g) + H 2O(g) O 2(g) + H 2(g) Enthalpy data for the reacting species are given in the table below. Substance O(g) H 2O(g) O 2(g) H 2(g) ΔH / kj mol
RECIPROCATING COMPRESSOR CALCULATION SHEET ISOTHERMAL COMPRESSION Gas properties, flowrate and conditions. Compressor Calculation Sheet
 RECIPRCATING CMPRESSR CALCULATIN SHEET ISTHERMAL CMPRESSIN Gas properties, flowrate and conditions 1 Gas name Air Item or symbol Quantity Unit Item or symbol Quantity Unit Formula 2 Suction pressure, ps
RECIPRCATING CMPRESSR CALCULATIN SHEET ISTHERMAL CMPRESSIN Gas properties, flowrate and conditions 1 Gas name Air Item or symbol Quantity Unit Item or symbol Quantity Unit Formula 2 Suction pressure, ps
PROPERTY TABLES AND CHARTS (SI UNITS)
 PROPERY ABLES AND CHARS (SI UNIS) APPENDIX 1 able A 1 Molar mass, gas constant, and critical-point properties able A 2 Ideal-gas specific heats of varios common gases able A 3 Properties of common liqids,
PROPERY ABLES AND CHARS (SI UNIS) APPENDIX 1 able A 1 Molar mass, gas constant, and critical-point properties able A 2 Ideal-gas specific heats of varios common gases able A 3 Properties of common liqids,
ΜΜ917-Σχεδιασμός Ενεργειακών Συστημάτων
 ΜΜ917-Σχεδιασμός Ενεργειακών Συστημάτων Ψυκτικός Κύκλος Παρουσίαση και περιγραφή τρόπου λειτουργίας των επιμέρους τμημάτων Βιομηχανικής Ψυκτικής Μονάδας ημήτρης Τζιουρτζιούμης ρ. Μηχανολόγος Μηχανικός
ΜΜ917-Σχεδιασμός Ενεργειακών Συστημάτων Ψυκτικός Κύκλος Παρουσίαση και περιγραφή τρόπου λειτουργίας των επιμέρους τμημάτων Βιομηχανικής Ψυκτικής Μονάδας ημήτρης Τζιουρτζιούμης ρ. Μηχανολόγος Μηχανικός
Η ΕΠΙΔΡΑΣΗ ΤΗΣ ΑΙΘΑΝΟΛΗΣ,ΤΗΣ ΜΕΘΑΝΟΛΗΣ ΚΑΙ ΤΟΥ ΑΙΘΥΛΟΤΡΙΤΟΤΑΓΗ ΒΟΥΤΥΛΑΙΘΕΡΑ ΣΤΙΣ ΙΔΙΟΤΗΤΕΣ ΤΗΣ ΒΕΝΖΙΝΗΣ
 ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙΔΕΥΤΙΚΟ ΙΔΡΥΜΑ ΚΑΒΑΛΑΣ ΣΧΟΛΗ ΤΕΧΝΟΛΟΓΙΚΩΝ ΕΦΑΡΜΟΓΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ Η ΕΠΙΔΡΑΣΗ ΤΗΣ ΑΙΘΑΝΟΛΗΣ,ΤΗΣ ΜΕΘΑΝΟΛΗΣ ΚΑΙ ΤΟΥ ΑΙΘΥΛΟΤΡΙΤΟΤΑΓΗ ΒΟΥΤΥΛΑΙΘΕΡΑ ΣΤΙΣ ΙΔΙΟΤΗΤΕΣ ΤΗΣ ΒΕΝΖΙΝΗΣ ΟΝΟΜΑΤΕΠΩΝΥΜΟ
ΤΕΧΝΟΛΟΓΙΚΟ ΕΚΠΑΙΔΕΥΤΙΚΟ ΙΔΡΥΜΑ ΚΑΒΑΛΑΣ ΣΧΟΛΗ ΤΕΧΝΟΛΟΓΙΚΩΝ ΕΦΑΡΜΟΓΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ Η ΕΠΙΔΡΑΣΗ ΤΗΣ ΑΙΘΑΝΟΛΗΣ,ΤΗΣ ΜΕΘΑΝΟΛΗΣ ΚΑΙ ΤΟΥ ΑΙΘΥΛΟΤΡΙΤΟΤΑΓΗ ΒΟΥΤΥΛΑΙΘΕΡΑ ΣΤΙΣ ΙΔΙΟΤΗΤΕΣ ΤΗΣ ΒΕΝΖΙΝΗΣ ΟΝΟΜΑΤΕΠΩΝΥΜΟ
Example Sheet 3 Solutions
 Example Sheet 3 Solutions. i Regular Sturm-Liouville. ii Singular Sturm-Liouville mixed boundary conditions. iii Not Sturm-Liouville ODE is not in Sturm-Liouville form. iv Regular Sturm-Liouville note
Example Sheet 3 Solutions. i Regular Sturm-Liouville. ii Singular Sturm-Liouville mixed boundary conditions. iii Not Sturm-Liouville ODE is not in Sturm-Liouville form. iv Regular Sturm-Liouville note
PETROSKILLS COPYRIGHT
 Contents Solution Gas-Oil Ratio... 2... 2... 2... 2 Formation Volume Factor... 3... 3... 3... 3 Viscosity... 4... 4... 4... 4 Density... 5 Bubble Point Pressure... 5... 6... 6... 6 Compressibility... 6
Contents Solution Gas-Oil Ratio... 2... 2... 2... 2 Formation Volume Factor... 3... 3... 3... 3 Viscosity... 4... 4... 4... 4 Density... 5 Bubble Point Pressure... 5... 6... 6... 6 Compressibility... 6
Πανεπιστήµιο Κρήτης - Τµήµα Επιστήµης Υπολογιστών. ΗΥ-570: Στατιστική Επεξεργασία Σήµατος. ιδάσκων : Α. Μουχτάρης. εύτερη Σειρά Ασκήσεων.
 Πανεπιστήµιο Κρήτης - Τµήµα Επιστήµης Υπολογιστών ΗΥ-570: Στατιστική Επεξεργασία Σήµατος 2015 ιδάσκων : Α. Μουχτάρης εύτερη Σειρά Ασκήσεων Λύσεις Ασκηση 1. 1. Consder the gven expresson for R 1/2 : R 1/2
Πανεπιστήµιο Κρήτης - Τµήµα Επιστήµης Υπολογιστών ΗΥ-570: Στατιστική Επεξεργασία Σήµατος 2015 ιδάσκων : Α. Μουχτάρης εύτερη Σειρά Ασκήσεων Λύσεις Ασκηση 1. 1. Consder the gven expresson for R 1/2 : R 1/2
 v R = c v 2 v P = R v = v 3 v n P = n 3 v 1 v = n 1 n P 1 P = c 1 c P 1 P = n 1 c 1 n 3 c = v 1 v 3 3 3 c P P = c v 4.5 P = c v 4 P = c v 3.5 v 1 = 24 v 2 = 18 c 1 c 1 4.5 = v 1 c 2 v 4.5 2 = 244.5 = 3.65
v R = c v 2 v P = R v = v 3 v n P = n 3 v 1 v = n 1 n P 1 P = c 1 c P 1 P = n 1 c 1 n 3 c = v 1 v 3 3 3 c P P = c v 4.5 P = c v 4 P = c v 3.5 v 1 = 24 v 2 = 18 c 1 c 1 4.5 = v 1 c 2 v 4.5 2 = 244.5 = 3.65
ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΤΜΗΜΑ ΧΗΜΙΚΩΝ ΜΗΧΑΝΙΚΩΝ ΕΡΓΑΣΤΗΡΙΟ ΣΧΕΔΙΑΣΜΟΥ & ΑΝΑΛΥΣΗΣ ΔΙΕΡΓΑΣΙΩΝ
 ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΤΜΗΜΑ ΧΗΜΙΚΩΝ ΜΗΧΑΝΙΚΩΝ ΕΡΓΑΣΤΗΡΙΟ ΣΧΕΔΙΑΣΜΟΥ & ΑΝΑΛΥΣΗΣ ΔΙΕΡΓΑΣΙΩΝ Μάθημα: ΤΕΧΝΙΚΗ ΦΥΣΙΚΩΝ ΔΙΕΡΓΑΣΙΩΝ ΙΙ 6ο Εξάμηνο Χημικών Μηχανικών ΞΗΡΑΝΤΗΡΕΣ 1 Characteristics of Food Dryers
ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΤΜΗΜΑ ΧΗΜΙΚΩΝ ΜΗΧΑΝΙΚΩΝ ΕΡΓΑΣΤΗΡΙΟ ΣΧΕΔΙΑΣΜΟΥ & ΑΝΑΛΥΣΗΣ ΔΙΕΡΓΑΣΙΩΝ Μάθημα: ΤΕΧΝΙΚΗ ΦΥΣΙΚΩΝ ΔΙΕΡΓΑΣΙΩΝ ΙΙ 6ο Εξάμηνο Χημικών Μηχανικών ΞΗΡΑΝΤΗΡΕΣ 1 Characteristics of Food Dryers
Jesse Maassen and Mark Lundstrom Purdue University November 25, 2013
 Notes on Average Scattering imes and Hall Factors Jesse Maassen and Mar Lundstrom Purdue University November 5, 13 I. Introduction 1 II. Solution of the BE 1 III. Exercises: Woring out average scattering
Notes on Average Scattering imes and Hall Factors Jesse Maassen and Mar Lundstrom Purdue University November 5, 13 I. Introduction 1 II. Solution of the BE 1 III. Exercises: Woring out average scattering
n b n Mn3C = n Mn /3 = 0,043 mol free Fe n Fe = n Fe 3n Fe3C = 16,13 mol, no free C b Mn3C = -Δ f G for Mn 3 C +3 b Mn + b C = 1907,9 kj/mol
 PET 44304 015 Exercses 3+4 f 4 10 Feb + 13 Feb 015 1. deal gas: Δs = s - s 1 = c p ln(t /T 1 ) - R ln(p /p 1 ) (T
PET 44304 015 Exercses 3+4 f 4 10 Feb + 13 Feb 015 1. deal gas: Δs = s - s 1 = c p ln(t /T 1 ) - R ln(p /p 1 ) (T
Συστήματα Βιομηχανικών Διεργασιών 6ο εξάμηνο
 Συστήματα Βιομηχανικών Διεργασιών 6ο εξάμηνο Μέρος 1 ο : Εισαγωγικά (διαστ., πυκν., θερμ., πίεση, κτλ.) Μέρος 2 ο : Ισοζύγια μάζας Μέρος 3 ο : 8 ο μάθημα Εκτός ύλης ΔΠΘ-ΜΠΔ Συστήματα Βιομηχανικών Διεργασιών
Συστήματα Βιομηχανικών Διεργασιών 6ο εξάμηνο Μέρος 1 ο : Εισαγωγικά (διαστ., πυκν., θερμ., πίεση, κτλ.) Μέρος 2 ο : Ισοζύγια μάζας Μέρος 3 ο : 8 ο μάθημα Εκτός ύλης ΔΠΘ-ΜΠΔ Συστήματα Βιομηχανικών Διεργασιών
Matrices and Determinants
 Matrices and Determinants SUBJECTIVE PROBLEMS: Q 1. For what value of k do the following system of equations possess a non-trivial (i.e., not all zero) solution over the set of rationals Q? x + ky + 3z
Matrices and Determinants SUBJECTIVE PROBLEMS: Q 1. For what value of k do the following system of equations possess a non-trivial (i.e., not all zero) solution over the set of rationals Q? x + ky + 3z
Group 30. Contents.
 Group 30 Contents Pump type Page Pump type Page Pump type Page 30A(C)...X002H 4 30A(C)...X013H 5 30A(C)...X068H 6 30A(C)...X068HU 7 30A(C)...X136H 8 30A(C)...X136Y 9 30A(C)...X146H 10 30A(C)...X160H 11
Group 30 Contents Pump type Page Pump type Page Pump type Page 30A(C)...X002H 4 30A(C)...X013H 5 30A(C)...X068H 6 30A(C)...X068HU 7 30A(C)...X136H 8 30A(C)...X136Y 9 30A(C)...X146H 10 30A(C)...X160H 11
Inverse trigonometric functions & General Solution of Trigonometric Equations. ------------------ ----------------------------- -----------------
 Inverse trigonometric functions & General Solution of Trigonometric Equations. 1. Sin ( ) = a) b) c) d) Ans b. Solution : Method 1. Ans a: 17 > 1 a) is rejected. w.k.t Sin ( sin ) = d is rejected. If sin
Inverse trigonometric functions & General Solution of Trigonometric Equations. 1. Sin ( ) = a) b) c) d) Ans b. Solution : Method 1. Ans a: 17 > 1 a) is rejected. w.k.t Sin ( sin ) = d is rejected. If sin
Answers to practice exercises
 Answers to practice exercises Chapter Exercise (Page 5). 9 kg 2. 479 mm. 66 4. 565 5. 225 6. 26 7. 07,70 8. 4 9. 487 0. 70872. $5, Exercise 2 (Page 6). (a) 468 (b) 868 2. (a) 827 (b) 458. (a) 86 kg (b)
Answers to practice exercises Chapter Exercise (Page 5). 9 kg 2. 479 mm. 66 4. 565 5. 225 6. 26 7. 07,70 8. 4 9. 487 0. 70872. $5, Exercise 2 (Page 6). (a) 468 (b) 868 2. (a) 827 (b) 458. (a) 86 kg (b)
ME 365: SYSTEMS, MEASUREMENTS, AND CONTROL (SMAC) I
 ME 365: SYSTEMS, MEASUREMENTS, AND CONTROL SMAC) I Dynamicresponseof 2 nd ordersystem Prof.SongZhangMEG088) Solutions to ODEs Forann@thorderLTIsystem a n yn) + a n 1 y n 1) ++ a 1 "y + a 0 y = b m u m)
ME 365: SYSTEMS, MEASUREMENTS, AND CONTROL SMAC) I Dynamicresponseof 2 nd ordersystem Prof.SongZhangMEG088) Solutions to ODEs Forann@thorderLTIsystem a n yn) + a n 1 y n 1) ++ a 1 "y + a 0 y = b m u m)
Assalamu `alaikum wr. wb.
 LUMP SUM Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. LUMP SUM Lump sum lump sum lump sum. lump sum fixed price lump sum lump
LUMP SUM Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. LUMP SUM Lump sum lump sum lump sum. lump sum fixed price lump sum lump
LUNGOO R. Control Engineering for Development of a Mechanical Ventilator for ICU Use Spontaneous Breathing Lung Simulator LUNGOO
 ol. 6, No.3 7/(7) IU UNGOO ontrol Engineering for Development of a Mechanical entilator for IU Use Spontaneous Breathing ung Simulator UNGOO Kenji OZAKI*yoji IIDA*Kazutoshi SOGA* and Yasuhiro UENO* A lung
ol. 6, No.3 7/(7) IU UNGOO ontrol Engineering for Development of a Mechanical entilator for IU Use Spontaneous Breathing ung Simulator UNGOO Kenji OZAKI*yoji IIDA*Kazutoshi SOGA* and Yasuhiro UENO* A lung
RECIPROCATING COMPRESSOR CALCULATION SHEET
 RECIPRCATING CMPRESSR CALCULATIN SHEET WITH INTERCLER Gas properties, flowrate and conditions 1 Gas name Air Item or symbol Quantity Unit Item or symbol Quantity Unit 2 Suction pressure, ps 1.013 bar A
RECIPRCATING CMPRESSR CALCULATIN SHEET WITH INTERCLER Gas properties, flowrate and conditions 1 Gas name Air Item or symbol Quantity Unit Item or symbol Quantity Unit 2 Suction pressure, ps 1.013 bar A
Radiation Stress Concerned with the force (or momentum flux) exerted on the right hand side of a plane by water on the left hand side of the plane.
 upplement on Radiation tress and Wave etup/et down Radiation tress oncerned wit te force (or momentum flu) eerted on te rit and side of a plane water on te left and side of te plane. plane z "Radiation
upplement on Radiation tress and Wave etup/et down Radiation tress oncerned wit te force (or momentum flu) eerted on te rit and side of a plane water on te left and side of te plane. plane z "Radiation
( ) Sine wave travelling to the right side
 SOUND WAVES (1) Sound wave: Varia2on of density of air Change in density at posi2on x and 2me t: Δρ(x,t) = Δρ m sin kx ωt (2) Sound wave: Varia2on of pressure Bulk modulus B is defined as: B = V dp dv
SOUND WAVES (1) Sound wave: Varia2on of density of air Change in density at posi2on x and 2me t: Δρ(x,t) = Δρ m sin kx ωt (2) Sound wave: Varia2on of pressure Bulk modulus B is defined as: B = V dp dv
The Simply Typed Lambda Calculus
 Type Inference Instead of writing type annotations, can we use an algorithm to infer what the type annotations should be? That depends on the type system. For simple type systems the answer is yes, and
Type Inference Instead of writing type annotations, can we use an algorithm to infer what the type annotations should be? That depends on the type system. For simple type systems the answer is yes, and
RAC. Technical Data Book. RAC(Max Heat) for North America (R410A, 60Hz, HP) Model : AR09/12KSWSPWKNCV AR09/12KSWSPWKXCV
 RAC Technical Data Book RAC(Max Heat) for North America (R410A, 60Hz, HP) Model : AR09/12KSWSPWKNCV AR09/12KSWSPWKXCV History Version Modification Date Remark Ver.1.0 Release RAC (Max Heat) TDB for North
RAC Technical Data Book RAC(Max Heat) for North America (R410A, 60Hz, HP) Model : AR09/12KSWSPWKNCV AR09/12KSWSPWKXCV History Version Modification Date Remark Ver.1.0 Release RAC (Max Heat) TDB for North
MSM Men who have Sex with Men HIV -
 ,**, The Japanese Society for AIDS Research The Journal of AIDS Research HIV,0 + + + + +,,, +, : HIV : +322,*** HIV,0,, :., n,0,,. + 2 2, CD. +3-ml n,, AIDS 3 ARC 3 +* 1. A, MSM Men who have Sex with Men
,**, The Japanese Society for AIDS Research The Journal of AIDS Research HIV,0 + + + + +,,, +, : HIV : +322,*** HIV,0,, :., n,0,,. + 2 2, CD. +3-ml n,, AIDS 3 ARC 3 +* 1. A, MSM Men who have Sex with Men
P AND P. P : actual probability. P : risk neutral probability. Realtionship: mutual absolute continuity P P. For example:
 (B t, S (t) t P AND P,..., S (p) t ): securities P : actual probability P : risk neutral probability Realtionship: mutual absolute continuity P P For example: P : ds t = µ t S t dt + σ t S t dw t P : ds
(B t, S (t) t P AND P,..., S (p) t ): securities P : actual probability P : risk neutral probability Realtionship: mutual absolute continuity P P For example: P : ds t = µ t S t dt + σ t S t dw t P : ds
Swirl diffusers, Variable swirl diffusers Swirl diffusers
 , Variable swirl diffusers Swirl diffuser OD-9 Square or round front mask Square or radial deflector arrangement Plastic deflectors Possible volume control damper in spigot Foam sealing on the flange St
, Variable swirl diffusers Swirl diffuser OD-9 Square or round front mask Square or radial deflector arrangement Plastic deflectors Possible volume control damper in spigot Foam sealing on the flange St
Right Rear Door. Let's now finish the door hinge saga with the right rear door
 Right Rear Door Let's now finish the door hinge saga with the right rear door You may have been already guessed my steps, so there is not much to describe in detail. Old upper one file:///c /Documents
Right Rear Door Let's now finish the door hinge saga with the right rear door You may have been already guessed my steps, so there is not much to describe in detail. Old upper one file:///c /Documents
SMD Power Inductor. - SPRH127 Series. Marking. 1 Marking Outline: 1 Appearance and dimensions (mm)
 Marking Outline: Low DCR, high rated current. Magnetic shielded structure Lead free product, RoHS compliant. RoHS Carrier tape packing, suitable for SMT process. SMT Widely used in buck converter, laptop,
Marking Outline: Low DCR, high rated current. Magnetic shielded structure Lead free product, RoHS compliant. RoHS Carrier tape packing, suitable for SMT process. SMT Widely used in buck converter, laptop,
ANSWERSHEET (TOPIC = DIFFERENTIAL CALCULUS) COLLECTION #2. h 0 h h 0 h h 0 ( ) g k = g 0 + g 1 + g g 2009 =?
 Teko Classes IITJEE/AIEEE Maths by SUHAAG SIR, Bhopal, Ph (0755) 3 00 000 www.tekoclasses.com ANSWERSHEET (TOPIC DIFFERENTIAL CALCULUS) COLLECTION # Question Type A.Single Correct Type Q. (A) Sol least
Teko Classes IITJEE/AIEEE Maths by SUHAAG SIR, Bhopal, Ph (0755) 3 00 000 www.tekoclasses.com ANSWERSHEET (TOPIC DIFFERENTIAL CALCULUS) COLLECTION # Question Type A.Single Correct Type Q. (A) Sol least
Question 8.1 Noting that h = h(t) for ideal gases, hence, h 1 = h 2 since T 1 = T 2 = 25 C. From the steady energy equation: P 2
 Quetion 8. Noting tt ( or idel ge, ence, ince 5. From te tedy energy eqution: Q & & 5 k e rte o entroy cnge o ir i: Q Q& ir 5 k ΔS& AIR ir 0.0889 k/k 98 K cont. 0 y Quetion 8. e roertie o te ter re 00
Quetion 8. Noting tt ( or idel ge, ence, ince 5. From te tedy energy eqution: Q & & 5 k e rte o entroy cnge o ir i: Q Q& ir 5 k ΔS& AIR ir 0.0889 k/k 98 K cont. 0 y Quetion 8. e roertie o te ter re 00
Bounding Nonsplitting Enumeration Degrees
 Bounding Nonsplitting Enumeration Degrees Thomas F. Kent Andrea Sorbi Università degli Studi di Siena Italia July 18, 2007 Goal: Introduce a form of Σ 0 2-permitting for the enumeration degrees. Till now,
Bounding Nonsplitting Enumeration Degrees Thomas F. Kent Andrea Sorbi Università degli Studi di Siena Italia July 18, 2007 Goal: Introduce a form of Σ 0 2-permitting for the enumeration degrees. Till now,
On a four-dimensional hyperbolic manifold with finite volume
 BULETINUL ACADEMIEI DE ŞTIINŢE A REPUBLICII MOLDOVA. MATEMATICA Numbers 2(72) 3(73), 2013, Pages 80 89 ISSN 1024 7696 On a four-dimensional hyperbolic manifold with finite volume I.S.Gutsul Abstract. In
BULETINUL ACADEMIEI DE ŞTIINŢE A REPUBLICII MOLDOVA. MATEMATICA Numbers 2(72) 3(73), 2013, Pages 80 89 ISSN 1024 7696 On a four-dimensional hyperbolic manifold with finite volume I.S.Gutsul Abstract. In
RECIPROCATING COMPRESSOR CALCULATION SHEET
 Gas properties, flowrate and conditions 1 Gas name Air RECIPRCATING CMPRESSR CALCULATIN SHEET WITH INTERCLER ( Sheet : 1. f 4.) Item or symbol Quantity Unit Item or symbol Quantity Unit 2 Suction pressure,
Gas properties, flowrate and conditions 1 Gas name Air RECIPRCATING CMPRESSR CALCULATIN SHEET WITH INTERCLER ( Sheet : 1. f 4.) Item or symbol Quantity Unit Item or symbol Quantity Unit 2 Suction pressure,
PETROSKILLS COPYRIGHT
 Contents Dew Point... 2 SI Conversions... 2 Output... 2 Input... 2 Solution Condensate-Oil Ratio... 3 SI Conversions... 3 Output... 3 Input... 3 Gas Density... 4 SI Conversions... 5 Output... 5 Input...
Contents Dew Point... 2 SI Conversions... 2 Output... 2 Input... 2 Solution Condensate-Oil Ratio... 3 SI Conversions... 3 Output... 3 Input... 3 Gas Density... 4 SI Conversions... 5 Output... 5 Input...
Areas and Lengths in Polar Coordinates
 Kiryl Tsishchanka Areas and Lengths in Polar Coordinates In this section we develop the formula for the area of a region whose boundary is given by a polar equation. We need to use the formula for the
Kiryl Tsishchanka Areas and Lengths in Polar Coordinates In this section we develop the formula for the area of a region whose boundary is given by a polar equation. We need to use the formula for the
Notes on the Open Economy
 Notes on the Open Econom Ben J. Heijdra Universit of Groningen April 24 Introduction In this note we stud the two-countr model of Table.4 in more detail. restated here for convenience. The model is Table.4.
Notes on the Open Econom Ben J. Heijdra Universit of Groningen April 24 Introduction In this note we stud the two-countr model of Table.4 in more detail. restated here for convenience. The model is Table.4.
1 Complete Set of Grassmann States
 Physcs 610 Homework 8 Solutons 1 Complete Set of Grassmann States For Θ, Θ, Θ, Θ each ndependent n-member sets of Grassmann varables, and usng the summaton conventon ΘΘ Θ Θ Θ Θ, prove the dentty e ΘΘ dθ
Physcs 610 Homework 8 Solutons 1 Complete Set of Grassmann States For Θ, Θ, Θ, Θ each ndependent n-member sets of Grassmann varables, and usng the summaton conventon ΘΘ Θ Θ Θ Θ, prove the dentty e ΘΘ dθ
Approximation of distance between locations on earth given by latitude and longitude
 Approximation of distance between locations on earth given by latitude and longitude Jan Behrens 2012-12-31 In this paper we shall provide a method to approximate distances between two points on earth
Approximation of distance between locations on earth given by latitude and longitude Jan Behrens 2012-12-31 In this paper we shall provide a method to approximate distances between two points on earth
Lowara SPECIFICATIONS
 SH Series Centrifugal pumps entirely made of AISI 36 stainless steel according to EN 733 (ex DIN 24255). Designed to pump hot, cold and moderately aggressive liquids. Available versions: SHE Close-coupled
SH Series Centrifugal pumps entirely made of AISI 36 stainless steel according to EN 733 (ex DIN 24255). Designed to pump hot, cold and moderately aggressive liquids. Available versions: SHE Close-coupled
CONCENTRATIVE PROPERTIES OF AQUEOUS SOLUTIONS: DENSITY, REFRACTIVE INDEX, FREEZING POINT DEPRESSION, AND VISCOSITY
 DENSITY, REFRACTIVE INDEX, FREEZING POINT DEPRESSION, AND VISCOSITY This table gives properties of aqueous solutions of 66 substances as a function of concentration. All data refer to a temperature of
DENSITY, REFRACTIVE INDEX, FREEZING POINT DEPRESSION, AND VISCOSITY This table gives properties of aqueous solutions of 66 substances as a function of concentration. All data refer to a temperature of
Capacitors - Capacitance, Charge and Potential Difference
 Capacitors - Capacitance, Charge and Potential Difference Capacitors store electric charge. This ability to store electric charge is known as capacitance. A simple capacitor consists of 2 parallel metal
Capacitors - Capacitance, Charge and Potential Difference Capacitors store electric charge. This ability to store electric charge is known as capacitance. A simple capacitor consists of 2 parallel metal
Correction Table for an Alcoholometer Calibrated at 20 o C
 An alcoholometer is a device that measures the concentration of ethanol in a water-ethanol mixture (often in units of %abv percent alcohol by volume). The depth to which an alcoholometer sinks in a water-ethanol
An alcoholometer is a device that measures the concentration of ethanol in a water-ethanol mixture (often in units of %abv percent alcohol by volume). The depth to which an alcoholometer sinks in a water-ethanol
Απόκριση σε Μοναδιαία Ωστική Δύναμη (Unit Impulse) Απόκριση σε Δυνάμεις Αυθαίρετα Μεταβαλλόμενες με το Χρόνο. Απόστολος Σ.
 Απόκριση σε Δυνάμεις Αυθαίρετα Μεταβαλλόμενες με το Χρόνο The time integral of a force is referred to as impulse, is determined by and is obtained from: Newton s 2 nd Law of motion states that the action
Απόκριση σε Δυνάμεις Αυθαίρετα Μεταβαλλόμενες με το Χρόνο The time integral of a force is referred to as impulse, is determined by and is obtained from: Newton s 2 nd Law of motion states that the action
EE101: Resonance in RLC circuits
 EE11: Resonance in RLC circuits M. B. Patil mbatil@ee.iitb.ac.in www.ee.iitb.ac.in/~sequel Deartment of Electrical Engineering Indian Institute of Technology Bombay I V R V L V C I = I m = R + jωl + 1/jωC
EE11: Resonance in RLC circuits M. B. Patil mbatil@ee.iitb.ac.in www.ee.iitb.ac.in/~sequel Deartment of Electrical Engineering Indian Institute of Technology Bombay I V R V L V C I = I m = R + jωl + 1/jωC
TRIAXIAL TEST, CORPS OF ENGINEERS FORMAT
 TRIAXIAL TEST, CORPS OF ENGINEERS FORMAT .5 C, φ, deg Tan(φ) Total.7 2.2 Effective.98 8.33 Shear,.5.5.5 2 2.5 3 Total Normal, Effective Normal, Deviator,.5.25.75.5.25 2.5 5 7.5 Axial Strain, % Type of
TRIAXIAL TEST, CORPS OF ENGINEERS FORMAT .5 C, φ, deg Tan(φ) Total.7 2.2 Effective.98 8.33 Shear,.5.5.5 2 2.5 3 Total Normal, Effective Normal, Deviator,.5.25.75.5.25 2.5 5 7.5 Axial Strain, % Type of
