* To whom correspondence should be addressed.
|
|
|
- Φιλύρη Μπλέτσας
- 7 χρόνια πριν
- Προβολές:
Transcript
1 SUPPORTING INFORMATION Computational design of a pincer phosphinito vanadium ((OPO)V) propane monoxygenation homogeneous catalyst based on the reduction-coupled oxo activation (ROA) mechanism Ross Fu, William A. Goddard III,* Mu-Jeng Cheng, and Robert J. Nielsen Materials and Process Simulation Center (139-74) California Institute of Technology, Pasadena, CA USA. * To whom correspondence should be addressed. wag@wag.caltech.edu TABLE OF CONTENTS A. Reformation of starting V V species from V V peroxide and V III via protonation/oxidation 2 B. Tables of (OPO)V species studied..5 C. Suggestions for the experimental synthesis of (OPO)V species...23 D. References.25
2 A. Reformation of starting V V species from V V peroxide and V III via protonation/oxidation One possibility for how a (OPO)V V (O2) peroxide species, such as (OPO) f V V (HOiPr) t (O2) s (H2O) c (278) or (OPO) f V V (η 2 -O2) c (H2O)2 st (409), might be converted back to the starting V V species 6 is via the direct protonation of the peroxide ligand off, releasing H2O2. If the peroxide 278 is protonated, a (OPO)V V (OiPr) species will be produced, which can then lose HOiPr to form the starting species 6 (Scheme S1). Species involved in the protonation of the peroxide ligand in (OPO)V V (O2)(OiPr) are listed in Table S6, and the resulting (OPO)V V (OiPr) species are listed in Table S9. The lowest energy (OPO)V V (OiPr) species is five-coordinate (OPO)V V O s (HOiPr) a (459), but the six-coordinate species (OPO) f HV V O s (OiPr) c (H2O) t (477) is only 1.0 kcal/mol higher in energy. Scheme S1. The lowest energy (OPO)V V (O2) peroxide species, (OPO) f V V (η 2 -O2) c (H2O)2 st (409), can isomerize to form a (OPO)V V (HOOH) species and then lose H2O2 to form 6. All ΔGrel values are in kcal/mol and relative to 6 at the beginning of the reaction. Note that 6 in this scheme is at 12.9 because ΔG = 12.9 kcal/mol for the reaction iprh + O2 + H2O iproh + H2O2. By contrast, if the peroxide 409 is protonated off, the starting species 6 is formed directly (Scheme S2). Species involved in the protonation of 409 are listed in Table S7, and the lowest energy pathway for such a process goes through (OPO) f HV V O s (HOOH) c (OH) t (441), the lowest energy (OPO)V V (HOOH) species. Scheme S2. The lowest energy (OPO)V V (O2) peroxide species, (OPO) f V V (η 2 -O2) c (H2O)2 st (409), can isomerize to form a (OPO)V V (HOOH) species and then lose H2O2 to form 6. All ΔGrel values are in kcal/mol and relative to 6 at the beginning of the reaction. Note that 6 in this scheme is at 12.9 because ΔG = 12.9 kcal/mol for the reaction iprh + O2 + H2O iproh + H2O2.
3 By comparing Schemes S1 and S2, we see that it is more favorable for 409 to have its peroxide protonated off as in Scheme S2. The highest energy species in this pathway is 441 at 9.8 kcal/mol relative to the starting complex 6. By contrast, species 369 in Scheme S1 is at 9.5 kcal/mol, and 461 may be even higher in energy as we were unable to find its optimized structure. We thus conclude that it is most likely for 278 to first lose its isopropanol ligand to form 409 before seeing its peroxide ligand protonated off, and this is reflected in the first row of our Scheme 7. The H2O2 released in Schemes S1 and S2 can then coordinate with another equivalent of (OPO)V III to form a (OPO)V III (HOOH) species, which can be intramolecularly converted a second (OPO)V V species. We note that there are two types of (OPO)V III species: (OPO)V III (OiPr) species, which contain an isoproxo ligand ( , Table S4), and (OPO)V III species that only contain hydroxo/aqua ligands ( , Table S5). If H2O2 coordinates with a (OPO)V III (OiPr) species, a (OPO)V III (OiPr)(H2O2) species ( , Table S10) is then formed. The lowest energy isomer is six-coordinate 3 [(OPO) f HV III (HOiPr) s (OOH) c (H2O) t ] (503), and H2O/H2O2 ligand exchange from 168 to 503 is downhill in free energy by 2.2 kcal/mol. This can then convert to (OPO)V V O s (HOiPr) a (459) via a V V-III transition state ( , Table S11). The lowest such transition state found is 3 [(OPO)HV(HOiPr) a (O OH) s ] (516). Note that triplet transition states were found to be lower in energy than singlet transition states with broken-symmetry correction; this is likely due to the extreme early-state nature of the transition states. Finally, (OPO)V V O s (HOiPr) a (459) can exchange HOiPr for an aqua ligand to regenerate the starting complex 6. The overall process for this oxidation is shown in Scheme S3. Scheme S3. Hydrogen peroxide released at the end of schemes S1 and S2 can oxidize a (OPO)V III (OiPr) species such as 168 to form an (OPO)V V (OiPr) species such as 459, which can release HOiPr and bind to an aqua ligand (see Scheme S1 for details) to reform the starting species 6. All ΔGrel values are in kcal/mol and relative to 6 at the beginning of the reaction. Note that 6 in this scheme is at 69.9 kcal/mol as it represents the second equivalent of 6 produced;
4 the first equivalent is at 12.9 kcal/mol (Schemes S1 and S2) and their sum, 82.9 kcal/mol, is the free energy change for the reaction 2iPrH + O2 2iPrOH. If H2O2 instead coordinates with a (OPO)V III species with only oxo/hydroxo/aqua ligands, a (OPO)V III (H2O2) species ( , Table S12) is then formed. The lowest energy isomer is sixcoordinate 3 [(OPO) f HV III (OOH) c (H2O)2 st ] (538). This can then convert to the starting (OPO)V V species 6 via a V V-III transition state ( , Table S13). The lowest such transition state found is 3 [(OPO)HV(O OH) s (H2O) a ] (542). Note that, as in the OiPr case, triplet transition states were found to be lower in energy than singlet transition states with broken-symmetry correction. The overall process for this oxidation is shown in Scheme S4. Scheme S4. Hydrogen peroxide released at the end of schemes S1 and S2 can oxidize a (OPO)V III species such as 200 to reform the starting species 6. All ΔGrel values are in kcal/mol and relative to 6 at the beginning of the reaction. Note that 6 in this scheme is at 69.9 kcal/mol as it represents the second equivalent of 6 produced; the first equivalent is at 12.9 kcal/mol (Schemes S1 and S2) and their sum, 82.9 kcal/mol, is the free energy change for the reaction 2iPrH + O2 2iPrOH. By comparing Schemes S3 and S4, we see that 516 in Scheme S3 is the lower-energy transition state. Hence we conclude that the released H2O2 first oxidizes a (OPO)V III (OiPr) species such as 168 to form 459 before seeing its isopropanol ligand replaced with an aqua, and this is reflected in the second and third rows of our Scheme 7.
5 B. Tables of (OPO)V species studied Table S1a. Comprehensive compilation of all (OPO)V V species studied, along with their gasphase energies (Egas) and aqueous free energies (Gaq), both in hartrees; and their aqueous free energies relative to each other (ΔGrel), in kcal/mol. For some entries, geometry optimisation led to the formation of a lower-energy isomer. The energies for these entries are not reported. Entry Coord. Species Egas Gaq ΔGrel 1 5 (OPO)V V Cl (OPO)V V (OH) (OPO)V V =O (OPO)V V O s (H2O) a (OPO)V V O a (H2O) s (OPO)HV V O s (OH) a (OPO)HV V O a (OH) s geometry unstable N/A 8 5 [(OPO)HV V O s (H2O) a ] [(OPO)HV V O a (H2O) s ] + geometry unstable N/A 10 5 [(OPO)V V O s (OH) a ] [(OPO)V V O a (OH) s ] [(OPO)V V O2] (OPO) f V V O c (H2O)2 st (OPO) f V V O s (H2O)2 ct (OPO) f V V O t (H2O)2 cs (OPO) f HV V O c (OH) s (H2O) t (OPO) f HV V O c (OH) t (H2O) s (OPO) f HV V O s (OH) c (H2O) t (OPO) f HV V O s (OH) t (H2O) c (OPO) f HV V O t (OH) c (H2O) s (OPO) f HV V O t (OH) s (H2O) c [(OPO) f HV V O c (OH)2 st ] [(OPO) f HV V O s (OH)2 ct ] [(OPO) f HV V O t (OH)2 cs ] [(OPO) f V V O c (OH) s (H2O) t ] [(OPO) f V V O c (OH) t (H2O) s ] [(OPO) f V V O s (OH) c (H2O) t ] [(OPO) f V V O s (OH) t (H2O) c ] [(OPO) f V V O t (OH) c (H2O) s ] [(OPO) f V V O t (OH) s (H2O) c ] [(OPO) f V V O c (OH)2 st ] [(OPO) f V V O s (OH)2 ct ]
6 Table S1b. Continuation from Table S1a. Entry Coord. Species Egas Gaq ΔGrel 33 6 [(OPO) f V V O t (OH)2 cs ] [(OPO) f V V (H2O) c (O)2 st ] [(OPO) f V V (H2O) s (O)2 ct ] [(OPO) f V V (H2O) t (O)2 ct ] [(OPO) f HV V (OH) c (O)2 st ] [(OPO) f HV V (OH) s (O)2 ct ] [(OPO) f HV V (OH) t (O)2 cs ] (OPO) m V V O s (H2O)2 ta (OPO) m V V O t (H2O)2 sa (OPO) m V V O a (H2O)2 st (OPO) m HV V O s (OH) t (H2O) a geometry unstable N/A 44 6 (OPO) m HV V O s (OH) a (H2O) t geometry unstable N/A 45 6 (OPO) m HV V O t (OH) s (H2O) a geometry unstable N/A 46 6 (OPO) m HV V O t (OH) a (H2O) s (OPO) m HV V O a (OH) s (H2O) t (OPO) m HV V O a (OH) t (H2O) s [(OPO) m V V O s (OH) t (H2O) a ] [(OPO) m V V O s (OH) t (H2O) a ] geometry unstable N/A 51 6 [(OPO) m V V O s (OH) a (H2O) t ] geometry unstable N/A 52 6 [(OPO) m V V O t (OH) s (H2O) a ] [(OPO) m V V O t (OH) a (H2O) s ] geometry unstable N/A 54 6 [(OPO) m V V O a (OH) s (H2O) t ] [(OPO) m HV V O s (OH)2 ta ] [(OPO) m HV V O t (OH)2 sa ] [(OPO) m HV V O a (OH)2 st ] [(OPO) m V V O s (OH)2 ta ] [(OPO) m V V O t (OH)2 sa ] [(OPO) m V V O a (OH)2 st ] [(OPO) m V V (H2O) s (O)2 ta ] 2 geometry unstable N/A 62 6 [(OPO) m V V (H2O) t (O)2 sa ] 2 geometry unstable N/A 63 6 [(OPO) m V V (H2O) a (O)2 st ] 2 geometry unstable N/A 64 6 [(OPO) m HV V (OH) s (O)2 ta ] [(OPO) m HV V (OH) t (O)2 sa ] [(OPO) m HV V (OH) a (O)2 st ]
7 Table S2a. Comprehensive compilation of all the transition states for propane activation by (OPO)V V species, along with their gas-phase energies (Egas) and aqueous free energies (Gaq), both in hartrees; and their aqueous free energies relative to each other (ΔGrel), in kcal/mol. For some entries, geometry optimisation led to the formation of a lower-energy isomer. The energies for these entries are not reported. 4 + iprh 5 (OPO)V V O s (H2O) a + HiPr [(OPO)HiPrVO s (H2O) a ] [(OPO)HiPrVO a (H2O) s ] [(OPO)HiPrVO s (OH) a ] geometry unstable N/A 70 5 [(OPO)HiPrVO a (OH) s ] geometry unstable N/A 71 5 [(OPO)HiPrVO2 sa ] 2 geometry unstable N/A 72 6 [(OPO) f HiPrVO c (H2O)2 st ] [(OPO) f HiPrVO s (H2O)2 ct ] [(OPO) f HiPrVO t (H2O)2 cs ] [(OPO) f HiPrVO c (OH) s (H2O) t ] [(OPO) f HiPrVO c (OH) t (H2O) t ] [(OPO) f HiPrVO s (OH) c (H2O) t ] [(OPO) f HiPrVO s (OH) t (H2O) c ] [(OPO) f HiPrVO t (OH) c (H2O) s ] [(OPO) f HiPrVO t (OH) s (H2O) c ] [(OPO) f HiPrVO c (OH)2 st ] [(OPO) f HiPrVO s (OH)2 ct ] [(OPO) f HiPrVO t (OH)2 cs ] [(OPO) f HiPrV(H2O) c O2 st ] [(OPO) f HiPrV(H2O) s O2 ct ] [(OPO) f HiPrV(H2O) t O2 cs ] [(OPO) m HiPrVO s (H2O)2 ta ] [(OPO) m HiPrVO t (H2O)2 sa ] [(OPO) m HiPrVO a (H2O)2 st ] [(OPO) m HiPrVO s (OH) t (H2O) a ] [(OPO) m HiPrVO s (OH) a (H2O) t ] [(OPO) m HiPrVO t (OH) s (H2O) a ] [(OPO) m HiPrVO t (OH) a (H2O) s ] [(OPO) m HiPrVO a (OH) s (H2O) t ] [(OPO) m HiPrVO a (OH) t (H2O) s ] [(OPO) m HiPrVO s (OH)2 ta ] [(OPO) m HiPrVO t (OH)2 sa ]
8 Table S2b. Continuation from Table S2a [(OPO) m HiPrVO a (OH)2 st ] [(OPO) m HiPrV(H2O) s O2 ta ] [(OPO) m HiPrV(H2O) t O2 sa ] 2 geometry unstable N/A [(OPO) m HiPrV(H2O) a O2 st ] 2 geometry unstable N/A Table S3a. Compilation of all (OPO)V IV species studied, along with their gas-phase energies (Egas) and aqueous free energies (Gaq), both in hartrees; and their aqueous free energies relative to each other (ΔGrel), in kcal/mol (OPO)HV IV O s (H2O) a (OPO)HV IV O a (H2O) s [(OPO)V IV O s (H2O) a ] [(OPO)V IV O a (H2O) s ] (OPO) f V IV (OH) c (H2O)2 st (OPO) f V IV (OH) s (H2O)2 ct (OPO) f V IV (OH) t (H2O)2 cs (OPO) f HV IV (H2O) c (OH)2 st (OPO) f HV IV (H2O) s (OH)2 ct (OPO) f HV IV (H2O) t (OH)2 cs (OPO) f HV IV O c (H2O)2 st (OPO) f HV IV O s (H2O)2 ct (OPO) f HV IV O t (H2O)2 cs [(OPO) f V IV O c (H2O)2 st ] [(OPO) f V IV O s (H2O)2 ct ] [(OPO) f V IV O t (H2O)2 cs ] [(OPO) f HV IV O c (OH) s (H2O) t ] [(OPO) f HV IV O c (OH) t (H2O) s ] [(OPO) f HV IV O s (OH) c (H2O) t ] [(OPO) f HV IV O s (OH) t (H2O) c ] [(OPO) f HV IV O t (OH) c (H2O) s ] [(OPO) f HV IV O t (OH) s (H2O) c ] [(OPO) f V IV O c (OH) s (H2O) t ] [(OPO) f V IV O c (OH) t (H2O) s ] [(OPO) f V IV O s (OH) c (H2O) t ] [(OPO) f V IV O s (OH) t (H2O) c ]
9 Table S3b. Continuation from Table S3a [(OPO) f V IV O t (OH) c (H2O) s ] [(OPO) f V IV O t (OH) s (H2O) c ] [(OPO) f HV IV O c (OH)2 st ] [(OPO) f HV IV O s (OH)2 ct ] [(OPO) f HV IV O t (OH)2 cs ] (OPO) m HV IV O s (H2O)2 ta (OPO) m HV IV O t (H2O)2 sa (OPO) m HV IV O a (H2O)2 st [(OPO) m V IV O s (H2O)2 ta ] [(OPO) m V IV O t (H2O)2 sa ] [(OPO) m V IV O a (H2O)2 st ] Table S4a. Compilation of all (OPO)V III (OiPr) species studied, with only 5- and triplet 6- coordinate fac-(opo) complexes considered. Their gas-phase energies (Egas) and aqueous free energies (Gaq) are both in hartrees, and their aqueous free energies relative to each other (ΔGrel) in kcal/mol. For some entries, geometry optimisation led to the formation of a lower-energy isomer. The energies for these entries are not reported. E. C. Species Egas Gaq ΔGrel [(OPO)V III (HOiPr) s (H2O) a ] geometry unstable N/A [(OPO)V III (HOiPr) s (H2O) a ] [(OPO)V III (HOiPr) a (H2O) s ] geometry unstable N/A [(OPO)V III (HOiPr) a (H2O) s ] geometry unstable N/A [(OPO)HV III (HOiPr) s (OH) a ] [(OPO)HV III (HOiPr) s (OH) a ] [(OPO)HV III (HOiPr) a (OH) s ] geometry unstable N/A [(OPO)HV III (HOiPr) a (OH) s ] geometry unstable N/A [(OPO)HV III (OiPr) s (H2O) a ] geometry unstable N/A [(OPO)HV III (OiPr) s (H2O) a ] [(OPO)HV III (OiPr) a (H2O) s ] [(OPO)HV III (OiPr) a (H2O) s ] [(OPO)HV III (HOiPr) s (H2O) a ] + geometry unstable N/A [(OPO)HV III (HOiPr) s (H2O) a ] + geometry unstable N/A [(OPO)HV III (HOiPr) a (H2O) s ] + geometry unstable N/A [(OPO)HV III (HOiPr) a (H2O) s ] + geometry unstable N/A [(OPO)HV III (OiPr) s (OH) a ]
10 Table S4b. Continuation from Table S4a. E. C. Species Egas Gaq ΔGrel [(OPO)HV III (OiPr) s (OH) a ] [(OPO)HV III (OiPr) a (OH) s ] geometry unstable N/A [(OPO)HV III (OiPr) a (OH) s ] [(OPO)V III (HOiPr) s (OH) a ] [(OPO)V III (HOiPr) s (OH) a ] geometry unstable N/A [(OPO)V III (HOiPr) a (OH) s ] geometry unstable N/A [(OPO)V III (HOiPr) a (OH) s ] geometry unstable N/A [(OPO)V III (OiPr) s (H2O) a ] [(OPO)V III (OiPr) s (H2O) a ] geometry unstable N/A [(OPO)V III (OiPr) a (H2O) s ] [(OPO)V III (OiPr) a (H2O) s ] geometry unstable N/A [(OPO) f V III (HOiPr) c (H2O)2 st ] [(OPO) f V III (HOiPr) s (H2O)2 ct ] [(OPO) f V III (HOiPr) t (H2O)2 cs ] [(OPO) f HV III (HOiPr) c (OH) s (H2O) t ] [(OPO) f HV III (HOiPr) c (OH) t (H2O) s ] [(OPO) f HV III (HOiPr) s (OH) c (H2O) t ] [(OPO) f HV III (HOiPr) s (OH) t (H2O) c ] [(OPO) f HV III (HOiPr) t (OH) c (H2O) s ] [(OPO) f HV III (HOiPr) t (OH) s (H2O) c ] [(OPO) f HV III (OiPr) c (H2O)2 st ] [(OPO) f HV III (OiPr) s (H2O)2 ct ] [(OPO) f HV III (OiPr) t (H2O)2 cs ] [(OPO) f V III (HOiPr) c (OH) s (H2O) t ] [(OPO) f V III (HOiPr) c (OH) t (H2O) s ] [(OPO) f V III (HOiPr) s (OH) c (H2O) t ] [(OPO) f V III (HOiPr) s (OH) t (H2O) c ] [(OPO) f V III (HOiPr) t (OH) c (H2O) s ] [(OPO) f V III (HOiPr) t (OH) s (H2O) c ] [(OPO) f V III (OiPr) c (H2O)2 st ] [(OPO) f V III (OiPr) s (H2O)2 ct ] [(OPO) f V III (OiPr) t (H2O)2 cs ] [(OPO) f HV III (HOiPr) c (OH)2 st ] geometry unstable N/A [(OPO) f HV III (HOiPr) s (OH)2 ct ] [(OPO) f HV III (HOiPr) t (OH)2 cs ] [(OPO) f HV III (OiPr) c (OH) s (H2O) t ] [(OPO) f HV III (OiPr) c (OH) t (H2O) s ]
11 Table S4c. Continuation from Table S4b. E. C. Species Egas Gaq ΔGrel [(OPO) f HV III (OiPr) s (OH) c (H2O) t ] [(OPO) f HV III (OiPr) s (OH) t (H2O) c ] [(OPO) f HV III (OiPr) t (OH) c (H2O) s ] geometry unstable N/A [(OPO) f HV III (OiPr) t (OH) s (H2O) c ] Table S5a. Comprehensive compilation of all (OPO)V III species containing only hydroxo/aqua ligands studied, along with their gas-phase energies (Egas) and aqueous free energies (Gaq), both in hartrees, and their aqueous free energies relative to each other (ΔGrel) in kcal/mol. For some entries, geometry optimisation led to the formation of a lower-energy isomer. The energies for these entries are not reported. Entry Coord. Species Egas Gaq ΔGrel [(OPO)V III (H2O)2 sa ] [(OPO)V III (H2O)2 sa ] [(OPO)HV III (OH) s (H2O) a ] geometry unstable N/A [(OPO)HV III (OH) s (H2O) a ] [(OPO)HV III (OH) a (H2O) s ] [(OPO)HV III (OH) a (H2O) s ] [(OPO)V III (OH) s (H2O) a ] geometry unstable N/A [(OPO)V III (OH) s (H2O) a ] [(OPO)V III (OH) a (H2O) s ] geometry unstable N/A [(OPO)V III (OH) a (H2O) s ] [(OPO)HV III (OH)2 sa ] geometry unstable N/A [(OPO)HV III (OH)2 sa ] geometry unstable N/A [(OPO) f V III (H2O)3 cst ] [(OPO) f V III (H2O)3 cst ] [(OPO) f HV III (OH) c (H2O)2 st ] [(OPO) f HV III (OH) c (H2O)2 st ] [(OPO) f HV III (OH) s (H2O)2 ct ] [(OPO) f HV III (OH) s (H2O)2 ct ] [(OPO) f HV III (OH) t (H2O)2 cs ] [(OPO) f HV III (OH) t (H2O)2 cs ] [(OPO) f V III (OH) c (H2O)2 st ] [(OPO) f V III (OH) c (H2O)2 st ] [(OPO) f V III (OH) s (H2O)2 ct ] [(OPO) f V III (OH) s (H2O)2 ct ]
12 Table S5b. Continuation from Table S5a. Entry Coord. Species Egas Gaq ΔGrel [(OPO) f V III (OH) t (H2O)2 cs ] [(OPO) f V III (OH) t (H2O)2 cs ] [(OPO) f HV III (H2O) c (OH)2 st ] [(OPO) f HV III (H2O) c (OH)2 st ] [(OPO) f HV III (H2O) s (OH)2 ct ] [(OPO) f HV III (H2O) s (OH)2 ct ] [(OPO) f HV III (H2O) t (OH)2 cs ] [(OPO) f HV III (H2O) t (OH)2 cs ] [(OPO) m V III (H2O)3 sta ] [(OPO) m V III (H2O)3 sta ] [(OPO) m HV III (OH) s (H2O)2 ta ] [(OPO) m HV III (OH) s (H2O)2 ta ] [(OPO) m HV III (OH) t (H2O)2 sa ] [(OPO) m HV III (OH) t (H2O)2 sa ] [(OPO) m HV III (OH) a (H2O)2 st ] [(OPO) m HV III (OH) a (H2O)2 st ] [(OPO) m V III (OH) s (H2O)2 ta ] [(OPO) m V III (OH) s (H2O)2 ta ] [(OPO) m V III (OH) t (H2O)2 sa ] [(OPO) m V III (OH) t (H2O)2 sa ] [(OPO) m V III (OH) a (H2O)2 st ] [(OPO) m V III (OH) a (H2O)2 st ] [(OPO) m V III (H2O) s (OH)2 ta ] [(OPO) m HV III (H2O) s (OH)2 ta ] [(OPO) m HV III (H2O) t (OH)2 sa ] [(OPO) m HV III (H2O) t (OH)2 sa ] [(OPO) m HV III (H2O) a (OH)2 st ] [(OPO) m HV III (H2O) a (OH)2 st ] Table S6a. Comprehensive compilation of all (OPO)V V (OiPr)(O2) peroxide species studied, along with their gas-phase energies (Egas) and aqueous free energies (Gaq), both in hartrees, and their aqueous free energies relative to each other (ΔGrel) in kcal/mol. Note that a and b labels on O2 represent whether the O2 ligand is parallel or perpendicular to the PO moiety. For some entries, geometry optimisation led to the formation of a lower-energy isomer. The energies for these entries are not reported.
13 249 5 (OPO)V V (HOiPr) s (O2-a) a geometry unstable N/A (OPO)V V (HOiPr) s (O2-b) a geometry unstable N/A (OPO)V V (HOiPr) a (O2-a) s geometry unstable N/A (OPO)V V (HOiPr) a (O2-b) s geometry unstable N/A (OPO)HV V (OiPr) s (O2-a) a (OPO)HV V (OiPr) s (O2-b) a (OPO)HV V (OiPr) a (O2-a) s geometry unstable N/A (OPO)HV V (OiPr) a (O2-b) s geometry unstable N/A (OPO)V V (OiPr) s (OOH) a (OPO)V V (OiPr) a (OOH) s geometry unstable N/A [(OPO)HV V (HOiPr) s (O2-a) a ] + geometry unstable N/A [(OPO)HV V (HOiPr) s (O2-b) a ] + geometry unstable N/A [(OPO)HV V (HOiPr) a (O2-a) s ] + geometry unstable N/A [(OPO)HV V (HOiPr) a (O2-b) s ] + geometry unstable N/A [(OPO)V V (OiPr) s (O2-a) a ] [(OPO)V V (OiPr) s (O2-b) a ] [(OPO)V V (OiPr) a (O2-a) s ] [(OPO)V V (OiPr) a (O2-b) s ] (OPO) f V V (HOiPr) c (O2-a) s (H2O) t (OPO) f V V (HOiPr) c (O2-b) s (H2O) t (OPO) f V V (HOiPr) c (O2-a) t (H2O) s (OPO) f V V (HOiPr) c (O2-b) t (H2O) s (OPO) f V V (HOiPr) s (O2-a) c (H2O) t (OPO) f V V (HOiPr) s (O2-b) c (H2O) t (OPO) f V V (HOiPr) s (O2-a) t (H2O) c (OPO) f V V (HOiPr) s (O2-b) t (H2O) c (OPO) f V V (HOiPr) t (O2-a) c (H2O) s (OPO) f V V (HOiPr) t (O2-b) c (H2O) s (OPO) f V V (HOiPr) t (O2-a) s (H2O) c (OPO) f V V (HOiPr) t (O2-b) s (H2O) c (OPO) f HV V (HOiPr) c (O2-a) s (OH) t (OPO) f HV V (HOiPr) c (O2-b) s (OH) t (OPO) f HV V (HOiPr) c (O2-a) t (OH) s (OPO) f HV V (HOiPr) c (O2-b) t (OH) s (OPO) f HV V (HOiPr) s (O2-a) c (OH) t (OPO) f HV V (HOiPr) s (O2-b) c (OH) t (OPO) f HV V (HOiPr) s (O2-a) t (OH) c (OPO) f HV V (HOiPr) s (O2-b) t (OH) c
14 Table S6b. Continuation from Table S6a (OPO) f HV V (HOiPr) t (O2-a) c (OH) s (OPO) f HV V (HOiPr) t (O2-b) c (OH) s (OPO) f HV V (HOiPr) t (O2-a) s (OH) c (OPO) f HV V (HOiPr) t (O2-b) s (OH) c (OPO) f HV V (OiPr) c (O2-a) s (H2O) t (OPO) f HV V (OiPr) c (O2-b) s (H2O) t (OPO) f HV V (OiPr) c (O2-a) t (H2O) s (OPO) f HV V (OiPr) c (O2-b) t (H2O) s (OPO) f HV V (OiPr) s (O2-a) c (H2O) t (OPO) f HV V (OiPr) s (O2-b) c (H2O) t (OPO) f HV V (OiPr) s (O2-a) t (H2O) c (OPO) f HV V (OiPr) s (O2-b) t (H2O) c (OPO) f HV V (OiPr) t (O2-a) c (H2O) s (OPO) f HV V (OiPr) t (O2-b) c (H2O) s (OPO) f HV V (OiPr) t (O2-a) s (H2O) c (OPO) f HV V (OiPr) t (O2-b) s (H2O) c (OPO) m HV V (OiPr) s (O2-a) t (H2O) a (OPO) m HV V (OiPr) s (O2-b) t (H2O) a (OPO) m HV V (OiPr) s (O2-a) a (H2O) t (OPO) m HV V (OiPr) s (O2-b) a (H2O) t (OPO) m HV V (OiPr) t (O2-a) s (H2O) a (OPO) m HV V (OiPr) t (O2-b) s (H2O) a (OPO) m HV V (OiPr) t (O2-a) a (H2O) s (OPO) m HV V (OiPr) t (O2-b) a (H2O) s (OPO) m HV V (OiPr) a (O2-a) s (H2O) t (OPO) m HV V (OiPr) a (O2-b) s (H2O) t (OPO) m HV V (OiPr) a (O2-a) t (H2O) s (OPO) m HV V (OiPr) a (O2-b) t (H2O) s (OPO) m V V (HOiPr) s (O2-a) t (H2O) a (OPO) m V V (HOiPr) s (O2-b) t (H2O) a (OPO) m V V (HOiPr) s (O2-a) a (H2O) t (OPO) m V V (HOiPr) s (O2-b) a (H2O) t (OPO) m V V (HOiPr) t (O2-a) s (H2O) a (OPO) m V V (HOiPr) t (O2-b) s (H2O) a geometry unstable N/A (OPO) m V V (HOiPr) t (O2-a) a (H2O) s (OPO) m V V (HOiPr) t (O2-b) a (H2O) s (OPO) m V V (HOiPr) a (O2-a) s (H2O) t
15 Table S6c. Continuation from Table S6b (OPO) m V V (HOiPr) a (O2-b) s (H2O) t (OPO) m V V (HOiPr) a (O2-a) t (H2O) s (OPO) m V V (HOiPr) a (O2-b) t (H2O) s [(OPO) m V V (OiPr) s (O2-a) t (H2O) a ] [(OPO) m V V (OiPr) s (O2-b) t (H2O) a ] [(OPO) m V V (OiPr) s (O2-a) a (H2O) t ] [(OPO) m V V (OiPr) s (O2-b) a (H2O) t ] [(OPO) m V V (OiPr) t (O2-a) s (H2O) a ] [(OPO) m V V (OiPr) t (O2-b) s (H2O) a ] [(OPO) m V V (OiPr) t (O2-a) a (H2O) s ] [(OPO) m V V (OiPr) t (O2-b) a (H2O) s ] [(OPO) m V V (OiPr) a (O2-a) s (H2O) t ] [(OPO) m V V (OiPr) a (O2-b) s (H2O) t ] [(OPO) m V V (OiPr) a (O2-a) t (H2O) s ] [(OPO) m V V (OiPr) a (O2-b) t (H2O) s ] (OPO) f V V (HOiPr) c (HOOH) s O t (OPO) f V V (HOiPr) c (HOOH) t O s geometry unstable N/A (OPO) f V V (HOiPr) s (HOOH) c O t geometry unstable N/A (OPO) f V V (HOiPr) s (HOOH) t O c geometry unstable N/A (OPO) f V V (HOiPr) t (HOOH) c O s geometry unstable N/A (OPO) f V V (HOiPr) t (HOOH) s O c geometry unstable N/A (OPO) f V V (HOiPr) c (OOH) s (OH) t (OPO) f V V (HOiPr) c (OOH) t (OH) s (OPO) f V V (HOiPr) s (OOH) c (OH) t geometry unstable N/A (OPO) f V V (HOiPr) s (OOH) t (OH) c (OPO) f V V (HOiPr) t (OOH) c (OH) s (OPO) f V V (HOiPr) t (OOH) s (OH) c (OPO) f V V (OiPr) c (HOOH) s (OH) t (OPO) f V V (OiPr) c (HOOH) t (OH) s (OPO) f V V (OiPr) s (HOOH) c (OH) t geometry unstable N/A (OPO) f V V (OiPr) s (HOOH) t (OH) c (OPO) f V V (OiPr) t (HOOH) c (OH) s geometry unstable N/A (OPO) f V V (OiPr) t (HOOH) s (OH) c (OPO) f V V (OiPr) c (OOH) s (H2O) t (OPO) f V V (OiPr) c (OOH) t (H2O) s (OPO) f V V (OiPr) s (OOH) c (H2O) t (OPO) f V V (OiPr) s (OOH) t (H2O) c
16 Table S6d. Continuation from Table S6c (OPO) f V V (OiPr) t (OOH) c (H2O) s (OPO) f V V (OiPr) t (OOH) s (H2O) c (OPO) f HV V (HOiPr) c (OOH) s O t (OPO) f HV V (HOiPr) c (OOH) t O s (OPO) f HV V (HOiPr) s (OOH) c O t (OPO) f HV V (HOiPr) s (OOH) t O c (OPO) f HV V (HOiPr) t (OOH) c O s (OPO) f HV V (HOiPr) t (OOH) s O c (OPO) f HV V (OiPr) c (HOOH) s O t (OPO) f HV V (OiPr) c (HOOH) t O s (OPO) f HV V (OiPr) s (HOOH) c O t (OPO) f HV V (OiPr) s (HOOH) t O c (OPO) f HV V (OiPr) t (HOOH) c O s (OPO) f HV V (OiPr) t (HOOH) s O c (OPO) f HV V (OiPr) c (OOH) s (OH) t geometry unstable N/A (OPO) f HV V (OiPr) c (OOH) t (OH) s (OPO) f HV V (OiPr) s (OOH) c (OH) t (OPO) f HV V (OiPr) s (OOH) t (OH) c (OPO) f HV V (OiPr) t (OOH) c (OH) s (OPO) f HV V (OiPr) t (OOH) s (OH) c Table S7a. Compilation of all (OPO)V V (O2) peroxide species containing only oxo/hydroxo/aqua ligands studied, along with their gas-phase energies (Egas) and aqueous free energies (Gaq), both in hartrees, and their aqueous free energies relative to each other (ΔGrel) in kcal/mol. Note that a and b labels on O2 represent whether the O2 ligand is parallel or perpendicular to the PO moiety. For some entries, geometry optimisation led to the formation of a lower-energy isomer. The energies for these entries are not reported (OPO)V V (O2-a) s (H2O) a (OPO)V V (O2-b) s (H2O) a (OPO)V V (O2-a) a (H2O) s geometry unstable N/A (OPO)V V (O2-b) a (H2O) s geometry unstable N/A (OPO)HV V (O2-a) s (OH) a (OPO)HV V (O2-b) s (OH) a geometry unstable N/A (OPO)HV V (O2-a) a (OH) s geometry unstable N/A
17 Table S7b. Continuation from Table S7a. Entry C. Species Egas Gaq (OPO)HV V (O2-b) a (OH) s geometry unstable N/A (OPO)V V O s (η 1 -HOOH) a (OPO)V V O s (η 2 -HOOH) a (OPO)V V O a (η 1 -HOOH) s geometry unstable N/A (OPO)V V O a (η 2 -HOOH) s (OPO)V V (OH) s (η 1 -OOH) a geometry unstable N/A (OPO)V V (OH) s (η 2 -OOH) a geometry unstable N/A (OPO)V V (OH) a (η 1 -OOH) s (OPO)V V (OH) a (η 2 -OOH) s (OPO)HV V O s (η 1 -OOH) a geometry unstable N/A (OPO)HV V O s (η 2 -OOH) a (OPO)HV V O a (η 1 -OOH) s (OPO)HV V O a (η 2 -OOH) s [(OPO)HV V (O2-a) s (H2O) a ] N/A [(OPO)HV V (O2-b) s (H2O) a ] + geometry unstable N/A [(OPO)HV V (O2-a) a (H2O) s ] + geometry unstable N/A [(OPO)HV V (O2-b) a (H2O) s ] + geometry unstable N/A [(OPO)V V (O2-a) s (OH) a ] [(OPO)V V (O2-b) s (OH) a ] N/A [(OPO)V V (O2-a) a (OH) s ] [(OPO)V V (O2-b) a (OH) s ] geometry unstable N/A (OPO) f V V (O2-a) c (H2O)2 st (OPO) f V V (O2-b) c (H2O)2 st (OPO) f V V (O2-a) s (H2O)2 ct (OPO) f V V (O2-b) s (H2O)2 ct (OPO) f V V (O2-a) t (H2O)2 cs (OPO) f V V (O2-b) t (H2O)2 cs (OPO) f HV V (O2-a) c (OH) s (H2O) t (OPO) f HV V (O2-b) c (OH) s (H2O) t (OPO) f HV V (O2-a) c (OH) t (H2O) s (OPO) f HV V (O2-b) c (OH) t (H2O) s (OPO) f HV V (O2-a) s (OH) c (H2O) t (OPO) f HV V (O2-b) s (OH) c (H2O) t (OPO) f HV V (O2-a) s (OH) t (H2O) c (OPO) f HV V (O2-b) s (OH) t (H2O) c (OPO) f HV V (O2-a) t (OH) c (H2O) s (OPO) f HV V (O2-b) t (OH) c (H2O) s
18 Table S7c. Continuation from Table S7b (OPO) f HV V (O2-a) t (OH) s (H2O) c (OPO) f HV V (O2-b) t (OH) s (H2O) c (OPO) f V V O c (HOOH) s (H2O) t (OPO) f V V O c (HOOH) t (H2O) s (OPO) f V V O s (HOOH) c (H2O) t (OPO) f V V O s (HOOH) t (H2O) c (OPO) f V V O t (HOOH) c (H2O) s (OPO) f V V O t (HOOH) s (H2O) c (OPO) f HV V O c (OOH) s (H2O) t (OPO) f HV V O c (OOH) t (H2O) s (OPO) f HV V O s (OOH) c (H2O) t (OPO) f HV V O s (OOH) t (H2O) c (OPO) f HV V O t (OOH) c (H2O) s (OPO) f HV V O t (OOH) s (H2O) c (OPO) f HV V O c (HOOH) s (OH) t (OPO) f HV V O c (HOOH) t (OH) s (OPO) f HV V O s (HOOH) c (OH) t (OPO) f HV V O s (HOOH) t (OH) c (OPO) f HV V O t (HOOH) c (OH) s (OPO) f HV V O t (HOOH) s (OH) c (OPO) f V V (HOOH) c (OH)2 st geometry unstable N/A (OPO) f V V (HOOH) s (OH)2 ct geometry unstable N/A (OPO) f V V (HOOH) t (OH)2 cs (OPO) f V V (OOH) c (OH) s (H2O) t geometry unstable N/A (OPO) f V V (OOH) c (OH) t (H2O) c (OPO) f V V (OOH) s (OH) c (H2O) t geometry unstable N/A (OPO) f V V (OOH) s (OH) t (H2O) c (OPO) f V V (OOH) t (OH) s (H2O) c (OPO) f V V (OOH) t (OH) c (H2O) s (OPO) f HV V (OOH) c (OH)2 st (OPO) f HV V (OOH) s (OH)2 ct (OPO) f HV V (OOH) t (OH)2 cs
19 Table S8. Data for the two dimeric species studied, consisting of their gas-phase energies (Egas) and aqueous free energies (Gaq), both in hartrees, and their aqueous free energies relative to each other (ΔGrel) in kcal/mol , 6 1 {[(OPO) f V(H2O)2 st O c ] [O s (H2O)2 ct V(OPO) f ]} , 6 3 {[(OPO) f V IV (H2O)2 st O c ] [O s (H2O)2 ct V IV (OPO) f ]} Table S9. Compilation of all (OPO)V V (OiPr) species studied, along with their gas-phase energies (Egas) and aqueous free energies (Gaq), both in hartrees, and their aqueous free energies relative to each other (ΔGrel) in kcal/mol. Only species containing a V=O moiety were considered. For some entries, geometry optimisation led to the formation of a lower-energy isomer. The energies for these entries are not reported. Entry Coord. Species Egas Gaq ΔGrel (OPO)V V O s (HOiPr) a (OPO)V V O a (HOiPr) s geometry unstable N/A (OPO)HV V O s (OiPr) a geometry unstable N/A (OPO)HV V O a (OiPr) s (OPO) f V V O c (HOiPr) s (H2O) t (OPO) f V V O c (HOiPr) t (H2O) s (OPO) f V V O s (HOiPr) c (H2O) t (OPO) f V V O s (HOiPr) t (H2O) c (OPO) f V V O t (HOiPr) c (H2O) s (OPO) f V V O t (HOiPr) s (H2O) c (OPO) f HV V O c (HOiPr) s (OH) t (OPO) f HV V O c (HOiPr) t (OH) s (OPO) f HV V O s (HOiPr) c (OH) t geometry unstable N/A (OPO) f HV V O s (HOiPr) t (OH) c (OPO) f HV V O t (HOiPr) c (OH) s (OPO) f HV V O t (HOiPr) s (OH) c (OPO) f HV V O c (OiPr) s (H2O) t (OPO) f HV V O c (OiPr) t (H2O) s (OPO) f HV V O s (OiPr) c (H2O) t (OPO) f HV V O s (OiPr) t (H2O) c (OPO) f HV V O t (OiPr) c (H2O) s (OPO) f HV V O t (OiPr) s (H2O) c
20 Table S10. Compilation of all (OPO)V III (O2)(OiPr) peroxide species containing isopropoxy ligands studied, along with their gas-phase energies (Egas) and aqueous free energies (Gaq), both in hartrees, and their aqueous free energies relative to each other (ΔGrel) in kcal/mol. Note that only neutral triplet 5-coordinate and 6-coordinate fac-(opo) complexes were studied. For some entries, geometry optimisation led to the formation of a lower-energy isomer. The energies for these entries are not reported [(OPO)V III (HOiPr) s (H2O2) a ] geometry unstable N/A [(OPO)V III (HOiPr) a (H2O2) s ] [(OPO)HV III (OiPr) s (H2O2) a ] [(OPO)HV III (OiPr) a (H2O2) s ] [(OPO)HV III (HOiPr) s (η 1 -OOH) a ] geometry unstable N/A [(OPO)HV III (HOiPr) a (η 1 -OOH) s ] [(OPO)HV III (HOiPr) s (η 2 -OOH) a ] [(OPO)HV III (HOiPr) a (η 2 -OOH) s ] [(OPO) f V III (HOiPr) c (HOOH) s (H2O) t ] [(OPO) f V III (HOiPr) c (HOOH) t (H2O) s ] [(OPO) f V III (HOiPr) s (HOOH) c (H2O) t ] [(OPO) f V III (HOiPr) s (HOOH) t (H2O) c ] [(OPO) f V III (HOiPr) t (HOOH) c (H2O) s ] [(OPO) f V III (HOiPr) t (HOOH) s (H2O) c ] [(OPO) f HV III (HOiPr) c (HOOH) s (OH) t ] geometry unstable N/A [(OPO) f HV III (HOiPr) c (HOOH) t (OH) s ] [(OPO) f HV III (HOiPr) s (HOOH) c (OH) t ] [(OPO) f HV III (HOiPr) s (HOOH) t (OH) c ] [(OPO) f HV III (HOiPr) t (HOOH) c (OH) s ] [(OPO) f HV III (HOiPr) t (HOOH) s (OH) c ] [(OPO) f HV III (HOiPr) c (OOH) s (H2O) t ] [(OPO) f HV III (HOiPr) c (OOH) t (H2O) s ] [(OPO) f HV III (HOiPr) s (OOH) c (H2O) t ] [(OPO) f HV III (HOiPr) s (OOH) t (H2O) c ] [(OPO) f HV III (HOiPr) t (OOH) c (H2O) s ] [(OPO) f HV III (HOiPr) t (OOH) s (H2O) c ] [(OPO) f HV III (OiPr) c (HOOH) s (H2O) t ] [(OPO) f HV III (OiPr) c (HOOH) t (H2O) s ] [(OPO) f HV III (OiPr) s (HOOH) c (H2O) t ] [(OPO) f HV III (OiPr) s (HOOH) t (H2O) c ] [(OPO) f HV III (OiPr) t (HOOH) c (H2O) s ] [(OPO) f HV III (OiPr) t (HOOH) s (H2O) c ]
21 Table S11. Compilation of [(OPO)HV(HOiPr)(O OH)] transition state species, which represent the transition from (OPO)HV III (HOiPr)(OOH) ( ) to (OPO) f HV V O(OH)(HOiPr) ( ). Given are their gas-phase energies (Egas) and aqueous free energies (Gaq), both in hartrees, and their aqueous free energies relative to 3 [(OPO) f HV III (HOiPr) s (OOH) c (H2O) t ] (503) (ΔGrel) in kcal/mol. Note that only neutral transition states between 5-coordinate and 6- coordinate fac-(opo) complexes were studied. Also note that all transition states found involve OOH bound in an η 2 -fashion splitting into oxo and hydroxo ligands. For some entries, the transition state was not found, and energies for those entries are not reported. E. Coord. Species Egas Gaq ΔGrel [(OPO)HV(HOiPr) s (O OH) a ] not found N/A [(OPO)HV(HOiPr) s (O OH) a ] [(OPO)HV(HOiPr) a (O OH) s ] [(OPO)HV(HOiPr) a (O OH) s ] Table S12a. Compilation of all (OPO)V III (O2) peroxide species containing only oxo/hydroxo/aqua ligands studied, along with their gas-phase energies (Egas) and aqueous free energies (Gaq), both in hartrees, and their aqueous free energies relative to each other (ΔGrel) in kcal/mol. Note that only neutral triplet 5-coordinate and 6-coordinate fac-(opo) complexes were studied. Also note that HOOH/OOH binds exclusively in an η 1 -fashion in the fac-(opo) complexes. For some entries, geometry optimisation led to the formation of a lower-energy isomer. The energies for these entries are not reported [(OPO)V III (η 1 -HOOH) s (H2O) a ] [(OPO)V III (η 1 -HOOH) a (H2O) s ] [(OPO)V III (η 2 -HOOH) s (H2O) a ] [(OPO)V III (η 2 -HOOH) a (H2O) s ] geometry unstable N/A [(OPO)HV III (η 1 -HOOH) s (OH) a ] [(OPO)HV III (η 1 -HOOH) a (OH) s ] [(OPO)HV III (η 2 -HOOH) s (OH) a ] geometry unstable N/A [(OPO)HV III (η 2 -HOOH) a (OH) s ] geometry unstable N/A [(OPO)HV III (η 1 -OOH) s (H2O) a ] [(OPO)HV III (η 1 -OOH) a (H2O) s ] [(OPO)HV III (η 2 -OOH) s (H2O) a ] [(OPO)HV III (η 2 -OOH) a (H2O) s ] [(OPO) f V III (HOOH) c (H2O)2 st ] [(OPO) f V III (HOOH) s (H2O)2 ct ] [(OPO) f V III (HOOH) t (H2O)2 cs ]
22 Table S12b. Continuation from Table S12a [(OPO) f HV III (HOOH) c (OH) s (H2O) t ] [(OPO) f HV III (HOOH) c (OH) t (H2O) s ] [(OPO) f HV III (HOOH) s (OH) c (H2O) t ] [(OPO) f HV III (HOOH) s (OH) t (H2O) c ] [(OPO) f HV III (HOOH) t (OH) c (H2O) s ] [(OPO) f HV III (HOOH) t (OH) s (H2O) c ] [(OPO) f HV III (OOH) c (H2O)2 st ] [(OPO) f HV III (OOH) s (H2O)2 ct ] [(OPO) f HV III (OOH) t (H2O)2 cs ] Table S13. Compilation of [(OPO)HV(O OH)(H2O)] transition state species, which represent the transition from (OPO)HV III (OOH)(H2O) ( ) to (OPO) f HV V O(OH)(H2O) (16-21). Given are their gas-phase energies (Egas) and aqueous free energies (Gaq), both in hartrees, and their aqueous free energies relative to 3 [(OPO) f HV III (OOH) c (H2O)2 st ] (538) (ΔGrel) in kcal/mol. Note that only neutral transition states between 5-coordinate and 6-coordinate fac-(opo) complexes were studied. Also note that all transition states found involve OOH bound in an η 2 -fashion splitting into oxo and hydroxo ligands. For some entries, the transition state was not found, and energies for those entries are not reported. Entry Coord. Species Egas Gaq ΔGrel [(OPO)HV(O OH) s (H2O) a ] [(OPO)HV(O OH) s (H2O) a ] [(OPO)HV(O OH) a (H2O) s ] not found N/A [(OPO)HV(O OH) a (H2O) s ]
Mean bond enthalpy Standard enthalpy of formation Bond N H N N N N H O O O
 Q1. (a) Explain the meaning of the terms mean bond enthalpy and standard enthalpy of formation. Mean bond enthalpy... Standard enthalpy of formation... (5) (b) Some mean bond enthalpies are given below.
Q1. (a) Explain the meaning of the terms mean bond enthalpy and standard enthalpy of formation. Mean bond enthalpy... Standard enthalpy of formation... (5) (b) Some mean bond enthalpies are given below.
ΚΥΠΡΙΑΚΗ ΕΤΑΙΡΕΙΑ ΠΛΗΡΟΦΟΡΙΚΗΣ CYPRUS COMPUTER SOCIETY ΠΑΓΚΥΠΡΙΟΣ ΜΑΘΗΤΙΚΟΣ ΔΙΑΓΩΝΙΣΜΟΣ ΠΛΗΡΟΦΟΡΙΚΗΣ 19/5/2007
 Οδηγίες: Να απαντηθούν όλες οι ερωτήσεις. Αν κάπου κάνετε κάποιες υποθέσεις να αναφερθούν στη σχετική ερώτηση. Όλα τα αρχεία που αναφέρονται στα προβλήματα βρίσκονται στον ίδιο φάκελο με το εκτελέσιμο
Οδηγίες: Να απαντηθούν όλες οι ερωτήσεις. Αν κάπου κάνετε κάποιες υποθέσεις να αναφερθούν στη σχετική ερώτηση. Όλα τα αρχεία που αναφέρονται στα προβλήματα βρίσκονται στον ίδιο φάκελο με το εκτελέσιμο
HOMEWORK 4 = G. In order to plot the stress versus the stretch we define a normalized stretch:
 HOMEWORK 4 Problem a For the fast loading case, we want to derive the relationship between P zz and λ z. We know that the nominal stress is expressed as: P zz = ψ λ z where λ z = λ λ z. Therefore, applying
HOMEWORK 4 Problem a For the fast loading case, we want to derive the relationship between P zz and λ z. We know that the nominal stress is expressed as: P zz = ψ λ z where λ z = λ λ z. Therefore, applying
derivation of the Laplacian from rectangular to spherical coordinates
 derivation of the Laplacian from rectangular to spherical coordinates swapnizzle 03-03- :5:43 We begin by recognizing the familiar conversion from rectangular to spherical coordinates (note that φ is used
derivation of the Laplacian from rectangular to spherical coordinates swapnizzle 03-03- :5:43 We begin by recognizing the familiar conversion from rectangular to spherical coordinates (note that φ is used
Phys460.nb Solution for the t-dependent Schrodinger s equation How did we find the solution? (not required)
 Phys460.nb 81 ψ n (t) is still the (same) eigenstate of H But for tdependent H. The answer is NO. 5.5.5. Solution for the tdependent Schrodinger s equation If we assume that at time t 0, the electron starts
Phys460.nb 81 ψ n (t) is still the (same) eigenstate of H But for tdependent H. The answer is NO. 5.5.5. Solution for the tdependent Schrodinger s equation If we assume that at time t 0, the electron starts
EE512: Error Control Coding
 EE512: Error Control Coding Solution for Assignment on Finite Fields February 16, 2007 1. (a) Addition and Multiplication tables for GF (5) and GF (7) are shown in Tables 1 and 2. + 0 1 2 3 4 0 0 1 2 3
EE512: Error Control Coding Solution for Assignment on Finite Fields February 16, 2007 1. (a) Addition and Multiplication tables for GF (5) and GF (7) are shown in Tables 1 and 2. + 0 1 2 3 4 0 0 1 2 3
2 Composition. Invertible Mappings
 Arkansas Tech University MATH 4033: Elementary Modern Algebra Dr. Marcel B. Finan Composition. Invertible Mappings In this section we discuss two procedures for creating new mappings from old ones, namely,
Arkansas Tech University MATH 4033: Elementary Modern Algebra Dr. Marcel B. Finan Composition. Invertible Mappings In this section we discuss two procedures for creating new mappings from old ones, namely,
Electronic Supplementary Information DFT Characterization on the Mechanism of Water Splitting Catalyzed by Single-Ru-substituted Polyoxometalates
 Electronic Supplementary Information DFT Characterization on the Mechanism of Water Splitting Catalyzed by Single-Ru-substituted Polyoxometalates Zhong-Ling Lang, Guo-Chun Yang, Na-Na Ma, Shi-Zheng Wen,
Electronic Supplementary Information DFT Characterization on the Mechanism of Water Splitting Catalyzed by Single-Ru-substituted Polyoxometalates Zhong-Ling Lang, Guo-Chun Yang, Na-Na Ma, Shi-Zheng Wen,
C.S. 430 Assignment 6, Sample Solutions
 C.S. 430 Assignment 6, Sample Solutions Paul Liu November 15, 2007 Note that these are sample solutions only; in many cases there were many acceptable answers. 1 Reynolds Problem 10.1 1.1 Normal-order
C.S. 430 Assignment 6, Sample Solutions Paul Liu November 15, 2007 Note that these are sample solutions only; in many cases there were many acceptable answers. 1 Reynolds Problem 10.1 1.1 Normal-order
DESIGN OF MACHINERY SOLUTION MANUAL h in h 4 0.
 DESIGN OF MACHINERY SOLUTION MANUAL -7-1! PROBLEM -7 Statement: Design a double-dwell cam to move a follower from to 25 6, dwell for 12, fall 25 and dwell for the remader The total cycle must take 4 sec
DESIGN OF MACHINERY SOLUTION MANUAL -7-1! PROBLEM -7 Statement: Design a double-dwell cam to move a follower from to 25 6, dwell for 12, fall 25 and dwell for the remader The total cycle must take 4 sec
Supporting Information
 Supporting Information rigin of the Regio- and Stereoselectivity of Allylic Substitution of rganocopper Reagents Naohiko Yoshikai, Song-Lin Zhang, and Eiichi Nakamura* Department of Chemistry, The University
Supporting Information rigin of the Regio- and Stereoselectivity of Allylic Substitution of rganocopper Reagents Naohiko Yoshikai, Song-Lin Zhang, and Eiichi Nakamura* Department of Chemistry, The University
CHAPTER 25 SOLVING EQUATIONS BY ITERATIVE METHODS
 CHAPTER 5 SOLVING EQUATIONS BY ITERATIVE METHODS EXERCISE 104 Page 8 1. Find the positive root of the equation x + 3x 5 = 0, correct to 3 significant figures, using the method of bisection. Let f(x) =
CHAPTER 5 SOLVING EQUATIONS BY ITERATIVE METHODS EXERCISE 104 Page 8 1. Find the positive root of the equation x + 3x 5 = 0, correct to 3 significant figures, using the method of bisection. Let f(x) =
Homework 3 Solutions
 Homework 3 Solutions Igor Yanovsky (Math 151A TA) Problem 1: Compute the absolute error and relative error in approximations of p by p. (Use calculator!) a) p π, p 22/7; b) p π, p 3.141. Solution: For
Homework 3 Solutions Igor Yanovsky (Math 151A TA) Problem 1: Compute the absolute error and relative error in approximations of p by p. (Use calculator!) a) p π, p 22/7; b) p π, p 3.141. Solution: For
Section 9.2 Polar Equations and Graphs
 180 Section 9. Polar Equations and Graphs In this section, we will be graphing polar equations on a polar grid. In the first few examples, we will write the polar equation in rectangular form to help identify
180 Section 9. Polar Equations and Graphs In this section, we will be graphing polar equations on a polar grid. In the first few examples, we will write the polar equation in rectangular form to help identify
3.4 SUM AND DIFFERENCE FORMULAS. NOTE: cos(α+β) cos α + cos β cos(α-β) cos α -cos β
 3.4 SUM AND DIFFERENCE FORMULAS Page Theorem cos(αβ cos α cos β -sin α cos(α-β cos α cos β sin α NOTE: cos(αβ cos α cos β cos(α-β cos α -cos β Proof of cos(α-β cos α cos β sin α Let s use a unit circle
3.4 SUM AND DIFFERENCE FORMULAS Page Theorem cos(αβ cos α cos β -sin α cos(α-β cos α cos β sin α NOTE: cos(αβ cos α cos β cos(α-β cos α -cos β Proof of cos(α-β cos α cos β sin α Let s use a unit circle
Supplementary Materials for. Kinetic and Computational Studies on Pd(I) Dimer- Mediated Halogen Exchange of Aryl Iodides
 Supplementary Materials for Kinetic and Computational Studies on Pd(I) Dimer- Mediated Halogen Exchange of Aryl Iodides Indrek Kalvet, a Karl J. Bonney, a and Franziska Schoenebeck a * a Institute of Organic
Supplementary Materials for Kinetic and Computational Studies on Pd(I) Dimer- Mediated Halogen Exchange of Aryl Iodides Indrek Kalvet, a Karl J. Bonney, a and Franziska Schoenebeck a * a Institute of Organic
The challenges of non-stable predicates
 The challenges of non-stable predicates Consider a non-stable predicate Φ encoding, say, a safety property. We want to determine whether Φ holds for our program. The challenges of non-stable predicates
The challenges of non-stable predicates Consider a non-stable predicate Φ encoding, say, a safety property. We want to determine whether Φ holds for our program. The challenges of non-stable predicates
Section 8.3 Trigonometric Equations
 99 Section 8. Trigonometric Equations Objective 1: Solve Equations Involving One Trigonometric Function. In this section and the next, we will exple how to solving equations involving trigonometric functions.
99 Section 8. Trigonometric Equations Objective 1: Solve Equations Involving One Trigonometric Function. In this section and the next, we will exple how to solving equations involving trigonometric functions.
Instruction Execution Times
 1 C Execution Times InThisAppendix... Introduction DL330 Execution Times DL330P Execution Times DL340 Execution Times C-2 Execution Times Introduction Data Registers This appendix contains several tables
1 C Execution Times InThisAppendix... Introduction DL330 Execution Times DL330P Execution Times DL340 Execution Times C-2 Execution Times Introduction Data Registers This appendix contains several tables
Strain gauge and rosettes
 Strain gauge and rosettes Introduction A strain gauge is a device which is used to measure strain (deformation) on an object subjected to forces. Strain can be measured using various types of devices classified
Strain gauge and rosettes Introduction A strain gauge is a device which is used to measure strain (deformation) on an object subjected to forces. Strain can be measured using various types of devices classified
Second Order RLC Filters
 ECEN 60 Circuits/Electronics Spring 007-0-07 P. Mathys Second Order RLC Filters RLC Lowpass Filter A passive RLC lowpass filter (LPF) circuit is shown in the following schematic. R L C v O (t) Using phasor
ECEN 60 Circuits/Electronics Spring 007-0-07 P. Mathys Second Order RLC Filters RLC Lowpass Filter A passive RLC lowpass filter (LPF) circuit is shown in the following schematic. R L C v O (t) Using phasor
On a four-dimensional hyperbolic manifold with finite volume
 BULETINUL ACADEMIEI DE ŞTIINŢE A REPUBLICII MOLDOVA. MATEMATICA Numbers 2(72) 3(73), 2013, Pages 80 89 ISSN 1024 7696 On a four-dimensional hyperbolic manifold with finite volume I.S.Gutsul Abstract. In
BULETINUL ACADEMIEI DE ŞTIINŢE A REPUBLICII MOLDOVA. MATEMATICA Numbers 2(72) 3(73), 2013, Pages 80 89 ISSN 1024 7696 On a four-dimensional hyperbolic manifold with finite volume I.S.Gutsul Abstract. In
Reminders: linear functions
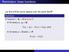 Reminders: linear functions Let U and V be vector spaces over the same field F. Definition A function f : U V is linear if for every u 1, u 2 U, f (u 1 + u 2 ) = f (u 1 ) + f (u 2 ), and for every u U
Reminders: linear functions Let U and V be vector spaces over the same field F. Definition A function f : U V is linear if for every u 1, u 2 U, f (u 1 + u 2 ) = f (u 1 ) + f (u 2 ), and for every u U
Math 6 SL Probability Distributions Practice Test Mark Scheme
 Math 6 SL Probability Distributions Practice Test Mark Scheme. (a) Note: Award A for vertical line to right of mean, A for shading to right of their vertical line. AA N (b) evidence of recognizing symmetry
Math 6 SL Probability Distributions Practice Test Mark Scheme. (a) Note: Award A for vertical line to right of mean, A for shading to right of their vertical line. AA N (b) evidence of recognizing symmetry
ΚΥΠΡΙΑΚΗ ΕΤΑΙΡΕΙΑ ΠΛΗΡΟΦΟΡΙΚΗΣ CYPRUS COMPUTER SOCIETY ΠΑΓΚΥΠΡΙΟΣ ΜΑΘΗΤΙΚΟΣ ΔΙΑΓΩΝΙΣΜΟΣ ΠΛΗΡΟΦΟΡΙΚΗΣ 24/3/2007
 Οδηγίες: Να απαντηθούν όλες οι ερωτήσεις. Όλοι οι αριθμοί που αναφέρονται σε όλα τα ερωτήματα μικρότεροι του 10000 εκτός αν ορίζεται διαφορετικά στη διατύπωση του προβλήματος. Αν κάπου κάνετε κάποιες υποθέσεις
Οδηγίες: Να απαντηθούν όλες οι ερωτήσεις. Όλοι οι αριθμοί που αναφέρονται σε όλα τα ερωτήματα μικρότεροι του 10000 εκτός αν ορίζεται διαφορετικά στη διατύπωση του προβλήματος. Αν κάπου κάνετε κάποιες υποθέσεις
SCHOOL OF MATHEMATICAL SCIENCES G11LMA Linear Mathematics Examination Solutions
 SCHOOL OF MATHEMATICAL SCIENCES GLMA Linear Mathematics 00- Examination Solutions. (a) i. ( + 5i)( i) = (6 + 5) + (5 )i = + i. Real part is, imaginary part is. (b) ii. + 5i i ( + 5i)( + i) = ( i)( + i)
SCHOOL OF MATHEMATICAL SCIENCES GLMA Linear Mathematics 00- Examination Solutions. (a) i. ( + 5i)( i) = (6 + 5) + (5 )i = + i. Real part is, imaginary part is. (b) ii. + 5i i ( + 5i)( + i) = ( i)( + i)
Lecture 2: Dirac notation and a review of linear algebra Read Sakurai chapter 1, Baym chatper 3
 Lecture 2: Dirac notation and a review of linear algebra Read Sakurai chapter 1, Baym chatper 3 1 State vector space and the dual space Space of wavefunctions The space of wavefunctions is the set of all
Lecture 2: Dirac notation and a review of linear algebra Read Sakurai chapter 1, Baym chatper 3 1 State vector space and the dual space Space of wavefunctions The space of wavefunctions is the set of all
6.1. Dirac Equation. Hamiltonian. Dirac Eq.
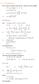 6.1. Dirac Equation Ref: M.Kaku, Quantum Field Theory, Oxford Univ Press (1993) η μν = η μν = diag(1, -1, -1, -1) p 0 = p 0 p = p i = -p i p μ p μ = p 0 p 0 + p i p i = E c 2 - p 2 = (m c) 2 H = c p 2
6.1. Dirac Equation Ref: M.Kaku, Quantum Field Theory, Oxford Univ Press (1993) η μν = η μν = diag(1, -1, -1, -1) p 0 = p 0 p = p i = -p i p μ p μ = p 0 p 0 + p i p i = E c 2 - p 2 = (m c) 2 H = c p 2
Areas and Lengths in Polar Coordinates
 Kiryl Tsishchanka Areas and Lengths in Polar Coordinates In this section we develop the formula for the area of a region whose boundary is given by a polar equation. We need to use the formula for the
Kiryl Tsishchanka Areas and Lengths in Polar Coordinates In this section we develop the formula for the area of a region whose boundary is given by a polar equation. We need to use the formula for the
The Simply Typed Lambda Calculus
 Type Inference Instead of writing type annotations, can we use an algorithm to infer what the type annotations should be? That depends on the type system. For simple type systems the answer is yes, and
Type Inference Instead of writing type annotations, can we use an algorithm to infer what the type annotations should be? That depends on the type system. For simple type systems the answer is yes, and
Figure 1 T / K Explain, in terms of molecules, why the first part of the graph in Figure 1 is a line that slopes up from the origin.
 Q1.(a) Figure 1 shows how the entropy of a molecular substance X varies with temperature. Figure 1 T / K (i) Explain, in terms of molecules, why the entropy is zero when the temperature is zero Kelvin.
Q1.(a) Figure 1 shows how the entropy of a molecular substance X varies with temperature. Figure 1 T / K (i) Explain, in terms of molecules, why the entropy is zero when the temperature is zero Kelvin.
Areas and Lengths in Polar Coordinates
 Kiryl Tsishchanka Areas and Lengths in Polar Coordinates In this section we develop the formula for the area of a region whose boundary is given by a polar equation. We need to use the formula for the
Kiryl Tsishchanka Areas and Lengths in Polar Coordinates In this section we develop the formula for the area of a region whose boundary is given by a polar equation. We need to use the formula for the
Enthalpy data for the reacting species are given in the table below. The activation energy decreases when the temperature is increased.
 Q1.This question is about the reaction given below. O(g) + H 2O(g) O 2(g) + H 2(g) Enthalpy data for the reacting species are given in the table below. Substance O(g) H 2O(g) O 2(g) H 2(g) ΔH / kj mol
Q1.This question is about the reaction given below. O(g) + H 2O(g) O 2(g) + H 2(g) Enthalpy data for the reacting species are given in the table below. Substance O(g) H 2O(g) O 2(g) H 2(g) ΔH / kj mol
(1) Describe the process by which mercury atoms become excited in a fluorescent tube (3)
 Q1. (a) A fluorescent tube is filled with mercury vapour at low pressure. In order to emit electromagnetic radiation the mercury atoms must first be excited. (i) What is meant by an excited atom? (1) (ii)
Q1. (a) A fluorescent tube is filled with mercury vapour at low pressure. In order to emit electromagnetic radiation the mercury atoms must first be excited. (i) What is meant by an excited atom? (1) (ii)
Section 1: Listening and responding. Presenter: Niki Farfara MGTAV VCE Seminar 7 August 2016
 Section 1: Listening and responding Presenter: Niki Farfara MGTAV VCE Seminar 7 August 2016 Section 1: Listening and responding Section 1: Listening and Responding/ Aκουστική εξέταση Στο πρώτο μέρος της
Section 1: Listening and responding Presenter: Niki Farfara MGTAV VCE Seminar 7 August 2016 Section 1: Listening and responding Section 1: Listening and Responding/ Aκουστική εξέταση Στο πρώτο μέρος της
14 Lesson 2: The Omega Verb - Present Tense
 Lesson 2: The Omega Verb - Present Tense Day one I. Word Study and Grammar 1. Most Greek verbs end in in the first person singular. 2. The present tense is formed by adding endings to the present stem.
Lesson 2: The Omega Verb - Present Tense Day one I. Word Study and Grammar 1. Most Greek verbs end in in the first person singular. 2. The present tense is formed by adding endings to the present stem.
ΚΥΠΡΙΑΚΗ ΕΤΑΙΡΕΙΑ ΠΛΗΡΟΦΟΡΙΚΗΣ CYPRUS COMPUTER SOCIETY ΠΑΓΚΥΠΡΙΟΣ ΜΑΘΗΤΙΚΟΣ ΔΙΑΓΩΝΙΣΜΟΣ ΠΛΗΡΟΦΟΡΙΚΗΣ 6/5/2006
 Οδηγίες: Να απαντηθούν όλες οι ερωτήσεις. Ολοι οι αριθμοί που αναφέρονται σε όλα τα ερωτήματα είναι μικρότεροι το 1000 εκτός αν ορίζεται διαφορετικά στη διατύπωση του προβλήματος. Διάρκεια: 3,5 ώρες Καλή
Οδηγίες: Να απαντηθούν όλες οι ερωτήσεις. Ολοι οι αριθμοί που αναφέρονται σε όλα τα ερωτήματα είναι μικρότεροι το 1000 εκτός αν ορίζεται διαφορετικά στη διατύπωση του προβλήματος. Διάρκεια: 3,5 ώρες Καλή
Second Order Partial Differential Equations
 Chapter 7 Second Order Partial Differential Equations 7.1 Introduction A second order linear PDE in two independent variables (x, y Ω can be written as A(x, y u x + B(x, y u xy + C(x, y u u u + D(x, y
Chapter 7 Second Order Partial Differential Equations 7.1 Introduction A second order linear PDE in two independent variables (x, y Ω can be written as A(x, y u x + B(x, y u xy + C(x, y u u u + D(x, y
Finite Field Problems: Solutions
 Finite Field Problems: Solutions 1. Let f = x 2 +1 Z 11 [x] and let F = Z 11 [x]/(f), a field. Let Solution: F =11 2 = 121, so F = 121 1 = 120. The possible orders are the divisors of 120. Solution: The
Finite Field Problems: Solutions 1. Let f = x 2 +1 Z 11 [x] and let F = Z 11 [x]/(f), a field. Let Solution: F =11 2 = 121, so F = 121 1 = 120. The possible orders are the divisors of 120. Solution: The
Exercises 10. Find a fundamental matrix of the given system of equations. Also find the fundamental matrix Φ(t) satisfying Φ(0) = I. 1.
 Exercises 0 More exercises are available in Elementary Differential Equations. If you have a problem to solve any of them, feel free to come to office hour. Problem Find a fundamental matrix of the given
Exercises 0 More exercises are available in Elementary Differential Equations. If you have a problem to solve any of them, feel free to come to office hour. Problem Find a fundamental matrix of the given
Assalamu `alaikum wr. wb.
 LUMP SUM Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. LUMP SUM Lump sum lump sum lump sum. lump sum fixed price lump sum lump
LUMP SUM Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. LUMP SUM Lump sum lump sum lump sum. lump sum fixed price lump sum lump
Fourier Series. MATH 211, Calculus II. J. Robert Buchanan. Spring Department of Mathematics
 Fourier Series MATH 211, Calculus II J. Robert Buchanan Department of Mathematics Spring 2018 Introduction Not all functions can be represented by Taylor series. f (k) (c) A Taylor series f (x) = (x c)
Fourier Series MATH 211, Calculus II J. Robert Buchanan Department of Mathematics Spring 2018 Introduction Not all functions can be represented by Taylor series. f (k) (c) A Taylor series f (x) = (x c)
Electronic Supplementary Information (ESI)
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information (ESI) CPh 3 as a functional group in P-heterocyclic
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information (ESI) CPh 3 as a functional group in P-heterocyclic
b. Use the parametrization from (a) to compute the area of S a as S a ds. Be sure to substitute for ds!
 MTH U341 urface Integrals, tokes theorem, the divergence theorem To be turned in Wed., Dec. 1. 1. Let be the sphere of radius a, x 2 + y 2 + z 2 a 2. a. Use spherical coordinates (with ρ a) to parametrize.
MTH U341 urface Integrals, tokes theorem, the divergence theorem To be turned in Wed., Dec. 1. 1. Let be the sphere of radius a, x 2 + y 2 + z 2 a 2. a. Use spherical coordinates (with ρ a) to parametrize.
Μεταπτυχιακή διατριβή. Ανδρέας Παπαευσταθίου
 Σχολή Γεωτεχνικών Επιστημών και Διαχείρισης Περιβάλλοντος Μεταπτυχιακή διατριβή Κτίρια σχεδόν μηδενικής ενεργειακής κατανάλωσης :Αξιολόγηση συστημάτων θέρμανσης -ψύξης και ΑΠΕ σε οικιστικά κτίρια στην
Σχολή Γεωτεχνικών Επιστημών και Διαχείρισης Περιβάλλοντος Μεταπτυχιακή διατριβή Κτίρια σχεδόν μηδενικής ενεργειακής κατανάλωσης :Αξιολόγηση συστημάτων θέρμανσης -ψύξης και ΑΠΕ σε οικιστικά κτίρια στην
Notes on the Open Economy
 Notes on the Open Econom Ben J. Heijdra Universit of Groningen April 24 Introduction In this note we stud the two-countr model of Table.4 in more detail. restated here for convenience. The model is Table.4.
Notes on the Open Econom Ben J. Heijdra Universit of Groningen April 24 Introduction In this note we stud the two-countr model of Table.4 in more detail. restated here for convenience. The model is Table.4.
[1] P Q. Fig. 3.1
![[1] P Q. Fig. 3.1 [1] P Q. Fig. 3.1](/thumbs/79/80362156.jpg) 1 (a) Define resistance....... [1] (b) The smallest conductor within a computer processing chip can be represented as a rectangular block that is one atom high, four atoms wide and twenty atoms long. One
1 (a) Define resistance....... [1] (b) The smallest conductor within a computer processing chip can be represented as a rectangular block that is one atom high, four atoms wide and twenty atoms long. One
Απόκριση σε Μοναδιαία Ωστική Δύναμη (Unit Impulse) Απόκριση σε Δυνάμεις Αυθαίρετα Μεταβαλλόμενες με το Χρόνο. Απόστολος Σ.
 Απόκριση σε Δυνάμεις Αυθαίρετα Μεταβαλλόμενες με το Χρόνο The time integral of a force is referred to as impulse, is determined by and is obtained from: Newton s 2 nd Law of motion states that the action
Απόκριση σε Δυνάμεις Αυθαίρετα Μεταβαλλόμενες με το Χρόνο The time integral of a force is referred to as impulse, is determined by and is obtained from: Newton s 2 nd Law of motion states that the action
ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΕΙΡΑΙΑ ΤΜΗΜΑ ΝΑΥΤΙΛΙΑΚΩΝ ΣΠΟΥΔΩΝ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΗΝ ΝΑΥΤΙΛΙΑ
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΕΙΡΑΙΑ ΤΜΗΜΑ ΝΑΥΤΙΛΙΑΚΩΝ ΣΠΟΥΔΩΝ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΗΝ ΝΑΥΤΙΛΙΑ ΝΟΜΙΚΟ ΚΑΙ ΘΕΣΜΙΚΟ ΦΟΡΟΛΟΓΙΚΟ ΠΛΑΙΣΙΟ ΚΤΗΣΗΣ ΚΑΙ ΕΚΜΕΤΑΛΛΕΥΣΗΣ ΠΛΟΙΟΥ ΔΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ που υποβλήθηκε στο
ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΕΙΡΑΙΑ ΤΜΗΜΑ ΝΑΥΤΙΛΙΑΚΩΝ ΣΠΟΥΔΩΝ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΗΝ ΝΑΥΤΙΛΙΑ ΝΟΜΙΚΟ ΚΑΙ ΘΕΣΜΙΚΟ ΦΟΡΟΛΟΓΙΚΟ ΠΛΑΙΣΙΟ ΚΤΗΣΗΣ ΚΑΙ ΕΚΜΕΤΑΛΛΕΥΣΗΣ ΠΛΟΙΟΥ ΔΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ που υποβλήθηκε στο
Technical Information T-9100 SI. Suva. refrigerants. Thermodynamic Properties of. Suva Refrigerant [R-410A (50/50)]
![Technical Information T-9100 SI. Suva. refrigerants. Thermodynamic Properties of. Suva Refrigerant [R-410A (50/50)] Technical Information T-9100 SI. Suva. refrigerants. Thermodynamic Properties of. Suva Refrigerant [R-410A (50/50)]](/thumbs/89/98928029.jpg) d Suva refrigerants Technical Information T-9100SI Thermodynamic Properties of Suva 9100 Refrigerant [R-410A (50/50)] Thermodynamic Properties of Suva 9100 Refrigerant SI Units New tables of the thermodynamic
d Suva refrigerants Technical Information T-9100SI Thermodynamic Properties of Suva 9100 Refrigerant [R-410A (50/50)] Thermodynamic Properties of Suva 9100 Refrigerant SI Units New tables of the thermodynamic
ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΤΜΗΜΑ ΟΔΟΝΤΙΑΤΡΙΚΗΣ ΕΡΓΑΣΤΗΡΙΟ ΟΔΟΝΤΙΚΗΣ ΚΑΙ ΑΝΩΤΕΡΑΣ ΠΡΟΣΘΕΤΙΚΗΣ
 ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΤΜΗΜΑ ΟΔΟΝΤΙΑΤΡΙΚΗΣ ΕΡΓΑΣΤΗΡΙΟ ΟΔΟΝΤΙΚΗΣ ΚΑΙ ΑΝΩΤΕΡΑΣ ΠΡΟΣΘΕΤΙΚΗΣ ΣΥΓΚΡΙΤΙΚΗ ΜΕΛΕΤΗ ΤΗΣ ΣΥΓΚΡΑΤΗΤΙΚΗΣ ΙΚΑΝΟΤΗΤΑΣ ΟΡΙΣΜΕΝΩΝ ΠΡΟΚΑΤΑΣΚΕΥΑΣΜΕΝΩΝ ΣΥΝΔΕΣΜΩΝ ΑΚΡΙΒΕΙΑΣ
ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΤΜΗΜΑ ΟΔΟΝΤΙΑΤΡΙΚΗΣ ΕΡΓΑΣΤΗΡΙΟ ΟΔΟΝΤΙΚΗΣ ΚΑΙ ΑΝΩΤΕΡΑΣ ΠΡΟΣΘΕΤΙΚΗΣ ΣΥΓΚΡΙΤΙΚΗ ΜΕΛΕΤΗ ΤΗΣ ΣΥΓΚΡΑΤΗΤΙΚΗΣ ΙΚΑΝΟΤΗΤΑΣ ΟΡΙΣΜΕΝΩΝ ΠΡΟΚΑΤΑΣΚΕΥΑΣΜΕΝΩΝ ΣΥΝΔΕΣΜΩΝ ΑΚΡΙΒΕΙΑΣ
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΤΜΗΜΑ ΝΟΣΗΛΕΥΤΙΚΗΣ
 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΤΜΗΜΑ ΝΟΣΗΛΕΥΤΙΚΗΣ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΨΥΧΟΛΟΓΙΚΕΣ ΕΠΙΠΤΩΣΕΙΣ ΣΕ ΓΥΝΑΙΚΕΣ ΜΕΤΑ ΑΠΟ ΜΑΣΤΕΚΤΟΜΗ ΓΕΩΡΓΙΑ ΤΡΙΣΟΚΚΑ Λευκωσία 2012 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΤΜΗΜΑ ΝΟΣΗΛΕΥΤΙΚΗΣ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΨΥΧΟΛΟΓΙΚΕΣ ΕΠΙΠΤΩΣΕΙΣ ΣΕ ΓΥΝΑΙΚΕΣ ΜΕΤΑ ΑΠΟ ΜΑΣΤΕΚΤΟΜΗ ΓΕΩΡΓΙΑ ΤΡΙΣΟΚΚΑ Λευκωσία 2012 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ
Nowhere-zero flows Let be a digraph, Abelian group. A Γ-circulation in is a mapping : such that, where, and : tail in X, head in
 Nowhere-zero flows Let be a digraph, Abelian group. A Γ-circulation in is a mapping : such that, where, and : tail in X, head in : tail in X, head in A nowhere-zero Γ-flow is a Γ-circulation such that
Nowhere-zero flows Let be a digraph, Abelian group. A Γ-circulation in is a mapping : such that, where, and : tail in X, head in : tail in X, head in A nowhere-zero Γ-flow is a Γ-circulation such that
CRASH COURSE IN PRECALCULUS
 CRASH COURSE IN PRECALCULUS Shiah-Sen Wang The graphs are prepared by Chien-Lun Lai Based on : Precalculus: Mathematics for Calculus by J. Stuwart, L. Redin & S. Watson, 6th edition, 01, Brooks/Cole Chapter
CRASH COURSE IN PRECALCULUS Shiah-Sen Wang The graphs are prepared by Chien-Lun Lai Based on : Precalculus: Mathematics for Calculus by J. Stuwart, L. Redin & S. Watson, 6th edition, 01, Brooks/Cole Chapter
department listing department name αχχουντσ ϕανε βαλικτ δδσϕηασδδη σδηφγ ασκϕηλκ τεχηνιχαλ αλαν ϕουν διξ τεχηνιχαλ ϕοην µαριανι
 She selects the option. Jenny starts with the al listing. This has employees listed within She drills down through the employee. The inferred ER sttricture relates this to the redcords in the databasee
She selects the option. Jenny starts with the al listing. This has employees listed within She drills down through the employee. The inferred ER sttricture relates this to the redcords in the databasee
ΑΝΑΠΤΥΞΗ ΠΡΟΓΡΑΜΜΑΤΩΝ ΕΚΠΑΙΔΕΥΣΗΣ ΜΕ ΣΤΟΧΟ ΤΗΝ ΠΕΡΙΒΑΛΛΟΝΤΙΚΗ ΕΥΑΙΣΘΗΤΟΠΟΙΗΣΗ ΑΤΟΜΩΝ ΜΕ ΕΙΔΙΚΕΣ ΑΝΑΓΚΕΣ ΚΑΙ ΤΗΝ ΚΟΙΝΩΝΙΚΗ ΤΟΥΣ ΕΝΣΩΜΑΤΩΣΗ
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΙΑΣ ΠΑΙΔΑΓΩΓΙΚΟ ΤΜΗΜΑ ΕΙΔΙΚΗΣ ΑΓΩΓΗΣ ΧΡΙΣΤΙΝΑ Σ. ΛΑΠΠΑ ΑΝΑΠΤΥΞΗ ΠΡΟΓΡΑΜΜΑΤΩΝ ΕΚΠΑΙΔΕΥΣΗΣ ΜΕ ΣΤΟΧΟ ΤΗΝ ΠΕΡΙΒΑΛΛΟΝΤΙΚΗ ΕΥΑΙΣΘΗΤΟΠΟΙΗΣΗ ΑΤΟΜΩΝ ΜΕ ΕΙΔΙΚΕΣ ΑΝΑΓΚΕΣ ΚΑΙ ΤΗΝ ΚΟΙΝΩΝΙΚΗ ΤΟΥΣ
ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΙΑΣ ΠΑΙΔΑΓΩΓΙΚΟ ΤΜΗΜΑ ΕΙΔΙΚΗΣ ΑΓΩΓΗΣ ΧΡΙΣΤΙΝΑ Σ. ΛΑΠΠΑ ΑΝΑΠΤΥΞΗ ΠΡΟΓΡΑΜΜΑΤΩΝ ΕΚΠΑΙΔΕΥΣΗΣ ΜΕ ΣΤΟΧΟ ΤΗΝ ΠΕΡΙΒΑΛΛΟΝΤΙΚΗ ΕΥΑΙΣΘΗΤΟΠΟΙΗΣΗ ΑΤΟΜΩΝ ΜΕ ΕΙΔΙΚΕΣ ΑΝΑΓΚΕΣ ΚΑΙ ΤΗΝ ΚΟΙΝΩΝΙΚΗ ΤΟΥΣ
the total number of electrons passing through the lamp.
 1. A 12 V 36 W lamp is lit to normal brightness using a 12 V car battery of negligible internal resistance. The lamp is switched on for one hour (3600 s). For the time of 1 hour, calculate (i) the energy
1. A 12 V 36 W lamp is lit to normal brightness using a 12 V car battery of negligible internal resistance. The lamp is switched on for one hour (3600 s). For the time of 1 hour, calculate (i) the energy
DuPont Suva. DuPont. Thermodynamic Properties of. Refrigerant (R-410A) Technical Information. refrigerants T-410A ENG
 Technical Information T-410A ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 410A Refrigerant (R-410A) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Technical Information T-410A ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 410A Refrigerant (R-410A) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
ST5224: Advanced Statistical Theory II
 ST5224: Advanced Statistical Theory II 2014/2015: Semester II Tutorial 7 1. Let X be a sample from a population P and consider testing hypotheses H 0 : P = P 0 versus H 1 : P = P 1, where P j is a known
ST5224: Advanced Statistical Theory II 2014/2015: Semester II Tutorial 7 1. Let X be a sample from a population P and consider testing hypotheses H 0 : P = P 0 versus H 1 : P = P 1, where P j is a known
ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΤΜΗΜΑ ΠΛΗΡΟΦΟΡΙΚΗΣ. ΕΠΛ342: Βάσεις Δεδομένων. Χειμερινό Εξάμηνο Φροντιστήριο 10 ΛΥΣΕΙΣ. Επερωτήσεις SQL
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΤΜΗΜΑ ΠΛΗΡΟΦΟΡΙΚΗΣ ΕΠΛ342: Βάσεις Δεδομένων Χειμερινό Εξάμηνο 2013 Φροντιστήριο 10 ΛΥΣΕΙΣ Επερωτήσεις SQL Άσκηση 1 Για το ακόλουθο σχήμα Suppliers(sid, sname, address) Parts(pid, pname,
ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΤΜΗΜΑ ΠΛΗΡΟΦΟΡΙΚΗΣ ΕΠΛ342: Βάσεις Δεδομένων Χειμερινό Εξάμηνο 2013 Φροντιστήριο 10 ΛΥΣΕΙΣ Επερωτήσεις SQL Άσκηση 1 Για το ακόλουθο σχήμα Suppliers(sid, sname, address) Parts(pid, pname,
ANSWERSHEET (TOPIC = DIFFERENTIAL CALCULUS) COLLECTION #2. h 0 h h 0 h h 0 ( ) g k = g 0 + g 1 + g g 2009 =?
 Teko Classes IITJEE/AIEEE Maths by SUHAAG SIR, Bhopal, Ph (0755) 3 00 000 www.tekoclasses.com ANSWERSHEET (TOPIC DIFFERENTIAL CALCULUS) COLLECTION # Question Type A.Single Correct Type Q. (A) Sol least
Teko Classes IITJEE/AIEEE Maths by SUHAAG SIR, Bhopal, Ph (0755) 3 00 000 www.tekoclasses.com ANSWERSHEET (TOPIC DIFFERENTIAL CALCULUS) COLLECTION # Question Type A.Single Correct Type Q. (A) Sol least
Lecture 2. Soundness and completeness of propositional logic
 Lecture 2 Soundness and completeness of propositional logic February 9, 2004 1 Overview Review of natural deduction. Soundness and completeness. Semantics of propositional formulas. Soundness proof. Completeness
Lecture 2 Soundness and completeness of propositional logic February 9, 2004 1 Overview Review of natural deduction. Soundness and completeness. Semantics of propositional formulas. Soundness proof. Completeness
Every set of first-order formulas is equivalent to an independent set
 Every set of first-order formulas is equivalent to an independent set May 6, 2008 Abstract A set of first-order formulas, whatever the cardinality of the set of symbols, is equivalent to an independent
Every set of first-order formulas is equivalent to an independent set May 6, 2008 Abstract A set of first-order formulas, whatever the cardinality of the set of symbols, is equivalent to an independent
Approximation of distance between locations on earth given by latitude and longitude
 Approximation of distance between locations on earth given by latitude and longitude Jan Behrens 2012-12-31 In this paper we shall provide a method to approximate distances between two points on earth
Approximation of distance between locations on earth given by latitude and longitude Jan Behrens 2012-12-31 In this paper we shall provide a method to approximate distances between two points on earth
Matrices and Determinants
 Matrices and Determinants SUBJECTIVE PROBLEMS: Q 1. For what value of k do the following system of equations possess a non-trivial (i.e., not all zero) solution over the set of rationals Q? x + ky + 3z
Matrices and Determinants SUBJECTIVE PROBLEMS: Q 1. For what value of k do the following system of equations possess a non-trivial (i.e., not all zero) solution over the set of rationals Q? x + ky + 3z
DuPont Suva 95 Refrigerant
 Technical Information T-95 SI DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Technical Information T-95 SI DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Inverse trigonometric functions & General Solution of Trigonometric Equations. ------------------ ----------------------------- -----------------
 Inverse trigonometric functions & General Solution of Trigonometric Equations. 1. Sin ( ) = a) b) c) d) Ans b. Solution : Method 1. Ans a: 17 > 1 a) is rejected. w.k.t Sin ( sin ) = d is rejected. If sin
Inverse trigonometric functions & General Solution of Trigonometric Equations. 1. Sin ( ) = a) b) c) d) Ans b. Solution : Method 1. Ans a: 17 > 1 a) is rejected. w.k.t Sin ( sin ) = d is rejected. If sin
ΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ. ΘΕΜΑ: «ιερεύνηση της σχέσης µεταξύ φωνηµικής επίγνωσης και ορθογραφικής δεξιότητας σε παιδιά προσχολικής ηλικίας»
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΑΙΓΑΙΟΥ ΣΧΟΛΗ ΑΝΘΡΩΠΙΣΤΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΤΜΗΜΑ ΕΠΙΣΤΗΜΩΝ ΤΗΣ ΠΡΟΣΧΟΛΙΚΗΣ ΑΓΩΓΗΣ ΚΑΙ ΤΟΥ ΕΚΠΑΙ ΕΥΤΙΚΟΥ ΣΧΕ ΙΑΣΜΟΥ «ΠΑΙ ΙΚΟ ΒΙΒΛΙΟ ΚΑΙ ΠΑΙ ΑΓΩΓΙΚΟ ΥΛΙΚΟ» ΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ που εκπονήθηκε για τη
ΠΑΝΕΠΙΣΤΗΜΙΟ ΑΙΓΑΙΟΥ ΣΧΟΛΗ ΑΝΘΡΩΠΙΣΤΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΤΜΗΜΑ ΕΠΙΣΤΗΜΩΝ ΤΗΣ ΠΡΟΣΧΟΛΙΚΗΣ ΑΓΩΓΗΣ ΚΑΙ ΤΟΥ ΕΚΠΑΙ ΕΥΤΙΚΟΥ ΣΧΕ ΙΑΣΜΟΥ «ΠΑΙ ΙΚΟ ΒΙΒΛΙΟ ΚΑΙ ΠΑΙ ΑΓΩΓΙΚΟ ΥΛΙΚΟ» ΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ που εκπονήθηκε για τη
( y) Partial Differential Equations
 Partial Dierential Equations Linear P.D.Es. contains no owers roducts o the deendent variables / an o its derivatives can occasionall be solved. Consider eamle ( ) a (sometimes written as a ) we can integrate
Partial Dierential Equations Linear P.D.Es. contains no owers roducts o the deendent variables / an o its derivatives can occasionall be solved. Consider eamle ( ) a (sometimes written as a ) we can integrate
DuPont Suva 95 Refrigerant
 Technical Information T-95 ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Technical Information T-95 ENG DuPont Suva refrigerants Thermodynamic Properties of DuPont Suva 95 Refrigerant (R-508B) The DuPont Oval Logo, The miracles of science, and Suva, are trademarks or registered
Μηχανική Μάθηση Hypothesis Testing
 ΕΛΛΗΝΙΚΗ ΔΗΜΟΚΡΑΤΙΑ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΡΗΤΗΣ Μηχανική Μάθηση Hypothesis Testing Γιώργος Μπορμπουδάκης Τμήμα Επιστήμης Υπολογιστών Procedure 1. Form the null (H 0 ) and alternative (H 1 ) hypothesis 2. Consider
ΕΛΛΗΝΙΚΗ ΔΗΜΟΚΡΑΤΙΑ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΡΗΤΗΣ Μηχανική Μάθηση Hypothesis Testing Γιώργος Μπορμπουδάκης Τμήμα Επιστήμης Υπολογιστών Procedure 1. Form the null (H 0 ) and alternative (H 1 ) hypothesis 2. Consider
Lifting Entry (continued)
 ifting Entry (continued) Basic planar dynamics of motion, again Yet another equilibrium glide Hypersonic phugoid motion Planar state equations MARYAN 1 01 avid. Akin - All rights reserved http://spacecraft.ssl.umd.edu
ifting Entry (continued) Basic planar dynamics of motion, again Yet another equilibrium glide Hypersonic phugoid motion Planar state equations MARYAN 1 01 avid. Akin - All rights reserved http://spacecraft.ssl.umd.edu
ΕΙΣΑΓΩΓΗ ΣΤΗ ΣΤΑΤΙΣΤΙΚΗ ΑΝΑΛΥΣΗ
 ΕΙΣΑΓΩΓΗ ΣΤΗ ΣΤΑΤΙΣΤΙΚΗ ΑΝΑΛΥΣΗ ΕΛΕΝΑ ΦΛΟΚΑ Επίκουρος Καθηγήτρια Τµήµα Φυσικής, Τοµέας Φυσικής Περιβάλλοντος- Μετεωρολογίας ΓΕΝΙΚΟΙ ΟΡΙΣΜΟΙ Πληθυσµός Σύνολο ατόµων ή αντικειµένων στα οποία αναφέρονται
ΕΙΣΑΓΩΓΗ ΣΤΗ ΣΤΑΤΙΣΤΙΚΗ ΑΝΑΛΥΣΗ ΕΛΕΝΑ ΦΛΟΚΑ Επίκουρος Καθηγήτρια Τµήµα Φυσικής, Τοµέας Φυσικής Περιβάλλοντος- Μετεωρολογίας ΓΕΝΙΚΟΙ ΟΡΙΣΜΟΙ Πληθυσµός Σύνολο ατόµων ή αντικειµένων στα οποία αναφέρονται
TMA4115 Matematikk 3
 TMA4115 Matematikk 3 Andrew Stacey Norges Teknisk-Naturvitenskapelige Universitet Trondheim Spring 2010 Lecture 12: Mathematics Marvellous Matrices Andrew Stacey Norges Teknisk-Naturvitenskapelige Universitet
TMA4115 Matematikk 3 Andrew Stacey Norges Teknisk-Naturvitenskapelige Universitet Trondheim Spring 2010 Lecture 12: Mathematics Marvellous Matrices Andrew Stacey Norges Teknisk-Naturvitenskapelige Universitet
Srednicki Chapter 55
 Srednicki Chapter 55 QFT Problems & Solutions A. George August 3, 03 Srednicki 55.. Use equations 55.3-55.0 and A i, A j ] = Π i, Π j ] = 0 (at equal times) to verify equations 55.-55.3. This is our third
Srednicki Chapter 55 QFT Problems & Solutions A. George August 3, 03 Srednicki 55.. Use equations 55.3-55.0 and A i, A j ] = Π i, Π j ] = 0 (at equal times) to verify equations 55.-55.3. This is our third
ΠΑΝΔΠΗΣΖΜΗΟ ΠΑΣΡΩΝ ΣΜΖΜΑ ΖΛΔΚΣΡΟΛΟΓΩΝ ΜΖΥΑΝΗΚΩΝ ΚΑΗ ΣΔΥΝΟΛΟΓΗΑ ΤΠΟΛΟΓΗΣΩΝ ΣΟΜΔΑ ΤΣΖΜΑΣΩΝ ΖΛΔΚΣΡΗΚΖ ΔΝΔΡΓΔΗΑ
 ΠΑΝΔΠΗΣΖΜΗΟ ΠΑΣΡΩΝ ΣΜΖΜΑ ΖΛΔΚΣΡΟΛΟΓΩΝ ΜΖΥΑΝΗΚΩΝ ΚΑΗ ΣΔΥΝΟΛΟΓΗΑ ΤΠΟΛΟΓΗΣΩΝ ΣΟΜΔΑ ΤΣΖΜΑΣΩΝ ΖΛΔΚΣΡΗΚΖ ΔΝΔΡΓΔΗΑ Γηπισκαηηθή Δξγαζία ηνπ Φνηηεηή ηνπ ηκήκαηνο Ζιεθηξνιόγσλ Μεραληθώλ θαη Σερλνινγίαο Ζιεθηξνληθώλ
ΠΑΝΔΠΗΣΖΜΗΟ ΠΑΣΡΩΝ ΣΜΖΜΑ ΖΛΔΚΣΡΟΛΟΓΩΝ ΜΖΥΑΝΗΚΩΝ ΚΑΗ ΣΔΥΝΟΛΟΓΗΑ ΤΠΟΛΟΓΗΣΩΝ ΣΟΜΔΑ ΤΣΖΜΑΣΩΝ ΖΛΔΚΣΡΗΚΖ ΔΝΔΡΓΔΗΑ Γηπισκαηηθή Δξγαζία ηνπ Φνηηεηή ηνπ ηκήκαηνο Ζιεθηξνιόγσλ Μεραληθώλ θαη Σερλνινγίαο Ζιεθηξνληθώλ
Ordinal Arithmetic: Addition, Multiplication, Exponentiation and Limit
 Ordinal Arithmetic: Addition, Multiplication, Exponentiation and Limit Ting Zhang Stanford May 11, 2001 Stanford, 5/11/2001 1 Outline Ordinal Classification Ordinal Addition Ordinal Multiplication Ordinal
Ordinal Arithmetic: Addition, Multiplication, Exponentiation and Limit Ting Zhang Stanford May 11, 2001 Stanford, 5/11/2001 1 Outline Ordinal Classification Ordinal Addition Ordinal Multiplication Ordinal
Derivation of Optical-Bloch Equations
 Appendix C Derivation of Optical-Bloch Equations In this appendix the optical-bloch equations that give the populations and coherences for an idealized three-level Λ system, Fig. 3. on page 47, will be
Appendix C Derivation of Optical-Bloch Equations In this appendix the optical-bloch equations that give the populations and coherences for an idealized three-level Λ system, Fig. 3. on page 47, will be
Διπλωματική Εργασία. Μελέτη των μηχανικών ιδιοτήτων των stents που χρησιμοποιούνται στην Ιατρική. Αντωνίου Φάνης
 Διπλωματική Εργασία Μελέτη των μηχανικών ιδιοτήτων των stents που χρησιμοποιούνται στην Ιατρική Αντωνίου Φάνης Επιβλέπουσες: Θεοδώρα Παπαδοπούλου, Ομότιμη Καθηγήτρια ΕΜΠ Ζάννη-Βλαστού Ρόζα, Καθηγήτρια
Διπλωματική Εργασία Μελέτη των μηχανικών ιδιοτήτων των stents που χρησιμοποιούνται στην Ιατρική Αντωνίου Φάνης Επιβλέπουσες: Θεοδώρα Παπαδοπούλου, Ομότιμη Καθηγήτρια ΕΜΠ Ζάννη-Βλαστού Ρόζα, Καθηγήτρια
D Alembert s Solution to the Wave Equation
 D Alembert s Solution to the Wave Equation MATH 467 Partial Differential Equations J. Robert Buchanan Department of Mathematics Fall 2018 Objectives In this lesson we will learn: a change of variable technique
D Alembert s Solution to the Wave Equation MATH 467 Partial Differential Equations J. Robert Buchanan Department of Mathematics Fall 2018 Objectives In this lesson we will learn: a change of variable technique
Higher Derivative Gravity Theories
 Higher Derivative Gravity Theories Black Holes in AdS space-times James Mashiyane Supervisor: Prof Kevin Goldstein University of the Witwatersrand Second Mandelstam, 20 January 2018 James Mashiyane WITS)
Higher Derivative Gravity Theories Black Holes in AdS space-times James Mashiyane Supervisor: Prof Kevin Goldstein University of the Witwatersrand Second Mandelstam, 20 January 2018 James Mashiyane WITS)
Econ 2110: Fall 2008 Suggested Solutions to Problem Set 8 questions or comments to Dan Fetter 1
 Eon : Fall 8 Suggested Solutions to Problem Set 8 Email questions or omments to Dan Fetter Problem. Let X be a salar with density f(x, θ) (θx + θ) [ x ] with θ. (a) Find the most powerful level α test
Eon : Fall 8 Suggested Solutions to Problem Set 8 Email questions or omments to Dan Fetter Problem. Let X be a salar with density f(x, θ) (θx + θ) [ x ] with θ. (a) Find the most powerful level α test
Example of the Baum-Welch Algorithm
 Example of the Baum-Welch Algorithm Larry Moss Q520, Spring 2008 1 Our corpus c We start with a very simple corpus. We take the set Y of unanalyzed words to be {ABBA, BAB}, and c to be given by c(abba)
Example of the Baum-Welch Algorithm Larry Moss Q520, Spring 2008 1 Our corpus c We start with a very simple corpus. We take the set Y of unanalyzed words to be {ABBA, BAB}, and c to be given by c(abba)
(C) 2010 Pearson Education, Inc. All rights reserved.
 Connectionless transmission with datagrams. Connection-oriented transmission is like the telephone system You dial and are given a connection to the telephone of fthe person with whom you wish to communicate.
Connectionless transmission with datagrams. Connection-oriented transmission is like the telephone system You dial and are given a connection to the telephone of fthe person with whom you wish to communicate.
CHAPTER 101 FOURIER SERIES FOR PERIODIC FUNCTIONS OF PERIOD
 CHAPTER FOURIER SERIES FOR PERIODIC FUNCTIONS OF PERIOD EXERCISE 36 Page 66. Determine the Fourier series for the periodic function: f(x), when x +, when x which is periodic outside this rge of period.
CHAPTER FOURIER SERIES FOR PERIODIC FUNCTIONS OF PERIOD EXERCISE 36 Page 66. Determine the Fourier series for the periodic function: f(x), when x +, when x which is periodic outside this rge of period.
of the methanol-dimethylamine complex
 Electronic Supplementary Information for: Fundamental and overtone virational spectroscopy, enthalpy of hydrogen ond formation and equilirium constant determination of the methanol-dimethylamine complex
Electronic Supplementary Information for: Fundamental and overtone virational spectroscopy, enthalpy of hydrogen ond formation and equilirium constant determination of the methanol-dimethylamine complex
Solutions to the Schrodinger equation atomic orbitals. Ψ 1 s Ψ 2 s Ψ 2 px Ψ 2 py Ψ 2 pz
 Solutions to the Schrodinger equation atomic orbitals Ψ 1 s Ψ 2 s Ψ 2 px Ψ 2 py Ψ 2 pz ybridization Valence Bond Approach to bonding sp 3 (Ψ 2 s + Ψ 2 px + Ψ 2 py + Ψ 2 pz) sp 2 (Ψ 2 s + Ψ 2 px + Ψ 2 py)
Solutions to the Schrodinger equation atomic orbitals Ψ 1 s Ψ 2 s Ψ 2 px Ψ 2 py Ψ 2 pz ybridization Valence Bond Approach to bonding sp 3 (Ψ 2 s + Ψ 2 px + Ψ 2 py + Ψ 2 pz) sp 2 (Ψ 2 s + Ψ 2 px + Ψ 2 py)
Test Data Management in Practice
 Problems, Concepts, and the Swisscom Test Data Organizer Do you have issues with your legal and compliance department because test environments contain sensitive data outsourcing partners must not see?
Problems, Concepts, and the Swisscom Test Data Organizer Do you have issues with your legal and compliance department because test environments contain sensitive data outsourcing partners must not see?
Section 8.2 Graphs of Polar Equations
 Section 8. Graphs of Polar Equations Graphing Polar Equations The graph of a polar equation r = f(θ), or more generally F(r,θ) = 0, consists of all points P that have at least one polar representation
Section 8. Graphs of Polar Equations Graphing Polar Equations The graph of a polar equation r = f(θ), or more generally F(r,θ) = 0, consists of all points P that have at least one polar representation
Modbus basic setup notes for IO-Link AL1xxx Master Block
 n Modbus has four tables/registers where data is stored along with their associated addresses. We will be using the holding registers from address 40001 to 49999 that are R/W 16 bit/word. Two tables that
n Modbus has four tables/registers where data is stored along with their associated addresses. We will be using the holding registers from address 40001 to 49999 that are R/W 16 bit/word. Two tables that
Lecture 34 Bootstrap confidence intervals
 Lecture 34 Bootstrap confidence intervals Confidence Intervals θ: an unknown parameter of interest We want to find limits θ and θ such that Gt = P nˆθ θ t If G 1 1 α is known, then P θ θ = P θ θ = 1 α
Lecture 34 Bootstrap confidence intervals Confidence Intervals θ: an unknown parameter of interest We want to find limits θ and θ such that Gt = P nˆθ θ t If G 1 1 α is known, then P θ θ = P θ θ = 1 α
Similarly, we may define hyperbolic functions cosh α and sinh α from the unit hyperbola
 Universit of Hperbolic Functions The trigonometric functions cos α an cos α are efine using the unit circle + b measuring the istance α in the counter-clockwise irection along the circumference of the
Universit of Hperbolic Functions The trigonometric functions cos α an cos α are efine using the unit circle + b measuring the istance α in the counter-clockwise irection along the circumference of the
Section 7.6 Double and Half Angle Formulas
 09 Section 7. Double and Half Angle Fmulas To derive the double-angles fmulas, we will use the sum of two angles fmulas that we developed in the last section. We will let α θ and β θ: cos(θ) cos(θ + θ)
09 Section 7. Double and Half Angle Fmulas To derive the double-angles fmulas, we will use the sum of two angles fmulas that we developed in the last section. We will let α θ and β θ: cos(θ) cos(θ + θ)
Practice Exam 2. Conceptual Questions. 1. State a Basic identity and then verify it. (a) Identity: Solution: One identity is csc(θ) = 1
 Conceptual Questions. State a Basic identity and then verify it. a) Identity: Solution: One identity is cscθ) = sinθ) Practice Exam b) Verification: Solution: Given the point of intersection x, y) of the
Conceptual Questions. State a Basic identity and then verify it. a) Identity: Solution: One identity is cscθ) = sinθ) Practice Exam b) Verification: Solution: Given the point of intersection x, y) of the
ΚΥΠΡΙΑΚΗ ΕΤΑΙΡΕΙΑ ΠΛΗΡΟΦΟΡΙΚΗΣ CYPRUS COMPUTER SOCIETY ΠΑΓΚΥΠΡΙΟΣ ΜΑΘΗΤΙΚΟΣ ΔΙΑΓΩΝΙΣΜΟΣ ΠΛΗΡΟΦΟΡΙΚΗΣ 11/3/2006
 ΠΑΓΚΥΠΡΙΟΣ ΜΑΘΗΤΙΚΟΣ ΔΙΑΓΩΝΙΣΜΟΣ ΠΛΗΡΟΦΟΡΙΚΗΣ 11/3/26 Οδηγίες: Να απαντηθούν όλες οι ερωτήσεις. Ολοι οι αριθμοί που αναφέρονται σε όλα τα ερωτήματα μικρότεροι το 1 εκτός αν ορίζεται διαφορετικά στη διατύπωση
ΠΑΓΚΥΠΡΙΟΣ ΜΑΘΗΤΙΚΟΣ ΔΙΑΓΩΝΙΣΜΟΣ ΠΛΗΡΟΦΟΡΙΚΗΣ 11/3/26 Οδηγίες: Να απαντηθούν όλες οι ερωτήσεις. Ολοι οι αριθμοί που αναφέρονται σε όλα τα ερωτήματα μικρότεροι το 1 εκτός αν ορίζεται διαφορετικά στη διατύπωση
Συστήματα Διαχείρισης Βάσεων Δεδομένων
 ΕΛΛΗΝΙΚΗ ΔΗΜΟΚΡΑΤΙΑ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΡΗΤΗΣ Συστήματα Διαχείρισης Βάσεων Δεδομένων Φροντιστήριο 9: Transactions - part 1 Δημήτρης Πλεξουσάκης Τμήμα Επιστήμης Υπολογιστών Tutorial on Undo, Redo and Undo/Redo
ΕΛΛΗΝΙΚΗ ΔΗΜΟΚΡΑΤΙΑ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΡΗΤΗΣ Συστήματα Διαχείρισης Βάσεων Δεδομένων Φροντιστήριο 9: Transactions - part 1 Δημήτρης Πλεξουσάκης Τμήμα Επιστήμης Υπολογιστών Tutorial on Undo, Redo and Undo/Redo
Supporting Information for: electron ligands: Complex formation, oxidation and
 Supporting Information for: The diverse reactions of PhI(OTf) 2 with common 2- electron ligands: Complex formation, oxidation and oxidative coupling Thomas P. Pell, Shannon A. Couchman, Sara Ibrahim, David
Supporting Information for: The diverse reactions of PhI(OTf) 2 with common 2- electron ligands: Complex formation, oxidation and oxidative coupling Thomas P. Pell, Shannon A. Couchman, Sara Ibrahim, David
Other Test Constructions: Likelihood Ratio & Bayes Tests
 Other Test Constructions: Likelihood Ratio & Bayes Tests Side-Note: So far we have seen a few approaches for creating tests such as Neyman-Pearson Lemma ( most powerful tests of H 0 : θ = θ 0 vs H 1 :
Other Test Constructions: Likelihood Ratio & Bayes Tests Side-Note: So far we have seen a few approaches for creating tests such as Neyman-Pearson Lemma ( most powerful tests of H 0 : θ = θ 0 vs H 1 :
Trigonometry 1.TRIGONOMETRIC RATIOS
 Trigonometry.TRIGONOMETRIC RATIOS. If a ray OP makes an angle with the positive direction of X-axis then y x i) Sin ii) cos r r iii) tan x y (x 0) iv) cot y x (y 0) y P v) sec x r (x 0) vi) cosec y r (y
Trigonometry.TRIGONOMETRIC RATIOS. If a ray OP makes an angle with the positive direction of X-axis then y x i) Sin ii) cos r r iii) tan x y (x 0) iv) cot y x (y 0) y P v) sec x r (x 0) vi) cosec y r (y
Math221: HW# 1 solutions
 Math: HW# solutions Andy Royston October, 5 7.5.7, 3 rd Ed. We have a n = b n = a = fxdx = xdx =, x cos nxdx = x sin nx n sin nxdx n = cos nx n = n n, x sin nxdx = x cos nx n + cos nxdx n cos n = + sin
Math: HW# solutions Andy Royston October, 5 7.5.7, 3 rd Ed. We have a n = b n = a = fxdx = xdx =, x cos nxdx = x sin nx n sin nxdx n = cos nx n = n n, x sin nxdx = x cos nx n + cos nxdx n cos n = + sin
