«Μηχανισµοί µεταγραφής Ρύθµιση γονιδιακής έκφρασης»
|
|
|
- Ευφήμιος Αντωνοπούλου
- 10 χρόνια πριν
- Προβολές:
Transcript
1 Εκπαιδευτικό Πρόγραµµα της Οµάδας Μοριακής Παθολογικής Ανατοµικής της Ενότητας «ΑΡΧΕΣ ΜΟΡΙΑΚΗΣ ΚΑΙ ΚΥΤΤΑΡΙΚΗΣ ΒΙΟΛΟΓΙΑΣ» Αθήνα, 6/2/2010 «Μηχανισµοί µεταγραφής Ρύθµιση γονιδιακής έκφρασης» ρ Α. Πίντζας ιευθυντής Ερευνών Επιστ. Υπεύθυνος Εργαστηρίου Γονιδιακής Ρύθµισης Ινστ. Βιολογικών Ερευνών και Βιοτεχνολογίας Εθνικό Ιδρυµα Ερευνών
2 Key Concepts In any given cell, most genes are expressed in low levels Only a small number of genes, whose products are specialised for the cell type are highly expressed ~10,000 expressed genes maybe common to most cell types of a higher eukaryote
3 Gene Transcription and Translation Transcription generates a single-stranded RNA identical in sequence with one of the strands of the duplex DNA. Translation converts the nucleotide sequence of mrna into the sequence of amino acids comprising a protein. The entire length of an mrna is not translated, but each mrna contains at least one coding region that is related to a protein sequence by the genetic code: each nucleotide triplet (codon) of the coding region represents one amino acid. Only one strand of a DNA duplex is transcribed into a messenger RNA. We distinguish the two strands of DNA as depicted in Figure 5.2: The strand of DNA that directs synthesis of the mrna via complementary base pairing is called the template strand or antisense strand. (Antisense is used as a general term to describe a sequence of DNA or RNA that is complementary to mrna.) The other DNA strand bears the same sequence as the mrna (except for possessing T instead of U), and is called the coding strand or sense strand.
4 Promoter structure and function (A) Organization of a generalized eukaryotic gene, showing the relative position of the transcription unit, basal promoter region (black box with bent arrow), and transcription factor binding sites (vertical bars). The position of transcription factor binding sites differs enormously between loci; (B) Idealized promoter in operation. Initiating transcription requires several dozen different proteins which interact with each other in specific ways. These include the RNA polymerase II holoenzyme complex ( 15 proteins); TATA-binding protein (TBP; 1 protein); TAFs (TBP-associated factors, also known as general transcription factors; 8 proteins); transcription factors; Transcription cofactors; and chromatin remodeling complexes
5 Introduction Initiation of transcription: a key mechanism to regulate gene expression -Initiation of transcription is a KEY step to the regulation of gene expression. -It requires the recognition of a particular gene promoter -This induces a multi-step recruitment of the pre-initiation complex. - Promoter recognition and positioning of the TFIID complex (transcription factor complex II D). The TATA box is involved. - The binding of TFIID induces the ordered recruitment of the other general transcription factors and RNA polymeraseii Promoter recognition is the most important step to initiate the transcription. TFIID plays a major role in this process. TFIID is composed of the TATA-Binding Protein (TBP) and several TBP-Associated Factors (TAFs) Pre-initiation complex: PIC In vitro, TBP is sufficient for promoter recognition and PIC assembly. What is the role of the TAFs? - Initiation of transcription
6 Introduction Transcription initiation: the in vivo situation and the role of TFIID In vitro situation/naked DNA chromatin In vivo situation/chromatin structure/other cis-and trans acting factors An entire TFIID is necessary for REGULATED transcription initiation in vivo: It controls the correct promoter recognition captured in chromatin structures and mediates bridging between activators and the basal transcription machinery. In vivo, TFIID is necessary to the control of transcription initiation TFIID, TBP and TAFs. -TFIID is a basal transcription factor, composed of TBP+TAFs (numbered 1,2 etc depending on their MW) -TAFs organized between them and around TBP in a particular order and stochiometry - Core TAFs necessary for stability of the complex. If one of these TAFs is missing, TFIID is destabilized and individual TAFs are degraded. Gangloff et al., Trends Biochem Sci., 2001 Muller and Tora, EMBO J., 2004
7 Introduction Other TAF-containing complexes are involved in transcriptional regulation HAT activity Muller and Tora, EMBO J., 2004
8 TFIID not so basal after all! TBP and TAFs are regulated Transcriptional regulation: rate, tissue specificity, isoforms Post-translational regulation: modifications, stability, cellular localization Alternative complexes: several TBP-containing and TBP-free TAF- containing complexes to control transcriptional initiation Target gene specificity, induced by cell signals and acting via specific interactions with TF Ex: TBP is up-regulated by the RAS signalling pathway; contribute to mechanisms of cellular transformation in colon carcinoma cell lines. (Johnson et al., Mol. Cell Biol.) a specialized isoform of TAF6 is induced and cleaved via apoptotic signaling pathways, to selectively alter transcription. (Bell et al., Mol. Cell.Biol.) Inactivation of TAF4 induces serum-independent autocrine growth and activation of the transforming growth factor beta signaling pathway. (Mengus et al., EMBO J.) Cell-type-selective induction of c-jun by TAF4b directs ovarian-specific transcription networks. (Geles et al., Proc Natl Acad Sci U S A
9 Οι µεταγραφικοί παράγοντες ρυθµιζονται απο κυτταρικά σήµατα Στο κλασικό µονοπάτι ΜΑΡ κινασών, επιτυγχάνεται µε την µετακίνηση της ίδιας της κινάσης ΜΑΡ στον πυρήνα, όπου φωσφορυλιώνει µεταγραφικούς παράγοντες- στόχους Ένα εναλλακτικό µονοπάτι είναι η φωσφορυλίωση ενός κυτταροπλασµατικού παράγοντα. Αυτός µπορεί να είναι ένας µεταγραφικός παράγοντας που στη συνέχεια µετακινείται στον πυρήνα ή µια πρωτεΐνη που ρυθµίζει έναν µεταγραφικό παράγοντα (π.χ. τον απελευθερώνει για να πάει στον πυρήνα)
10 Generalized signalling pathway
11 Ternary complexes of eukaryotic transcription factors and promoter DNA
12 οµές µεταγραφικών παραγόντων στο DNA ΙΙ
13 WNT signals through β-catenin
14 NF-κB activation
15 The interactions of Jun and Fos proteins components of AP-1 transcription factor The transcription factor AP-1 is formed by dimerization of Jun proteins (c-jun, JunB and JunD) or heterodimerization of a Jun protein with a Fos protein (c-fos, FosB, Fra-1 and Fra-2). AP-1 binds primarily to the 12-O-tetradecanoylphorbol-13- acetate (TPA) response element. Jun and Fos proteins contain a basic-region leucine zipper (bzip) domain and can interact with other bzip proteins, such as MAF and ATF, which allows them to target the camp response element (CRE), and with the p65 subunit of nuclear factor B (NF- B). A dominant-negative mutant form of c-jun (TAM67) interacts with any of the three Jun or four Fos family proteins and/or p65, inhibiting the transactivational activity of AP-1 and NF- B.
16 Regulation and function of genes coding for AP-1 factors Abundance of AP-1 proteins Activity of AP-1 factors Transcriptional regulation of genes Phosphorylation Modulation of protein stability JNKs (ubiquitination) c-jun ser 63, 73 Interaction with transcriptional coactivators e.g. CBP e.g. c-jun, c-fos
17 AP-1 and signal transduction c-fos c-jun Transcriptional activation of immediate early genes: c-fos, c-jun Cell surface stimuli P AAA AAA Translation P Heterodimer and homodimer formation Jun Fos Fra Jun Jun Jun JunJunB Fos Jun TGAGTCA DNA binding and regulation of target gene expression Fra JunB Jun Fos
18 Structure of the members of Ets family proteins Ets, DNA-binding (Ets) domain; HLH, helix loop helix domain (Pointed domain); AD, activation domain; ID, auto-inhibitory domain; RD, repression domain.
19 Functional cooperation of Ets family proteins with other transcription factors and co-activators on various cellular promoters and enhancers (a) Ets-1 or PEA-3 activates the MMP (matrix metalloproteinase) promoters in cooperation with AP1 (c-fos/c-jun) on the RRE (Ras-response element). (b) TCF (Elk-1, Sap-1) activates the c-fos promoter in cooperation with SRF (serum response factor). (c) PU.1 activates myeloid-specific promoters in cooperation with AML1 and C/EBP. (d) Fli-1 activates megakaryocyte-specific promoters in cooperation with GATA-1. (e) PU.1 activates the immunoglobulin (Ig) heavy (H) chain enhancer in cooperation with E12, Ets-1, TFE3 and Oct-2. (f) Ets-1 activates the T cell receptor (TCR) enhancer in cooperation with CREB/ATF, TCF-1, AML1 and GATA-3.
20 Schematics of transcriptional regulation of hematopoietic cell differentiation Schematics of transcriptional regulation of hematopoietic cell differentiation. HAB, hemangioblast; AB, angioblast; EC, Endothelial cell; HSC, hematopoietic stem cell; CMP, common myeloid progenitor; CLP, common lymphoid progenitor; MEP,myeloid-erythroid progenitor; GMP, granulocytic-monocytic progenitor; ProB, pro-b cell; ProT, pro-t cell; Ery, erythrocyte; Meg, megakaryocyte; Neu, neutrophil;m, macrophage; B, B cell; NK, natural killer cell; T, T cell. Note the levels of PU.1, GATA-1 and other lineage-specific transcription factors are critical to determine the subsequent cell fates of hematopoietic progenitors.
21 Signalling specificity in the TGF-ß superfamily
22 Οι 9 πρωτεΐνες Smad χωρίζονται λειτουργικά σε τρεις κατηγορίες: Οι εξειδικευµένοι ενεργοποιητές µονοπατιών είναι οι Smad 2 και 3 (που µεσολαβούν στην σηµατοδότηση TGFβ/ ακτιβίνης) και οι Smad 1 και 5 (που ενεργοποιούν τη σηµατοδότηση BMP). Η Smad 4 είναι συνπαράγοντας που µπορεί να διµερίζεται µε όλες τις εξειδικευµένες Smad. Οι ανασταλτικές Smad δρουν ως ανταγωνιστικοί αναστολείς των ενεργοποιητών Smad, προσδίδοντας ακόµη ένα επίπεδο πολυπλοκότητας στο µονοπάτι. Στην υπεροικογένεια TGFβ, κάθε προσδέτης ενεργοποιεί ένα συγκεκριµένο υποδοχέα που σηµατοδοτεί µέσω ενός χαρακτηριστικού συνδυασµού Smad πρωτεϊνών. ιάφορες άλλες πρωτεΐνες προσδένονται στα διµερή των Smad και επηρεάζουν την ικανότητά τους να επιδρούν στη µεταγραφή
23 The Smad family: Diagrammatic representation of the three subfamilies of Smads
24 Smad oligomerisation Pictorial representation of the plasma membrane receptor kinases that phosphorylate the C-termini of R-Smads (light colour), leading to homo-oligomerisation (a dimer shown for simplicity)
25 Smad nucleocytoplasmic shuttling The five pathways shown are: Smad2 nuclear import after release from SARA (pathway 1); Smad3 nuclear import mediated by importin-ß1 and Ran (pathway 2); Smad4 shuttling mediated by the exportin Crm1 (pathway 3); putative Smad2 (4) and Smad3 (5) export pathways marked with question marks
26 Transcriptional regulation by Smads Two examples, one for gene induction and one for gene repression are shown. Chromatin in nucleosomal configuration is depicted by an arrow indicating promoter activation and a vertical line depicting promoter silencing. Smads are shown as heterodimers of phosphorylated (small black circle) R-Smad Smad4
27 Chromatin changes involved transcriptional regulation Chromatin changes involved in epigenetic transcriptional repression. In unmethylated nucleosomal DNA, acetylation of core histones and binding of transcription factors to DNA leads to active transcription of genes by RNA polymerase II (`transcription on'). Methylation of CpG islands in DNA near to gene promoters is associated with chromatin changes such as deacetylation and methylation of core histones, and this leads to exclusion of transcription factors. This results in loss of gene transcription (`transcription off').
28 Various aspects of the transcription process and its regulation by histone modification (A) Schematic representation of a nucleosome. Yellow represents the histones. Dark red depicts the histone tail that can be modified to loosen DNA (purple) winding. The dark red circle represents a tail without an acetyl (Ac) group. The dark red 'banana shape' represents a histone tail with an acetyl group, relieving the tight packaging of the DNA. (B) Transcriptional repression and activation in chromatin. Yellow circles represent core histone octamers; in the upper panel, acetylated histone tails (dark red) are depicted emerging from the octamer. DNA is purple, and the solid black arrow represents complex movement. Both histone acetyltransferase (HAT; activation) and HDAC (repression) require several cofactors (for DNA binding, for recruitment of the complex, for remodelling of the DNA helix to reduce the accessibility of transcription factors) for their activity Biochemical Journal (2003) Volume 370
29 Chromatin structure regulates transcriptional activity Nucleosomes consist of DNA (black line) wrapped around histone octomers (purple). Post-translational modification of histone tails by methylation (Me), phosphorylation (P) or acetylation (Ac) can alter the higher-order nucleosome structure. Nucleosome structure can be regulated by ATP-dependent chromatin remodellers (yellow cylinders), and the opposing actions of histone acetyltransferases (HATs) and histone deacetylases (HDACs). Methyl-binding proteins, such as the methyl-cpg-binding protein (MECP2), target methylated DNA (yellow) and recruit HDACs. a DNA methylation and histone deacetylation induce a closed-chromatin configuration and transcriptional repression. b Histone acetylation and demethylation of DNA relaxes chromatin, and allows transcriptional activation.
Code Breaker. TEACHER s NOTES
 TEACHER s NOTES Time: 50 minutes Learning Outcomes: To relate the genetic code to the assembly of proteins To summarize factors that lead to different types of mutations To distinguish among positive,
TEACHER s NOTES Time: 50 minutes Learning Outcomes: To relate the genetic code to the assembly of proteins To summarize factors that lead to different types of mutations To distinguish among positive,
Βιολογικές Μεμβράνες και Μεταγωγή Σήματος
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΙΩΑΝΝΙΝΩΝ ΑΝΟΙΚΤΑ ΑΚΑΔΗΜΑΪΚΑ ΜΑΘΗΜΑΤΑ Βιολογικές Μεμβράνες και Μεταγωγή Σήματος Τερματισμός σηματοδότησης Διδάσκουσα: Καθ. Μαρία - Ελένη Ε. Λέκκα Άδειες Χρήσης Το παρόν εκπαιδευτικό υλικό
ΠΑΝΕΠΙΣΤΗΜΙΟ ΙΩΑΝΝΙΝΩΝ ΑΝΟΙΚΤΑ ΑΚΑΔΗΜΑΪΚΑ ΜΑΘΗΜΑΤΑ Βιολογικές Μεμβράνες και Μεταγωγή Σήματος Τερματισμός σηματοδότησης Διδάσκουσα: Καθ. Μαρία - Ελένη Ε. Λέκκα Άδειες Χρήσης Το παρόν εκπαιδευτικό υλικό
Μελέτη της έκφρασης του ογκοκατασταλτικού γονιδίου Cyld στον καρκίνο του μαστού
 Σχολή Θετικών Επιστημών Τμήμα Βιολογίας Πρόγραμμα Μεταπτυχιακών Σπουδών Κατεύθυνση: Εφαρμοσμένη γενετική και βιοτεχνολογία ΜΕΤΑΠΤΥΧΙΑΚΗ ΔΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ Μελέτη της έκφρασης του ογκοκατασταλτικού γονιδίου
Σχολή Θετικών Επιστημών Τμήμα Βιολογίας Πρόγραμμα Μεταπτυχιακών Σπουδών Κατεύθυνση: Εφαρμοσμένη γενετική και βιοτεχνολογία ΜΕΤΑΠΤΥΧΙΑΚΗ ΔΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ Μελέτη της έκφρασης του ογκοκατασταλτικού γονιδίου
ΚΥΠΡΙΑΚΗ ΕΤΑΙΡΕΙΑ ΠΛΗΡΟΦΟΡΙΚΗΣ CYPRUS COMPUTER SOCIETY ΠΑΓΚΥΠΡΙΟΣ ΜΑΘΗΤΙΚΟΣ ΔΙΑΓΩΝΙΣΜΟΣ ΠΛΗΡΟΦΟΡΙΚΗΣ 19/5/2007
 Οδηγίες: Να απαντηθούν όλες οι ερωτήσεις. Αν κάπου κάνετε κάποιες υποθέσεις να αναφερθούν στη σχετική ερώτηση. Όλα τα αρχεία που αναφέρονται στα προβλήματα βρίσκονται στον ίδιο φάκελο με το εκτελέσιμο
Οδηγίες: Να απαντηθούν όλες οι ερωτήσεις. Αν κάπου κάνετε κάποιες υποθέσεις να αναφερθούν στη σχετική ερώτηση. Όλα τα αρχεία που αναφέρονται στα προβλήματα βρίσκονται στον ίδιο φάκελο με το εκτελέσιμο
C.S. 430 Assignment 6, Sample Solutions
 C.S. 430 Assignment 6, Sample Solutions Paul Liu November 15, 2007 Note that these are sample solutions only; in many cases there were many acceptable answers. 1 Reynolds Problem 10.1 1.1 Normal-order
C.S. 430 Assignment 6, Sample Solutions Paul Liu November 15, 2007 Note that these are sample solutions only; in many cases there were many acceptable answers. 1 Reynolds Problem 10.1 1.1 Normal-order
Fractional Colorings and Zykov Products of graphs
 Fractional Colorings and Zykov Products of graphs Who? Nichole Schimanski When? July 27, 2011 Graphs A graph, G, consists of a vertex set, V (G), and an edge set, E(G). V (G) is any finite set E(G) is
Fractional Colorings and Zykov Products of graphs Who? Nichole Schimanski When? July 27, 2011 Graphs A graph, G, consists of a vertex set, V (G), and an edge set, E(G). V (G) is any finite set E(G) is
Κυτταρική Επικοινωνία Cell Communication
 Κυτταρική Επικοινωνία Cell Communication Η μοίρα του κυττάρου ρυθμίζεται από εξωτερικά σήματα (από το περιβάλλον και άλλα κύτταρα) survival division differentiation apoptosis Είδη κυτταρικής επικοινωνίας
Κυτταρική Επικοινωνία Cell Communication Η μοίρα του κυττάρου ρυθμίζεται από εξωτερικά σήματα (από το περιβάλλον και άλλα κύτταρα) survival division differentiation apoptosis Είδη κυτταρικής επικοινωνίας
Strain gauge and rosettes
 Strain gauge and rosettes Introduction A strain gauge is a device which is used to measure strain (deformation) on an object subjected to forces. Strain can be measured using various types of devices classified
Strain gauge and rosettes Introduction A strain gauge is a device which is used to measure strain (deformation) on an object subjected to forces. Strain can be measured using various types of devices classified
Πρόβλημα 1: Αναζήτηση Ελάχιστης/Μέγιστης Τιμής
 Πρόβλημα 1: Αναζήτηση Ελάχιστης/Μέγιστης Τιμής Να γραφεί πρόγραμμα το οποίο δέχεται ως είσοδο μια ακολουθία S από n (n 40) ακέραιους αριθμούς και επιστρέφει ως έξοδο δύο ακολουθίες από θετικούς ακέραιους
Πρόβλημα 1: Αναζήτηση Ελάχιστης/Μέγιστης Τιμής Να γραφεί πρόγραμμα το οποίο δέχεται ως είσοδο μια ακολουθία S από n (n 40) ακέραιους αριθμούς και επιστρέφει ως έξοδο δύο ακολουθίες από θετικούς ακέραιους
Instruction Execution Times
 1 C Execution Times InThisAppendix... Introduction DL330 Execution Times DL330P Execution Times DL340 Execution Times C-2 Execution Times Introduction Data Registers This appendix contains several tables
1 C Execution Times InThisAppendix... Introduction DL330 Execution Times DL330P Execution Times DL340 Execution Times C-2 Execution Times Introduction Data Registers This appendix contains several tables
Main source: "Discrete-time systems and computer control" by Α. ΣΚΟΔΡΑΣ ΨΗΦΙΑΚΟΣ ΕΛΕΓΧΟΣ ΔΙΑΛΕΞΗ 4 ΔΙΑΦΑΝΕΙΑ 1
 Main source: "Discrete-time systems and computer control" by Α. ΣΚΟΔΡΑΣ ΨΗΦΙΑΚΟΣ ΕΛΕΓΧΟΣ ΔΙΑΛΕΞΗ 4 ΔΙΑΦΑΝΕΙΑ 1 A Brief History of Sampling Research 1915 - Edmund Taylor Whittaker (1873-1956) devised a
Main source: "Discrete-time systems and computer control" by Α. ΣΚΟΔΡΑΣ ΨΗΦΙΑΚΟΣ ΕΛΕΓΧΟΣ ΔΙΑΛΕΞΗ 4 ΔΙΑΦΑΝΕΙΑ 1 A Brief History of Sampling Research 1915 - Edmund Taylor Whittaker (1873-1956) devised a
13o Μεμβρανικοί υποδοχείς με εσωτερική δραστικότητα κινάσης Ser/Thr 1. Σηματοδότηση μέσω TGFβ
 13 o TGF-β Μεμβρανικοί υποδοχείς με εσωτερική δραστικότητα κινάσης Ser/Thr 1. Σηματοδότηση μέσω TGFβ Ωρίμανση του μορίου TGFβ Ενεργοποίηση των υποδοχέων TGFβ Οι μεταγραφικοί παράγοντες Smads Η ρύθμιση
13 o TGF-β Μεμβρανικοί υποδοχείς με εσωτερική δραστικότητα κινάσης Ser/Thr 1. Σηματοδότηση μέσω TGFβ Ωρίμανση του μορίου TGFβ Ενεργοποίηση των υποδοχέων TGFβ Οι μεταγραφικοί παράγοντες Smads Η ρύθμιση
Démographie spatiale/spatial Demography
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΙΑΣ Démographie spatiale/spatial Demography Session 1: Introduction to spatial demography Basic concepts Michail Agorastakis Department of Planning & Regional Development Άδειες Χρήσης
ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΙΑΣ Démographie spatiale/spatial Demography Session 1: Introduction to spatial demography Basic concepts Michail Agorastakis Department of Planning & Regional Development Άδειες Χρήσης
(1) Describe the process by which mercury atoms become excited in a fluorescent tube (3)
 Q1. (a) A fluorescent tube is filled with mercury vapour at low pressure. In order to emit electromagnetic radiation the mercury atoms must first be excited. (i) What is meant by an excited atom? (1) (ii)
Q1. (a) A fluorescent tube is filled with mercury vapour at low pressure. In order to emit electromagnetic radiation the mercury atoms must first be excited. (i) What is meant by an excited atom? (1) (ii)
Lecture 2: Dirac notation and a review of linear algebra Read Sakurai chapter 1, Baym chatper 3
 Lecture 2: Dirac notation and a review of linear algebra Read Sakurai chapter 1, Baym chatper 3 1 State vector space and the dual space Space of wavefunctions The space of wavefunctions is the set of all
Lecture 2: Dirac notation and a review of linear algebra Read Sakurai chapter 1, Baym chatper 3 1 State vector space and the dual space Space of wavefunctions The space of wavefunctions is the set of all
ΣΥΝΤΟΜΟ ΒΙΟΓΡΑΦΙΚΟ ΣΗΜΕΙΩΜΑ. Δημήτρης Tζαμαρίας
 ΑΤΟΜΙΚΑ ΣΤΟΙΧΕΙΑ ΣΥΝΤΟΜΟ ΒΙΟΓΡΑΦΙΚΟ ΣΗΜΕΙΩΜΑ Δημήτρης Τζαμαρίας Oνοματεπώνυμο: Ημ/νία και Τόπος Γέννησης: Υπηκοότητα: Οικογενειακή κατάσταση: Δημήτρης Tζαμαρίας 11 Aπριλίου 1962, Zάκυνθος Eλληνική Έγγαμος
ΑΤΟΜΙΚΑ ΣΤΟΙΧΕΙΑ ΣΥΝΤΟΜΟ ΒΙΟΓΡΑΦΙΚΟ ΣΗΜΕΙΩΜΑ Δημήτρης Τζαμαρίας Oνοματεπώνυμο: Ημ/νία και Τόπος Γέννησης: Υπηκοότητα: Οικογενειακή κατάσταση: Δημήτρης Tζαμαρίας 11 Aπριλίου 1962, Zάκυνθος Eλληνική Έγγαμος
The Simply Typed Lambda Calculus
 Type Inference Instead of writing type annotations, can we use an algorithm to infer what the type annotations should be? That depends on the type system. For simple type systems the answer is yes, and
Type Inference Instead of writing type annotations, can we use an algorithm to infer what the type annotations should be? That depends on the type system. For simple type systems the answer is yes, and
Solutions to the Schrodinger equation atomic orbitals. Ψ 1 s Ψ 2 s Ψ 2 px Ψ 2 py Ψ 2 pz
 Solutions to the Schrodinger equation atomic orbitals Ψ 1 s Ψ 2 s Ψ 2 px Ψ 2 py Ψ 2 pz ybridization Valence Bond Approach to bonding sp 3 (Ψ 2 s + Ψ 2 px + Ψ 2 py + Ψ 2 pz) sp 2 (Ψ 2 s + Ψ 2 px + Ψ 2 py)
Solutions to the Schrodinger equation atomic orbitals Ψ 1 s Ψ 2 s Ψ 2 px Ψ 2 py Ψ 2 pz ybridization Valence Bond Approach to bonding sp 3 (Ψ 2 s + Ψ 2 px + Ψ 2 py + Ψ 2 pz) sp 2 (Ψ 2 s + Ψ 2 px + Ψ 2 py)
ΑΚΑ ΗΜΙΑ ΕΜΠΟΡΙΚΟΥ ΝΑΥΤΙΚΟΥ ΜΑΚΕ ΟΝΙΑΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ
 ΑΚΑ ΗΜΙΑ ΕΜΠΟΡΙΚΟΥ ΝΑΥΤΙΚΟΥ ΜΑΚΕ ΟΝΙΑΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΘΕΜΑ :ΤΥΠΟΙ ΑΕΡΟΣΥΜΠΙΕΣΤΩΝ ΚΑΙ ΤΡΟΠΟΙ ΛΕΙΤΟΥΡΓΙΑΣ ΣΠΟΥ ΑΣΤΡΙΑ: ΕΥΘΥΜΙΑ ΟΥ ΣΩΣΑΝΝΑ ΕΠΙΒΛΕΠΩΝ ΚΑΘΗΓΗΤΗΣ : ΓΟΥΛΟΠΟΥΛΟΣ ΑΘΑΝΑΣΙΟΣ 1 ΑΚΑ
ΑΚΑ ΗΜΙΑ ΕΜΠΟΡΙΚΟΥ ΝΑΥΤΙΚΟΥ ΜΑΚΕ ΟΝΙΑΣ ΣΧΟΛΗ ΜΗΧΑΝΙΚΩΝ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΘΕΜΑ :ΤΥΠΟΙ ΑΕΡΟΣΥΜΠΙΕΣΤΩΝ ΚΑΙ ΤΡΟΠΟΙ ΛΕΙΤΟΥΡΓΙΑΣ ΣΠΟΥ ΑΣΤΡΙΑ: ΕΥΘΥΜΙΑ ΟΥ ΣΩΣΑΝΝΑ ΕΠΙΒΛΕΠΩΝ ΚΑΘΗΓΗΤΗΣ : ΓΟΥΛΟΠΟΥΛΟΣ ΑΘΑΝΑΣΙΟΣ 1 ΑΚΑ
Section 9.2 Polar Equations and Graphs
 180 Section 9. Polar Equations and Graphs In this section, we will be graphing polar equations on a polar grid. In the first few examples, we will write the polar equation in rectangular form to help identify
180 Section 9. Polar Equations and Graphs In this section, we will be graphing polar equations on a polar grid. In the first few examples, we will write the polar equation in rectangular form to help identify
Potential Dividers. 46 minutes. 46 marks. Page 1 of 11
 Potential Dividers 46 minutes 46 marks Page 1 of 11 Q1. In the circuit shown in the figure below, the battery, of negligible internal resistance, has an emf of 30 V. The pd across the lamp is 6.0 V and
Potential Dividers 46 minutes 46 marks Page 1 of 11 Q1. In the circuit shown in the figure below, the battery, of negligible internal resistance, has an emf of 30 V. The pd across the lamp is 6.0 V and
EE512: Error Control Coding
 EE512: Error Control Coding Solution for Assignment on Finite Fields February 16, 2007 1. (a) Addition and Multiplication tables for GF (5) and GF (7) are shown in Tables 1 and 2. + 0 1 2 3 4 0 0 1 2 3
EE512: Error Control Coding Solution for Assignment on Finite Fields February 16, 2007 1. (a) Addition and Multiplication tables for GF (5) and GF (7) are shown in Tables 1 and 2. + 0 1 2 3 4 0 0 1 2 3
ΚΥΠΡΙΑΚΟΣ ΣΥΝΔΕΣΜΟΣ ΠΛΗΡΟΦΟΡΙΚΗΣ CYPRUS COMPUTER SOCIETY 21 ος ΠΑΓΚΥΠΡΙΟΣ ΜΑΘΗΤΙΚΟΣ ΔΙΑΓΩΝΙΣΜΟΣ ΠΛΗΡΟΦΟΡΙΚΗΣ Δεύτερος Γύρος - 30 Μαρτίου 2011
 Διάρκεια Διαγωνισμού: 3 ώρες Απαντήστε όλες τις ερωτήσεις Μέγιστο Βάρος (20 Μονάδες) Δίνεται ένα σύνολο από N σφαιρίδια τα οποία δεν έχουν όλα το ίδιο βάρος μεταξύ τους και ένα κουτί που αντέχει μέχρι
Διάρκεια Διαγωνισμού: 3 ώρες Απαντήστε όλες τις ερωτήσεις Μέγιστο Βάρος (20 Μονάδες) Δίνεται ένα σύνολο από N σφαιρίδια τα οποία δεν έχουν όλα το ίδιο βάρος μεταξύ τους και ένα κουτί που αντέχει μέχρι
ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΙΣ «ΚΛΙΝΙΚΕΣ ΚΑΙ ΚΛΙΝΙΚΟΕΡΓΑΣΤΗΡΙΑΚΕΣ ΙΑΤΡΙΚΕΣ ΕΙΔΙΚΟΤΗΤΕΣ»
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ ΥΓΕΙΑΣ ΤΜΗΜΑ ΙΑΤΡΙΚΗΣ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΙΣ «ΚΛΙΝΙΚΕΣ ΚΑΙ ΚΛΙΝΙΚΟΕΡΓΑΣΤΗΡΙΑΚΕΣ ΙΑΤΡΙΚΕΣ ΕΙΔΙΚΟΤΗΤΕΣ» ΑΡΝΗΤΙΚΗ ΡΥΘΜΙΣΗ ΤΗΣ ΜΕΤΑΒΙΒΑΣΗΣ ΤΟΥ ΣΗΜΑΤΟΣ ΤΗΣ
ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ ΥΓΕΙΑΣ ΤΜΗΜΑ ΙΑΤΡΙΚΗΣ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΙΣ «ΚΛΙΝΙΚΕΣ ΚΑΙ ΚΛΙΝΙΚΟΕΡΓΑΣΤΗΡΙΑΚΕΣ ΙΑΤΡΙΚΕΣ ΕΙΔΙΚΟΤΗΤΕΣ» ΑΡΝΗΤΙΚΗ ΡΥΘΜΙΣΗ ΤΗΣ ΜΕΤΑΒΙΒΑΣΗΣ ΤΟΥ ΣΗΜΑΤΟΣ ΤΗΣ
TMA4115 Matematikk 3
 TMA4115 Matematikk 3 Andrew Stacey Norges Teknisk-Naturvitenskapelige Universitet Trondheim Spring 2010 Lecture 12: Mathematics Marvellous Matrices Andrew Stacey Norges Teknisk-Naturvitenskapelige Universitet
TMA4115 Matematikk 3 Andrew Stacey Norges Teknisk-Naturvitenskapelige Universitet Trondheim Spring 2010 Lecture 12: Mathematics Marvellous Matrices Andrew Stacey Norges Teknisk-Naturvitenskapelige Universitet
CHAPTER 48 APPLICATIONS OF MATRICES AND DETERMINANTS
 CHAPTER 48 APPLICATIONS OF MATRICES AND DETERMINANTS EXERCISE 01 Page 545 1. Use matrices to solve: 3x + 4y x + 5y + 7 3x + 4y x + 5y 7 Hence, 3 4 x 0 5 y 7 The inverse of 3 4 5 is: 1 5 4 1 5 4 15 8 3
CHAPTER 48 APPLICATIONS OF MATRICES AND DETERMINANTS EXERCISE 01 Page 545 1. Use matrices to solve: 3x + 4y x + 5y + 7 3x + 4y x + 5y 7 Hence, 3 4 x 0 5 y 7 The inverse of 3 4 5 is: 1 5 4 1 5 4 15 8 3
Derivation of Optical-Bloch Equations
 Appendix C Derivation of Optical-Bloch Equations In this appendix the optical-bloch equations that give the populations and coherences for an idealized three-level Λ system, Fig. 3. on page 47, will be
Appendix C Derivation of Optical-Bloch Equations In this appendix the optical-bloch equations that give the populations and coherences for an idealized three-level Λ system, Fig. 3. on page 47, will be
High mobility group 1 HMG1
 Vol. 29, pp.705 ~ 711, 2001 High mobility group 1 HMG1 13 12 20 anti-neutrophil cytoplasmic antibodies, ANCA ANCA 1982 Davies 1980 1 high mobility group HMG1 HMG2 30 kd high mobility group HMGHMG HMG1
Vol. 29, pp.705 ~ 711, 2001 High mobility group 1 HMG1 13 12 20 anti-neutrophil cytoplasmic antibodies, ANCA ANCA 1982 Davies 1980 1 high mobility group HMG1 HMG2 30 kd high mobility group HMGHMG HMG1
Mean bond enthalpy Standard enthalpy of formation Bond N H N N N N H O O O
 Q1. (a) Explain the meaning of the terms mean bond enthalpy and standard enthalpy of formation. Mean bond enthalpy... Standard enthalpy of formation... (5) (b) Some mean bond enthalpies are given below.
Q1. (a) Explain the meaning of the terms mean bond enthalpy and standard enthalpy of formation. Mean bond enthalpy... Standard enthalpy of formation... (5) (b) Some mean bond enthalpies are given below.
3.4 SUM AND DIFFERENCE FORMULAS. NOTE: cos(α+β) cos α + cos β cos(α-β) cos α -cos β
 3.4 SUM AND DIFFERENCE FORMULAS Page Theorem cos(αβ cos α cos β -sin α cos(α-β cos α cos β sin α NOTE: cos(αβ cos α cos β cos(α-β cos α -cos β Proof of cos(α-β cos α cos β sin α Let s use a unit circle
3.4 SUM AND DIFFERENCE FORMULAS Page Theorem cos(αβ cos α cos β -sin α cos(α-β cos α cos β sin α NOTE: cos(αβ cos α cos β cos(α-β cos α -cos β Proof of cos(α-β cos α cos β sin α Let s use a unit circle
CHAPTER 25 SOLVING EQUATIONS BY ITERATIVE METHODS
 CHAPTER 5 SOLVING EQUATIONS BY ITERATIVE METHODS EXERCISE 104 Page 8 1. Find the positive root of the equation x + 3x 5 = 0, correct to 3 significant figures, using the method of bisection. Let f(x) =
CHAPTER 5 SOLVING EQUATIONS BY ITERATIVE METHODS EXERCISE 104 Page 8 1. Find the positive root of the equation x + 3x 5 = 0, correct to 3 significant figures, using the method of bisection. Let f(x) =
Inverse trigonometric functions & General Solution of Trigonometric Equations. ------------------ ----------------------------- -----------------
 Inverse trigonometric functions & General Solution of Trigonometric Equations. 1. Sin ( ) = a) b) c) d) Ans b. Solution : Method 1. Ans a: 17 > 1 a) is rejected. w.k.t Sin ( sin ) = d is rejected. If sin
Inverse trigonometric functions & General Solution of Trigonometric Equations. 1. Sin ( ) = a) b) c) d) Ans b. Solution : Method 1. Ans a: 17 > 1 a) is rejected. w.k.t Sin ( sin ) = d is rejected. If sin
PARTIAL NOTES for 6.1 Trigonometric Identities
 PARTIAL NOTES for 6.1 Trigonometric Identities tanθ = sinθ cosθ cotθ = cosθ sinθ BASIC IDENTITIES cscθ = 1 sinθ secθ = 1 cosθ cotθ = 1 tanθ PYTHAGOREAN IDENTITIES sin θ + cos θ =1 tan θ +1= sec θ 1 + cot
PARTIAL NOTES for 6.1 Trigonometric Identities tanθ = sinθ cosθ cotθ = cosθ sinθ BASIC IDENTITIES cscθ = 1 sinθ secθ = 1 cosθ cotθ = 1 tanθ PYTHAGOREAN IDENTITIES sin θ + cos θ =1 tan θ +1= sec θ 1 + cot
EPL 603 TOPICS IN SOFTWARE ENGINEERING. Lab 5: Component Adaptation Environment (COPE)
 EPL 603 TOPICS IN SOFTWARE ENGINEERING Lab 5: Component Adaptation Environment (COPE) Performing Static Analysis 1 Class Name: The fully qualified name of the specific class Type: The type of the class
EPL 603 TOPICS IN SOFTWARE ENGINEERING Lab 5: Component Adaptation Environment (COPE) Performing Static Analysis 1 Class Name: The fully qualified name of the specific class Type: The type of the class
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟΥ ΚΥΠΡΟΥ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ ΥΓΕΙΑΣ. Πτυχιακή εργασία
 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟΥ ΚΥΠΡΟΥ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ ΥΓΕΙΑΣ Πτυχιακή εργασία Η ΨΥΧΟΚΟΙΝΩΝΙΚΗ ΥΓΕΙΑ ΤΟΥ ΠΑΙΔΙΟΥ ΣΕ ΣΧΕΣΗ ΜΕ ΤΗΝ ΚΟΙΝΩΝΙΚΟΟΙΚΟΝΟΜΙΚΗ ΚΑΤΑΣΤΑΣΗ ΤΗΣ ΟΙΚΟΓΕΝΕΙΑΣ Μαρία Χρίστου Λεμεσός 2012 ΤΕΧΝΟΛΟΓΙΚΟ
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟΥ ΚΥΠΡΟΥ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ ΥΓΕΙΑΣ Πτυχιακή εργασία Η ΨΥΧΟΚΟΙΝΩΝΙΚΗ ΥΓΕΙΑ ΤΟΥ ΠΑΙΔΙΟΥ ΣΕ ΣΧΕΣΗ ΜΕ ΤΗΝ ΚΟΙΝΩΝΙΚΟΟΙΚΟΝΟΜΙΚΗ ΚΑΤΑΣΤΑΣΗ ΤΗΣ ΟΙΚΟΓΕΝΕΙΑΣ Μαρία Χρίστου Λεμεσός 2012 ΤΕΧΝΟΛΟΓΙΚΟ
SUPPLEMENTARY INFORMATION
 Sensitivity of [Ru(phen) 2 dppz] 2+ Light Switch Emission to Ionic Strength, Temperature, and DNA Sequence and Conformation Andrew W. McKinley, Per Lincoln and Eimer M. Tuite* SUPPLEMENTARY INFORMATION
Sensitivity of [Ru(phen) 2 dppz] 2+ Light Switch Emission to Ionic Strength, Temperature, and DNA Sequence and Conformation Andrew W. McKinley, Per Lincoln and Eimer M. Tuite* SUPPLEMENTARY INFORMATION
ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ
 ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΣΧΟΛΗ ΑΓΡΟΝΟΜΩΝ & ΤΟΠΟΓΡΑΦΩΝ ΜΗΧΑΝΙΚΩΝ ΤΟΜΕΑΣ ΓΕΩΓΡΑΦΙΑΣ ΚΑΙ ΠΕΡΙΦΕΡΕΙΑΚΟΥ ΣΧΕΔΙΑΣΜΟΥ ΔΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ «Η ΕΞΕΛΙΞΗ ΤΗΣ ΠΟΛΗΣ ΤΗΣ ΚΑΛΑΜΑΤΑΣ ΜΕ ΕΠΙΚΕΝΤΡΟ ΤΟ ΙΣΤΟΡΙΚΟ ΤΗΣ ΚΕΝΤΡΟ»
ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΣΧΟΛΗ ΑΓΡΟΝΟΜΩΝ & ΤΟΠΟΓΡΑΦΩΝ ΜΗΧΑΝΙΚΩΝ ΤΟΜΕΑΣ ΓΕΩΓΡΑΦΙΑΣ ΚΑΙ ΠΕΡΙΦΕΡΕΙΑΚΟΥ ΣΧΕΔΙΑΣΜΟΥ ΔΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ «Η ΕΞΕΛΙΞΗ ΤΗΣ ΠΟΛΗΣ ΤΗΣ ΚΑΛΑΜΑΤΑΣ ΜΕ ΕΠΙΚΕΝΤΡΟ ΤΟ ΙΣΤΟΡΙΚΟ ΤΗΣ ΚΕΝΤΡΟ»
Chapter 6: Systems of Linear Differential. be continuous functions on the interval
 Chapter 6: Systems of Linear Differential Equations Let a (t), a 2 (t),..., a nn (t), b (t), b 2 (t),..., b n (t) be continuous functions on the interval I. The system of n first-order differential equations
Chapter 6: Systems of Linear Differential Equations Let a (t), a 2 (t),..., a nn (t), b (t), b 2 (t),..., b n (t) be continuous functions on the interval I. The system of n first-order differential equations
Exercises 10. Find a fundamental matrix of the given system of equations. Also find the fundamental matrix Φ(t) satisfying Φ(0) = I. 1.
 Exercises 0 More exercises are available in Elementary Differential Equations. If you have a problem to solve any of them, feel free to come to office hour. Problem Find a fundamental matrix of the given
Exercises 0 More exercises are available in Elementary Differential Equations. If you have a problem to solve any of them, feel free to come to office hour. Problem Find a fundamental matrix of the given
Phys460.nb Solution for the t-dependent Schrodinger s equation How did we find the solution? (not required)
 Phys460.nb 81 ψ n (t) is still the (same) eigenstate of H But for tdependent H. The answer is NO. 5.5.5. Solution for the tdependent Schrodinger s equation If we assume that at time t 0, the electron starts
Phys460.nb 81 ψ n (t) is still the (same) eigenstate of H But for tdependent H. The answer is NO. 5.5.5. Solution for the tdependent Schrodinger s equation If we assume that at time t 0, the electron starts
6.1. Dirac Equation. Hamiltonian. Dirac Eq.
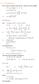 6.1. Dirac Equation Ref: M.Kaku, Quantum Field Theory, Oxford Univ Press (1993) η μν = η μν = diag(1, -1, -1, -1) p 0 = p 0 p = p i = -p i p μ p μ = p 0 p 0 + p i p i = E c 2 - p 2 = (m c) 2 H = c p 2
6.1. Dirac Equation Ref: M.Kaku, Quantum Field Theory, Oxford Univ Press (1993) η μν = η μν = diag(1, -1, -1, -1) p 0 = p 0 p = p i = -p i p μ p μ = p 0 p 0 + p i p i = E c 2 - p 2 = (m c) 2 H = c p 2
Matrices and Determinants
 Matrices and Determinants SUBJECTIVE PROBLEMS: Q 1. For what value of k do the following system of equations possess a non-trivial (i.e., not all zero) solution over the set of rationals Q? x + ky + 3z
Matrices and Determinants SUBJECTIVE PROBLEMS: Q 1. For what value of k do the following system of equations possess a non-trivial (i.e., not all zero) solution over the set of rationals Q? x + ky + 3z
Math 6 SL Probability Distributions Practice Test Mark Scheme
 Math 6 SL Probability Distributions Practice Test Mark Scheme. (a) Note: Award A for vertical line to right of mean, A for shading to right of their vertical line. AA N (b) evidence of recognizing symmetry
Math 6 SL Probability Distributions Practice Test Mark Scheme. (a) Note: Award A for vertical line to right of mean, A for shading to right of their vertical line. AA N (b) evidence of recognizing symmetry
ΚΥΠΡΙΑΚΗ ΕΤΑΙΡΕΙΑ ΠΛΗΡΟΦΟΡΙΚΗΣ CYPRUS COMPUTER SOCIETY ΠΑΓΚΥΠΡΙΟΣ ΜΑΘΗΤΙΚΟΣ ΔΙΑΓΩΝΙΣΜΟΣ ΠΛΗΡΟΦΟΡΙΚΗΣ 6/5/2006
 Οδηγίες: Να απαντηθούν όλες οι ερωτήσεις. Ολοι οι αριθμοί που αναφέρονται σε όλα τα ερωτήματα είναι μικρότεροι το 1000 εκτός αν ορίζεται διαφορετικά στη διατύπωση του προβλήματος. Διάρκεια: 3,5 ώρες Καλή
Οδηγίες: Να απαντηθούν όλες οι ερωτήσεις. Ολοι οι αριθμοί που αναφέρονται σε όλα τα ερωτήματα είναι μικρότεροι το 1000 εκτός αν ορίζεται διαφορετικά στη διατύπωση του προβλήματος. Διάρκεια: 3,5 ώρες Καλή
«ΑΓΡΟΤΟΥΡΙΣΜΟΣ ΚΑΙ ΤΟΠΙΚΗ ΑΝΑΠΤΥΞΗ: Ο ΡΟΛΟΣ ΤΩΝ ΝΕΩΝ ΤΕΧΝΟΛΟΓΙΩΝ ΣΤΗΝ ΠΡΟΩΘΗΣΗ ΤΩΝ ΓΥΝΑΙΚΕΙΩΝ ΣΥΝΕΤΑΙΡΙΣΜΩΝ»
 I ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΣΧΟΛΗ ΝΟΜΙΚΩΝ ΟΙΚΟΝΟΜΙΚΩΝ ΚΑΙ ΠΟΛΙΤΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΤΜΗΜΑ ΟΙΚΟΝΟΜΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΗΝ «ΔΙΟΙΚΗΣΗ ΚΑΙ ΟΙΚΟΝΟΜΙΑ» ΚΑΤΕΥΘΥΝΣΗ: ΟΙΚΟΝΟΜΙΚΗ
I ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΣΧΟΛΗ ΝΟΜΙΚΩΝ ΟΙΚΟΝΟΜΙΚΩΝ ΚΑΙ ΠΟΛΙΤΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΤΜΗΜΑ ΟΙΚΟΝΟΜΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΗΝ «ΔΙΟΙΚΗΣΗ ΚΑΙ ΟΙΚΟΝΟΜΙΑ» ΚΑΤΕΥΘΥΝΣΗ: ΟΙΚΟΝΟΜΙΚΗ
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΤΜΗΜΑ ΝΟΣΗΛΕΥΤΙΚΗΣ
 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΤΜΗΜΑ ΝΟΣΗΛΕΥΤΙΚΗΣ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΨΥΧΟΛΟΓΙΚΕΣ ΕΠΙΠΤΩΣΕΙΣ ΣΕ ΓΥΝΑΙΚΕΣ ΜΕΤΑ ΑΠΟ ΜΑΣΤΕΚΤΟΜΗ ΓΕΩΡΓΙΑ ΤΡΙΣΟΚΚΑ Λευκωσία 2012 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΤΜΗΜΑ ΝΟΣΗΛΕΥΤΙΚΗΣ ΠΤΥΧΙΑΚΗ ΕΡΓΑΣΙΑ ΨΥΧΟΛΟΓΙΚΕΣ ΕΠΙΠΤΩΣΕΙΣ ΣΕ ΓΥΝΑΙΚΕΣ ΜΕΤΑ ΑΠΟ ΜΑΣΤΕΚΤΟΜΗ ΓΕΩΡΓΙΑ ΤΡΙΣΟΚΚΑ Λευκωσία 2012 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ
SCHOOL OF MATHEMATICAL SCIENCES G11LMA Linear Mathematics Examination Solutions
 SCHOOL OF MATHEMATICAL SCIENCES GLMA Linear Mathematics 00- Examination Solutions. (a) i. ( + 5i)( i) = (6 + 5) + (5 )i = + i. Real part is, imaginary part is. (b) ii. + 5i i ( + 5i)( + i) = ( i)( + i)
SCHOOL OF MATHEMATICAL SCIENCES GLMA Linear Mathematics 00- Examination Solutions. (a) i. ( + 5i)( i) = (6 + 5) + (5 )i = + i. Real part is, imaginary part is. (b) ii. + 5i i ( + 5i)( + i) = ( i)( + i)
derivation of the Laplacian from rectangular to spherical coordinates
 derivation of the Laplacian from rectangular to spherical coordinates swapnizzle 03-03- :5:43 We begin by recognizing the familiar conversion from rectangular to spherical coordinates (note that φ is used
derivation of the Laplacian from rectangular to spherical coordinates swapnizzle 03-03- :5:43 We begin by recognizing the familiar conversion from rectangular to spherical coordinates (note that φ is used
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΜΗΧΑΝΙΚΗΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ. Πτυχιακή εργασία ΟΛΙΣΘΗΡΟΤΗΤΑ ΚΑΙ ΜΑΚΡΟΥΦΗ ΤΩΝ ΟΔΟΔΤΡΩΜΑΤΩΝ ΚΥΚΛΟΦΟΡΙΑΣ
 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΜΗΧΑΝΙΚΗΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ Πτυχιακή εργασία ΟΛΙΣΘΗΡΟΤΗΤΑ ΚΑΙ ΜΑΚΡΟΥΦΗ ΤΩΝ ΟΔΟΔΤΡΩΜΑΤΩΝ ΚΥΚΛΟΦΟΡΙΑΣ Χριστοδούλου Αντρέας Λεμεσός 2014 2 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΜΗΧΑΝΙΚΗΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ Πτυχιακή εργασία ΟΛΙΣΘΗΡΟΤΗΤΑ ΚΑΙ ΜΑΚΡΟΥΦΗ ΤΩΝ ΟΔΟΔΤΡΩΜΑΤΩΝ ΚΥΚΛΟΦΟΡΙΑΣ Χριστοδούλου Αντρέας Λεμεσός 2014 2 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ
[1] P Q. Fig. 3.1
![[1] P Q. Fig. 3.1 [1] P Q. Fig. 3.1](/thumbs/79/80362156.jpg) 1 (a) Define resistance....... [1] (b) The smallest conductor within a computer processing chip can be represented as a rectangular block that is one atom high, four atoms wide and twenty atoms long. One
1 (a) Define resistance....... [1] (b) The smallest conductor within a computer processing chip can be represented as a rectangular block that is one atom high, four atoms wide and twenty atoms long. One
ΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ. ΘΕΜΑ: «ιερεύνηση της σχέσης µεταξύ φωνηµικής επίγνωσης και ορθογραφικής δεξιότητας σε παιδιά προσχολικής ηλικίας»
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΑΙΓΑΙΟΥ ΣΧΟΛΗ ΑΝΘΡΩΠΙΣΤΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΤΜΗΜΑ ΕΠΙΣΤΗΜΩΝ ΤΗΣ ΠΡΟΣΧΟΛΙΚΗΣ ΑΓΩΓΗΣ ΚΑΙ ΤΟΥ ΕΚΠΑΙ ΕΥΤΙΚΟΥ ΣΧΕ ΙΑΣΜΟΥ «ΠΑΙ ΙΚΟ ΒΙΒΛΙΟ ΚΑΙ ΠΑΙ ΑΓΩΓΙΚΟ ΥΛΙΚΟ» ΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ που εκπονήθηκε για τη
ΠΑΝΕΠΙΣΤΗΜΙΟ ΑΙΓΑΙΟΥ ΣΧΟΛΗ ΑΝΘΡΩΠΙΣΤΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΤΜΗΜΑ ΕΠΙΣΤΗΜΩΝ ΤΗΣ ΠΡΟΣΧΟΛΙΚΗΣ ΑΓΩΓΗΣ ΚΑΙ ΤΟΥ ΕΚΠΑΙ ΕΥΤΙΚΟΥ ΣΧΕ ΙΑΣΜΟΥ «ΠΑΙ ΙΚΟ ΒΙΒΛΙΟ ΚΑΙ ΠΑΙ ΑΓΩΓΙΚΟ ΥΛΙΚΟ» ΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ που εκπονήθηκε για τη
Every set of first-order formulas is equivalent to an independent set
 Every set of first-order formulas is equivalent to an independent set May 6, 2008 Abstract A set of first-order formulas, whatever the cardinality of the set of symbols, is equivalent to an independent
Every set of first-order formulas is equivalent to an independent set May 6, 2008 Abstract A set of first-order formulas, whatever the cardinality of the set of symbols, is equivalent to an independent
ΑΝΩΤΑΤΗ ΣΧΟΛΗ ΠΑΙ ΑΓΩΓΙΚΗΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΚΗΣ ΕΚΠΑΙ ΕΥΣΗΣ ΠΑΡΑΔΟΤΕΟ ΕΠΙΣΤΗΜΟΝΙΚΗ ΕΡΓΑΣΙΑ ΣΕ ΔΙΕΘΝΕΣ ΕΠΙΣΤΗΜΟΝΙΚΟ ΠΕΡΙΟΔΙΚΟ
 ΑΝΩΤΑΤΗ ΣΧΟΛΗ ΠΑΙ ΑΓΩΓΙΚΗΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΚΗΣ ΕΚΠΑΙ ΕΥΣΗΣ (Α.Σ.ΠΑΙ.Τ.Ε.) «Αρχιμήδης ΙΙΙ Ενίσχυση Ερευνητικών ομάδων στην Α.Σ.ΠΑΙ.Τ.Ε.» Υποέργο: 8 Τίτλος: «Εκκεντρότητες αντισεισμικού σχεδιασμού ασύμμετρων
ΑΝΩΤΑΤΗ ΣΧΟΛΗ ΠΑΙ ΑΓΩΓΙΚΗΣ ΚΑΙ ΤΕΧΝΟΛΟΓΙΚΗΣ ΕΚΠΑΙ ΕΥΣΗΣ (Α.Σ.ΠΑΙ.Τ.Ε.) «Αρχιμήδης ΙΙΙ Ενίσχυση Ερευνητικών ομάδων στην Α.Σ.ΠΑΙ.Τ.Ε.» Υποέργο: 8 Τίτλος: «Εκκεντρότητες αντισεισμικού σχεδιασμού ασύμμετρων
Inferring regulatory subnetworks through the analysis of genome-wide expression profiles
 NATIONAL AND KAPODISTRIAN UNIVERSITY OF ATHENS SCHOOL OF SCIENCE DEPARTMENT OF INFORMATICS AND TELECOMMUNICATIONS POSTGRADUATE PROGRAM "INFORMATION TECHNOLOGIES IN MEDICINE AND BIOLOGY" MASTER S THESIS
NATIONAL AND KAPODISTRIAN UNIVERSITY OF ATHENS SCHOOL OF SCIENCE DEPARTMENT OF INFORMATICS AND TELECOMMUNICATIONS POSTGRADUATE PROGRAM "INFORMATION TECHNOLOGIES IN MEDICINE AND BIOLOGY" MASTER S THESIS
Mouse Gene 2.0 ST Array Shn3. Runx2 Shn3. Runx2 Shn3 Runx2. adult. (Schnurri, Shn)3/Hivep3. Runx2 Shn/Hivep. ST2 BMP-2 Shn3
 adult (Schnurri, Shn)3/Hivep3 Runx2 Shn/Hivep Shn3 Shn3 Shn3 Shn3 Shn1 Shn2 Shn3 gain-of-function loss-of-function Shn3 in vivo mimic MC3T3-E1 ST-2 ATDC5 C3H10T1/2 ALP RT-PCR alcian blue RT-PCR Affymetrix
adult (Schnurri, Shn)3/Hivep3 Runx2 Shn/Hivep Shn3 Shn3 Shn3 Shn3 Shn1 Shn2 Shn3 gain-of-function loss-of-function Shn3 in vivo mimic MC3T3-E1 ST-2 ATDC5 C3H10T1/2 ALP RT-PCR alcian blue RT-PCR Affymetrix
Second Order RLC Filters
 ECEN 60 Circuits/Electronics Spring 007-0-07 P. Mathys Second Order RLC Filters RLC Lowpass Filter A passive RLC lowpass filter (LPF) circuit is shown in the following schematic. R L C v O (t) Using phasor
ECEN 60 Circuits/Electronics Spring 007-0-07 P. Mathys Second Order RLC Filters RLC Lowpass Filter A passive RLC lowpass filter (LPF) circuit is shown in the following schematic. R L C v O (t) Using phasor
Other Test Constructions: Likelihood Ratio & Bayes Tests
 Other Test Constructions: Likelihood Ratio & Bayes Tests Side-Note: So far we have seen a few approaches for creating tests such as Neyman-Pearson Lemma ( most powerful tests of H 0 : θ = θ 0 vs H 1 :
Other Test Constructions: Likelihood Ratio & Bayes Tests Side-Note: So far we have seen a few approaches for creating tests such as Neyman-Pearson Lemma ( most powerful tests of H 0 : θ = θ 0 vs H 1 :
Απόκριση σε Μοναδιαία Ωστική Δύναμη (Unit Impulse) Απόκριση σε Δυνάμεις Αυθαίρετα Μεταβαλλόμενες με το Χρόνο. Απόστολος Σ.
 Απόκριση σε Δυνάμεις Αυθαίρετα Μεταβαλλόμενες με το Χρόνο The time integral of a force is referred to as impulse, is determined by and is obtained from: Newton s 2 nd Law of motion states that the action
Απόκριση σε Δυνάμεις Αυθαίρετα Μεταβαλλόμενες με το Χρόνο The time integral of a force is referred to as impulse, is determined by and is obtained from: Newton s 2 nd Law of motion states that the action
b. Use the parametrization from (a) to compute the area of S a as S a ds. Be sure to substitute for ds!
 MTH U341 urface Integrals, tokes theorem, the divergence theorem To be turned in Wed., Dec. 1. 1. Let be the sphere of radius a, x 2 + y 2 + z 2 a 2. a. Use spherical coordinates (with ρ a) to parametrize.
MTH U341 urface Integrals, tokes theorem, the divergence theorem To be turned in Wed., Dec. 1. 1. Let be the sphere of radius a, x 2 + y 2 + z 2 a 2. a. Use spherical coordinates (with ρ a) to parametrize.
Assalamu `alaikum wr. wb.
 LUMP SUM Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. LUMP SUM Lump sum lump sum lump sum. lump sum fixed price lump sum lump
LUMP SUM Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. Assalamu `alaikum wr. wb. LUMP SUM Wassalamu alaikum wr. wb. LUMP SUM Lump sum lump sum lump sum. lump sum fixed price lump sum lump
Splice site recognition between different organisms
 NATIONAL AND KAPODISTRIAN UNIVERSITY OF ATHENS SCHOOL OF SCIENCE DEPARTMENT OF INFORMATICS AND TELECOMMUNICATIONS INTERDEPARTMENTAL POSTGRADUATE PROGRAM "INFORMATION TECHNOLOGIES IN MEDICINE AND BIOLOGY"
NATIONAL AND KAPODISTRIAN UNIVERSITY OF ATHENS SCHOOL OF SCIENCE DEPARTMENT OF INFORMATICS AND TELECOMMUNICATIONS INTERDEPARTMENTAL POSTGRADUATE PROGRAM "INFORMATION TECHNOLOGIES IN MEDICINE AND BIOLOGY"
Calculating the propagation delay of coaxial cable
 Your source for quality GNSS Networking Solutions and Design Services! Page 1 of 5 Calculating the propagation delay of coaxial cable The delay of a cable or velocity factor is determined by the dielectric
Your source for quality GNSS Networking Solutions and Design Services! Page 1 of 5 Calculating the propagation delay of coaxial cable The delay of a cable or velocity factor is determined by the dielectric
Αστικές παρεμβάσεις ανάπλασης αδιαμόρφωτων χώρων. Δημιουργία βιώσιμου αστικού περιβάλλοντος και σύνδεση τριών κομβικών σημείων στην πόλη της Δράμας
 ΤΕΙ ΚΑΒΑΛΑΣ ΠΑΡΑΡΤΗΜΑ ΔΡΑΜΑΣ ΤΜΗΜΑ ΑΡΧΙΤΕΚΤΟΝΙΚΗΤΟΠΙΟΥ Αστικές παρεμβάσεις ανάπλασης αδιαμόρφωτων χώρων. Δημιουργία βιώσιμου αστικού περιβάλλοντος και σύνδεση τριών κομβικών σημείων στην πόλη της Δράμας
ΤΕΙ ΚΑΒΑΛΑΣ ΠΑΡΑΡΤΗΜΑ ΔΡΑΜΑΣ ΤΜΗΜΑ ΑΡΧΙΤΕΚΤΟΝΙΚΗΤΟΠΙΟΥ Αστικές παρεμβάσεις ανάπλασης αδιαμόρφωτων χώρων. Δημιουργία βιώσιμου αστικού περιβάλλοντος και σύνδεση τριών κομβικών σημείων στην πόλη της Δράμας
Ordinal Arithmetic: Addition, Multiplication, Exponentiation and Limit
 Ordinal Arithmetic: Addition, Multiplication, Exponentiation and Limit Ting Zhang Stanford May 11, 2001 Stanford, 5/11/2001 1 Outline Ordinal Classification Ordinal Addition Ordinal Multiplication Ordinal
Ordinal Arithmetic: Addition, Multiplication, Exponentiation and Limit Ting Zhang Stanford May 11, 2001 Stanford, 5/11/2001 1 Outline Ordinal Classification Ordinal Addition Ordinal Multiplication Ordinal
Κυτταρική Επικοινωνία Cell Communication
 Κυτταρική Επικοινωνία Cell Communication Κυτταρική Επικοινωνία Είναι η ικανότητα του κυττάρου να αποκρίνεται σε εξωτερικά σήματα (σήματα από το περιβάλλον του ή άλλα κύτταρα) Τα σήματα αυτά μπορεί να είναι
Κυτταρική Επικοινωνία Cell Communication Κυτταρική Επικοινωνία Είναι η ικανότητα του κυττάρου να αποκρίνεται σε εξωτερικά σήματα (σήματα από το περιβάλλον του ή άλλα κύτταρα) Τα σήματα αυτά μπορεί να είναι
Η ΨΥΧΙΑΤΡΙΚΗ - ΨΥΧΟΛΟΓΙΚΗ ΠΡΑΓΜΑΤΟΓΝΩΜΟΣΥΝΗ ΣΤΗΝ ΠΟΙΝΙΚΗ ΔΙΚΗ
 ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΝΟΜΙΚΗ ΣΧΟΛΗ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΤΟΜΕΑΣ ΙΣΤΟΡΙΑΣ ΦΙΛΟΣΟΦΙΑΣ ΚΑΙ ΚΟΙΝΩΝΙΟΛΟΓΙΑΣ ΤΟΥ ΔΙΚΑΙΟΥ Διπλωματική εργασία στο μάθημα «ΚΟΙΝΩΝΙΟΛΟΓΙΑ ΤΟΥ ΔΙΚΑΙΟΥ»
ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΝΟΜΙΚΗ ΣΧΟΛΗ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΤΟΜΕΑΣ ΙΣΤΟΡΙΑΣ ΦΙΛΟΣΟΦΙΑΣ ΚΑΙ ΚΟΙΝΩΝΙΟΛΟΓΙΑΣ ΤΟΥ ΔΙΚΑΙΟΥ Διπλωματική εργασία στο μάθημα «ΚΟΙΝΩΝΙΟΛΟΓΙΑ ΤΟΥ ΔΙΚΑΙΟΥ»
Καραπέτσας Θανάσης. Διπλωματική Εργασία:
 Διαπανεπιστημιακό Πρόγραμμα Μεταπτυχιακών Σπουδών «Κλινική Φαρμακολογία & Θεραπευτική» Επιβλέπων: Δρ. Αλ. Γαλάνης, Λέκτορας Μορ. Βιολογίας, «Σχεδιασμός Ειδικών Πεπτιδίων Αναστολέων της Αλληλεπίδρασης του
Διαπανεπιστημιακό Πρόγραμμα Μεταπτυχιακών Σπουδών «Κλινική Φαρμακολογία & Θεραπευτική» Επιβλέπων: Δρ. Αλ. Γαλάνης, Λέκτορας Μορ. Βιολογίας, «Σχεδιασμός Ειδικών Πεπτιδίων Αναστολέων της Αλληλεπίδρασης του
Study of In-vehicle Sound Field Creation by Simultaneous Equation Method
 Study of In-vehicle Sound Field Creation by Simultaneous Equation Method Kensaku FUJII Isao WAKABAYASI Tadashi UJINO Shigeki KATO Abstract FUJITSU TEN Limited has developed "TOYOTA remium Sound System"
Study of In-vehicle Sound Field Creation by Simultaneous Equation Method Kensaku FUJII Isao WAKABAYASI Tadashi UJINO Shigeki KATO Abstract FUJITSU TEN Limited has developed "TOYOTA remium Sound System"
Space-Time Symmetries
 Chapter Space-Time Symmetries In classical fiel theory any continuous symmetry of the action generates a conserve current by Noether's proceure. If the Lagrangian is not invariant but only shifts by a
Chapter Space-Time Symmetries In classical fiel theory any continuous symmetry of the action generates a conserve current by Noether's proceure. If the Lagrangian is not invariant but only shifts by a
ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΤΜΗΜΑ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ ΥΠΟΛΟΓΙΣΤΩΝ ΤΟΜΕΑΣ ΣΥΣΤΗΜΑΤΩΝ ΗΛΕΚΤΡΙΚΗΣ ΕΝΕΡΓΕΙΑΣ
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΤΜΗΜΑ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ ΥΠΟΛΟΓΙΣΤΩΝ ΤΟΜΕΑΣ ΣΥΣΤΗΜΑΤΩΝ ΗΛΕΚΤΡΙΚΗΣ ΕΝΕΡΓΕΙΑΣ Διπλωματική Εργασία του φοιτητή του τμήματος Ηλεκτρολόγων Μηχανικών και Τεχνολογίας Ηλεκτρονικών
ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΤΜΗΜΑ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΤΕΧΝΟΛΟΓΙΑΣ ΥΠΟΛΟΓΙΣΤΩΝ ΤΟΜΕΑΣ ΣΥΣΤΗΜΑΤΩΝ ΗΛΕΚΤΡΙΚΗΣ ΕΝΕΡΓΕΙΑΣ Διπλωματική Εργασία του φοιτητή του τμήματος Ηλεκτρολόγων Μηχανικών και Τεχνολογίας Ηλεκτρονικών
Homework 8 Model Solution Section
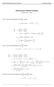 MATH 004 Homework Solution Homework 8 Model Solution Section 14.5 14.6. 14.5. Use the Chain Rule to find dz where z cosx + 4y), x 5t 4, y 1 t. dz dx + dy y sinx + 4y)0t + 4) sinx + 4y) 1t ) 0t + 4t ) sinx
MATH 004 Homework Solution Homework 8 Model Solution Section 14.5 14.6. 14.5. Use the Chain Rule to find dz where z cosx + 4y), x 5t 4, y 1 t. dz dx + dy y sinx + 4y)0t + 4) sinx + 4y) 1t ) 0t + 4t ) sinx
Table S1. All regions of WCC enrichment at p<0.001 and z score >3.09
 Table S. All regions of WCC enrichment at p3.9 N. crassa contig start nt # of reads z score promoter coding region intergenic chrvii_contig7. 776 738 5.66 frq chrvi_contig7. 696.93 vvd
Table S. All regions of WCC enrichment at p3.9 N. crassa contig start nt # of reads z score promoter coding region intergenic chrvii_contig7. 776 738 5.66 frq chrvi_contig7. 696.93 vvd
Areas and Lengths in Polar Coordinates
 Kiryl Tsishchanka Areas and Lengths in Polar Coordinates In this section we develop the formula for the area of a region whose boundary is given by a polar equation. We need to use the formula for the
Kiryl Tsishchanka Areas and Lengths in Polar Coordinates In this section we develop the formula for the area of a region whose boundary is given by a polar equation. We need to use the formula for the
2 Composition. Invertible Mappings
 Arkansas Tech University MATH 4033: Elementary Modern Algebra Dr. Marcel B. Finan Composition. Invertible Mappings In this section we discuss two procedures for creating new mappings from old ones, namely,
Arkansas Tech University MATH 4033: Elementary Modern Algebra Dr. Marcel B. Finan Composition. Invertible Mappings In this section we discuss two procedures for creating new mappings from old ones, namely,
ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΤΜΗΜΑ ΟΔΟΝΤΙΑΤΡΙΚΗΣ ΕΡΓΑΣΤΗΡΙΟ ΟΔΟΝΤΙΚΗΣ ΚΑΙ ΑΝΩΤΕΡΑΣ ΠΡΟΣΘΕΤΙΚΗΣ
 ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΤΜΗΜΑ ΟΔΟΝΤΙΑΤΡΙΚΗΣ ΕΡΓΑΣΤΗΡΙΟ ΟΔΟΝΤΙΚΗΣ ΚΑΙ ΑΝΩΤΕΡΑΣ ΠΡΟΣΘΕΤΙΚΗΣ ΣΥΓΚΡΙΤΙΚΗ ΜΕΛΕΤΗ ΤΗΣ ΣΥΓΚΡΑΤΗΤΙΚΗΣ ΙΚΑΝΟΤΗΤΑΣ ΟΡΙΣΜΕΝΩΝ ΠΡΟΚΑΤΑΣΚΕΥΑΣΜΕΝΩΝ ΣΥΝΔΕΣΜΩΝ ΑΚΡΙΒΕΙΑΣ
ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΤΜΗΜΑ ΟΔΟΝΤΙΑΤΡΙΚΗΣ ΕΡΓΑΣΤΗΡΙΟ ΟΔΟΝΤΙΚΗΣ ΚΑΙ ΑΝΩΤΕΡΑΣ ΠΡΟΣΘΕΤΙΚΗΣ ΣΥΓΚΡΙΤΙΚΗ ΜΕΛΕΤΗ ΤΗΣ ΣΥΓΚΡΑΤΗΤΙΚΗΣ ΙΚΑΝΟΤΗΤΑΣ ΟΡΙΣΜΕΝΩΝ ΠΡΟΚΑΤΑΣΚΕΥΑΣΜΕΝΩΝ ΣΥΝΔΕΣΜΩΝ ΑΚΡΙΒΕΙΑΣ
Second Order Partial Differential Equations
 Chapter 7 Second Order Partial Differential Equations 7.1 Introduction A second order linear PDE in two independent variables (x, y Ω can be written as A(x, y u x + B(x, y u xy + C(x, y u u u + D(x, y
Chapter 7 Second Order Partial Differential Equations 7.1 Introduction A second order linear PDE in two independent variables (x, y Ω can be written as A(x, y u x + B(x, y u xy + C(x, y u u u + D(x, y
Η εντόπιση και οι μεταβολές της Ινχιμπίνης, Ακτιβίνης και Φολιστατίνης σε φυσιολογικούς και παθολογικούς πλακούντες με ανοσοϊστοχημική μέθοδο
 ΔΗΜΗΤΡΙΟΣ ΚΟΡΦΙΑΣ Η εντόπιση και οι μεταβολές της Ινχιμπίνης, Ακτιβίνης και Φολιστατίνης σε φυσιολογικούς και παθολογικούς πλακούντες με ανοσοϊστοχημική μέθοδο ΠΕΡΙΛΗΨΗ Η οικογένεια των αυξητικών παραγόντων
ΔΗΜΗΤΡΙΟΣ ΚΟΡΦΙΑΣ Η εντόπιση και οι μεταβολές της Ινχιμπίνης, Ακτιβίνης και Φολιστατίνης σε φυσιολογικούς και παθολογικούς πλακούντες με ανοσοϊστοχημική μέθοδο ΠΕΡΙΛΗΨΗ Η οικογένεια των αυξητικών παραγόντων
2. THEORY OF EQUATIONS. PREVIOUS EAMCET Bits.
 EAMCET-. THEORY OF EQUATIONS PREVIOUS EAMCET Bits. Each of the roots of the equation x 6x + 6x 5= are increased by k so that the new transformed equation does not contain term. Then k =... - 4. - Sol.
EAMCET-. THEORY OF EQUATIONS PREVIOUS EAMCET Bits. Each of the roots of the equation x 6x + 6x 5= are increased by k so that the new transformed equation does not contain term. Then k =... - 4. - Sol.
14 Lesson 2: The Omega Verb - Present Tense
 Lesson 2: The Omega Verb - Present Tense Day one I. Word Study and Grammar 1. Most Greek verbs end in in the first person singular. 2. The present tense is formed by adding endings to the present stem.
Lesson 2: The Omega Verb - Present Tense Day one I. Word Study and Grammar 1. Most Greek verbs end in in the first person singular. 2. The present tense is formed by adding endings to the present stem.
Long intergenic non-coding RNA expression signature in human breast cancer
 Long intergenic non-coding RNA expression signature in human breast cancer Yanfeng Zhang 1,5,#, Erin K. Wagner 2,#, Xingyi Guo 1, Isaac May 3, Qiuyin Cai 1, Wei Zheng 1, Chunyan He 2,4,*, Jirong Long 1,*
Long intergenic non-coding RNA expression signature in human breast cancer Yanfeng Zhang 1,5,#, Erin K. Wagner 2,#, Xingyi Guo 1, Isaac May 3, Qiuyin Cai 1, Wei Zheng 1, Chunyan He 2,4,*, Jirong Long 1,*
ΙΩΑΝΝΗ ΑΘ. ΠΑΠΑΪΩΑΝΝΟΥ
 ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΣΧΟΛΗ ΓΕΩΠΟΝΙΑΣ, ΔΑΣΟΛΟΓΙΑΣ ΚΑΙ ΦΥΣΙΚΟΥ ΠΕΡΙΒΑΛΛΟΝΤΟΣ ΤΜΗΜΑ ΓΕΩΠΟΝΙΑΣ ΤΟΜΕΑΣ ΑΓΡΟΤΙΚΗΣ ΟΙΚΟΝΟΜΙΑΣ ΙΩΑΝΝΗ ΑΘ. ΠΑΠΑΪΩΑΝΝΟΥ Πτυχιούχου Γεωπόνου Κατόχου Μεταπτυχιακού
ΑΡΙΣΤΟΤΕΛΕΙΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΘΕΣΣΑΛΟΝΙΚΗΣ ΣΧΟΛΗ ΓΕΩΠΟΝΙΑΣ, ΔΑΣΟΛΟΓΙΑΣ ΚΑΙ ΦΥΣΙΚΟΥ ΠΕΡΙΒΑΛΛΟΝΤΟΣ ΤΜΗΜΑ ΓΕΩΠΟΝΙΑΣ ΤΟΜΕΑΣ ΑΓΡΟΤΙΚΗΣ ΟΙΚΟΝΟΜΙΑΣ ΙΩΑΝΝΗ ΑΘ. ΠΑΠΑΪΩΑΝΝΟΥ Πτυχιούχου Γεωπόνου Κατόχου Μεταπτυχιακού
«Χρήσεις γης, αξίες γης και κυκλοφοριακές ρυθμίσεις στο Δήμο Χαλκιδέων. Η μεταξύ τους σχέση και εξέλιξη.»
 ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΣΧΟΛΗ ΑΓΡΟΝΟΜΩΝ ΚΑΙ ΤΟΠΟΓΡΑΦΩΝ ΜΗΧΑΝΙΚΩΝ ΤΟΜΕΑΣ ΓΕΩΓΡΑΦΙΑΣ ΚΑΙ ΠΕΡΙΦΕΡΕΙΑΚΟΥ ΣΧΕΔΙΑΣΜΟΥ ΔΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ: «Χρήσεις γης, αξίες γης και κυκλοφοριακές ρυθμίσεις στο Δήμο Χαλκιδέων.
ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΣΧΟΛΗ ΑΓΡΟΝΟΜΩΝ ΚΑΙ ΤΟΠΟΓΡΑΦΩΝ ΜΗΧΑΝΙΚΩΝ ΤΟΜΕΑΣ ΓΕΩΓΡΑΦΙΑΣ ΚΑΙ ΠΕΡΙΦΕΡΕΙΑΚΟΥ ΣΧΕΔΙΑΣΜΟΥ ΔΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ: «Χρήσεις γης, αξίες γης και κυκλοφοριακές ρυθμίσεις στο Δήμο Χαλκιδέων.
ΠΑΡΑΜΕΤΡΟΙ ΕΠΗΡΕΑΣΜΟΥ ΤΗΣ ΑΝΑΓΝΩΣΗΣ- ΑΠΟΚΩΔΙΚΟΠΟΙΗΣΗΣ ΤΗΣ BRAILLE ΑΠΟ ΑΤΟΜΑ ΜΕ ΤΥΦΛΩΣΗ
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΜΑΚΕΔΟΝΙΑΣ ΟΙΚΟΝΟΜΙΚΩΝ ΚΑΙ ΚΟΙΝΩΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΤΜΗΜΑ ΕΚΠΑΙΔΕΥΤΙΚΗΣ ΚΑΙ ΚΟΙΝΩΝΙΚΗΣ ΠΟΛΙΤΙΚΗΣ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΠΑΡΑΜΕΤΡΟΙ ΕΠΗΡΕΑΣΜΟΥ ΤΗΣ ΑΝΑΓΝΩΣΗΣ- ΑΠΟΚΩΔΙΚΟΠΟΙΗΣΗΣ ΤΗΣ BRAILLE
ΠΑΝΕΠΙΣΤΗΜΙΟ ΜΑΚΕΔΟΝΙΑΣ ΟΙΚΟΝΟΜΙΚΩΝ ΚΑΙ ΚΟΙΝΩΝΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΤΜΗΜΑ ΕΚΠΑΙΔΕΥΤΙΚΗΣ ΚΑΙ ΚΟΙΝΩΝΙΚΗΣ ΠΟΛΙΤΙΚΗΣ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΠΑΡΑΜΕΤΡΟΙ ΕΠΗΡΕΑΣΜΟΥ ΤΗΣ ΑΝΑΓΝΩΣΗΣ- ΑΠΟΚΩΔΙΚΟΠΟΙΗΣΗΣ ΤΗΣ BRAILLE
DESIGN OF MACHINERY SOLUTION MANUAL h in h 4 0.
 DESIGN OF MACHINERY SOLUTION MANUAL -7-1! PROBLEM -7 Statement: Design a double-dwell cam to move a follower from to 25 6, dwell for 12, fall 25 and dwell for the remader The total cycle must take 4 sec
DESIGN OF MACHINERY SOLUTION MANUAL -7-1! PROBLEM -7 Statement: Design a double-dwell cam to move a follower from to 25 6, dwell for 12, fall 25 and dwell for the remader The total cycle must take 4 sec
Lecture 2. Soundness and completeness of propositional logic
 Lecture 2 Soundness and completeness of propositional logic February 9, 2004 1 Overview Review of natural deduction. Soundness and completeness. Semantics of propositional formulas. Soundness proof. Completeness
Lecture 2 Soundness and completeness of propositional logic February 9, 2004 1 Overview Review of natural deduction. Soundness and completeness. Semantics of propositional formulas. Soundness proof. Completeness
the total number of electrons passing through the lamp.
 1. A 12 V 36 W lamp is lit to normal brightness using a 12 V car battery of negligible internal resistance. The lamp is switched on for one hour (3600 s). For the time of 1 hour, calculate (i) the energy
1. A 12 V 36 W lamp is lit to normal brightness using a 12 V car battery of negligible internal resistance. The lamp is switched on for one hour (3600 s). For the time of 1 hour, calculate (i) the energy
Finite Field Problems: Solutions
 Finite Field Problems: Solutions 1. Let f = x 2 +1 Z 11 [x] and let F = Z 11 [x]/(f), a field. Let Solution: F =11 2 = 121, so F = 121 1 = 120. The possible orders are the divisors of 120. Solution: The
Finite Field Problems: Solutions 1. Let f = x 2 +1 Z 11 [x] and let F = Z 11 [x]/(f), a field. Let Solution: F =11 2 = 121, so F = 121 1 = 120. The possible orders are the divisors of 120. Solution: The
NATIONAL AND KAPODISTRIAN UNIVERSITY OF ATHENS SCHOOL OF SCIENCE FACULTY OF INFORMATICS AND TELECOMMUNICATIONS
 NATIONAL AND KAPODISTRIAN UNIVERSITY OF ATHENS SCHOOL OF SCIENCE FACULTY OF INFORMATICS AND TELECOMMUNICATIONS INTERDICIPLINARY POSTGRADUATE PROGRAMME "INFORMATION TECHNOLOGIES IN MEDICINE AND BIOLOGY"
NATIONAL AND KAPODISTRIAN UNIVERSITY OF ATHENS SCHOOL OF SCIENCE FACULTY OF INFORMATICS AND TELECOMMUNICATIONS INTERDICIPLINARY POSTGRADUATE PROGRAMME "INFORMATION TECHNOLOGIES IN MEDICINE AND BIOLOGY"
Supplemental Table S1. Tumor specific networks are enriched with somatically mutated genes (taken from the database COSMIC)
 Additional file 1 Supplemental Table S1. Tumor specific networks are enriched with somatically mutated genes (taken from the database COSMIC) COSMIC genes in tumor network COSMIC genes not in tumor network
Additional file 1 Supplemental Table S1. Tumor specific networks are enriched with somatically mutated genes (taken from the database COSMIC) COSMIC genes in tumor network COSMIC genes not in tumor network
* To whom correspondence should be addressed.
 SUPPORTING INFORMATION Computational design of a pincer phosphinito vanadium ((OPO)V) propane monoxygenation homogeneous catalyst based on the reduction-coupled oxo activation (ROA) mechanism Ross Fu,
SUPPORTING INFORMATION Computational design of a pincer phosphinito vanadium ((OPO)V) propane monoxygenation homogeneous catalyst based on the reduction-coupled oxo activation (ROA) mechanism Ross Fu,
ΠΡΟΣΚΛΗΣΗ ΕΚΔΗΛΩΣΗΣ ΕΝΔΙΑΦΕΡΟΝΤΟΣ
 1 ΕΛΛΗΝΙΚΗ ΔΗΜΟΚΡΑΤΙΑ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΡΗΤΗΣ ΕΙΔΙΚΟΣ ΛΟΓΑΡΙΑΣΜΟΣ ΑΝΑΡΤΗΤΕΑ ΣΤΟ ΔΙΑΔΙΚΤΥΟ ΠΡΟΣΚΛΗΣΗ ΕΚΔΗΛΩΣΗΣ ΕΝΔΙΑΦΕΡΟΝΤΟΣ Ηράκλειο, 04.02.2014 Αρ. πρωτ. 1054 Ο Ειδικός Λογαριασμός του Πανεπιστημίου Κρήτης
1 ΕΛΛΗΝΙΚΗ ΔΗΜΟΚΡΑΤΙΑ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΡΗΤΗΣ ΕΙΔΙΚΟΣ ΛΟΓΑΡΙΑΣΜΟΣ ΑΝΑΡΤΗΤΕΑ ΣΤΟ ΔΙΑΔΙΚΤΥΟ ΠΡΟΣΚΛΗΣΗ ΕΚΔΗΛΩΣΗΣ ΕΝΔΙΑΦΕΡΟΝΤΟΣ Ηράκλειο, 04.02.2014 Αρ. πρωτ. 1054 Ο Ειδικός Λογαριασμός του Πανεπιστημίου Κρήτης
Test Data Management in Practice
 Problems, Concepts, and the Swisscom Test Data Organizer Do you have issues with your legal and compliance department because test environments contain sensitive data outsourcing partners must not see?
Problems, Concepts, and the Swisscom Test Data Organizer Do you have issues with your legal and compliance department because test environments contain sensitive data outsourcing partners must not see?
SCITECH Volume 13, Issue 2 RESEARCH ORGANISATION Published online: March 29, 2018
 Journal of rogressive Research in Mathematics(JRM) ISSN: 2395-028 SCITECH Volume 3, Issue 2 RESEARCH ORGANISATION ublished online: March 29, 208 Journal of rogressive Research in Mathematics www.scitecresearch.com/journals
Journal of rogressive Research in Mathematics(JRM) ISSN: 2395-028 SCITECH Volume 3, Issue 2 RESEARCH ORGANISATION ublished online: March 29, 208 Journal of rogressive Research in Mathematics www.scitecresearch.com/journals
Homework 3 Solutions
 Homework 3 Solutions Igor Yanovsky (Math 151A TA) Problem 1: Compute the absolute error and relative error in approximations of p by p. (Use calculator!) a) p π, p 22/7; b) p π, p 3.141. Solution: For
Homework 3 Solutions Igor Yanovsky (Math 151A TA) Problem 1: Compute the absolute error and relative error in approximations of p by p. (Use calculator!) a) p π, p 22/7; b) p π, p 3.141. Solution: For
ΑΝΙΧΝΕΥΣΗ ΓΕΓΟΝΟΤΩΝ ΒΗΜΑΤΙΣΜΟΥ ΜΕ ΧΡΗΣΗ ΕΠΙΤΑΧΥΝΣΙΟΜΕΤΡΩΝ ΔΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ
 ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΣΧΟΛΗ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΜΗΧΑΝΙΚΩΝ ΥΠΟΛΟΓΙΣΤΩΝ ΤΟΜΕΑΣ ΕΠΙΚΟΙΝΩΝΙΩΝ ΗΛΕΚΤΡΟΝΙΚΗΣ ΚΑΙ ΣΥΣΤΗΜΑΤΩΝ ΠΛΗΡΟΦΟΡΙΚΗΣ ΑΝΙΧΝΕΥΣΗ ΓΕΓΟΝΟΤΩΝ ΒΗΜΑΤΙΣΜΟΥ ΜΕ ΧΡΗΣΗ ΕΠΙΤΑΧΥΝΣΙΟΜΕΤΡΩΝ
ΕΘΝΙΚΟ ΜΕΤΣΟΒΙΟ ΠΟΛΥΤΕΧΝΕΙΟ ΣΧΟΛΗ ΗΛΕΚΤΡΟΛΟΓΩΝ ΜΗΧΑΝΙΚΩΝ ΚΑΙ ΜΗΧΑΝΙΚΩΝ ΥΠΟΛΟΓΙΣΤΩΝ ΤΟΜΕΑΣ ΕΠΙΚΟΙΝΩΝΙΩΝ ΗΛΕΚΤΡΟΝΙΚΗΣ ΚΑΙ ΣΥΣΤΗΜΑΤΩΝ ΠΛΗΡΟΦΟΡΙΚΗΣ ΑΝΙΧΝΕΥΣΗ ΓΕΓΟΝΟΤΩΝ ΒΗΜΑΤΙΣΜΟΥ ΜΕ ΧΡΗΣΗ ΕΠΙΤΑΧΥΝΣΙΟΜΕΤΡΩΝ
Approximation of distance between locations on earth given by latitude and longitude
 Approximation of distance between locations on earth given by latitude and longitude Jan Behrens 2012-12-31 In this paper we shall provide a method to approximate distances between two points on earth
Approximation of distance between locations on earth given by latitude and longitude Jan Behrens 2012-12-31 In this paper we shall provide a method to approximate distances between two points on earth
Πτυχιακή Εργασία Η ΠΟΙΟΤΗΤΑ ΖΩΗΣ ΤΩΝ ΑΣΘΕΝΩΝ ΜΕ ΣΤΗΘΑΓΧΗ
 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ ΥΓΕΙΑΣ Πτυχιακή Εργασία Η ΠΟΙΟΤΗΤΑ ΖΩΗΣ ΤΩΝ ΑΣΘΕΝΩΝ ΜΕ ΣΤΗΘΑΓΧΗ Νικόλας Χριστοδούλου Λευκωσία, 2012 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ ΥΓΕΙΑΣ
ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ ΥΓΕΙΑΣ Πτυχιακή Εργασία Η ΠΟΙΟΤΗΤΑ ΖΩΗΣ ΤΩΝ ΑΣΘΕΝΩΝ ΜΕ ΣΤΗΘΑΓΧΗ Νικόλας Χριστοδούλου Λευκωσία, 2012 ΤΕΧΝΟΛΟΓΙΚΟ ΠΑΝΕΠΙΣΤΗΜΙΟ ΚΥΠΡΟΥ ΣΧΟΛΗ ΕΠΙΣΤΗΜΩΝ ΥΓΕΙΑΣ
ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΕΙΡΑΙΑ ΤΜΗΜΑ ΝΑΥΤΙΛΙΑΚΩΝ ΣΠΟΥΔΩΝ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΗΝ ΝΑΥΤΙΛΙΑ
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΕΙΡΑΙΑ ΤΜΗΜΑ ΝΑΥΤΙΛΙΑΚΩΝ ΣΠΟΥΔΩΝ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΗΝ ΝΑΥΤΙΛΙΑ ΝΟΜΙΚΟ ΚΑΙ ΘΕΣΜΙΚΟ ΦΟΡΟΛΟΓΙΚΟ ΠΛΑΙΣΙΟ ΚΤΗΣΗΣ ΚΑΙ ΕΚΜΕΤΑΛΛΕΥΣΗΣ ΠΛΟΙΟΥ ΔΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ που υποβλήθηκε στο
ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΕΙΡΑΙΑ ΤΜΗΜΑ ΝΑΥΤΙΛΙΑΚΩΝ ΣΠΟΥΔΩΝ ΠΡΟΓΡΑΜΜΑ ΜΕΤΑΠΤΥΧΙΑΚΩΝ ΣΠΟΥΔΩΝ ΣΤΗΝ ΝΑΥΤΙΛΙΑ ΝΟΜΙΚΟ ΚΑΙ ΘΕΣΜΙΚΟ ΦΟΡΟΛΟΓΙΚΟ ΠΛΑΙΣΙΟ ΚΤΗΣΗΣ ΚΑΙ ΕΚΜΕΤΑΛΛΕΥΣΗΣ ΠΛΟΙΟΥ ΔΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ που υποβλήθηκε στο
New bounds for spherical two-distance sets and equiangular lines
 New bounds for spherical two-distance sets and equiangular lines Michigan State University Oct 8-31, 016 Anhui University Definition If X = {x 1, x,, x N } S n 1 (unit sphere in R n ) and x i, x j = a
New bounds for spherical two-distance sets and equiangular lines Michigan State University Oct 8-31, 016 Anhui University Definition If X = {x 1, x,, x N } S n 1 (unit sphere in R n ) and x i, x j = a
The ε-pseudospectrum of a Matrix
 The ε-pseudospectrum of a Matrix Feb 16, 2015 () The ε-pseudospectrum of a Matrix Feb 16, 2015 1 / 18 1 Preliminaries 2 Definitions 3 Basic Properties 4 Computation of Pseudospectrum of 2 2 5 Problems
The ε-pseudospectrum of a Matrix Feb 16, 2015 () The ε-pseudospectrum of a Matrix Feb 16, 2015 1 / 18 1 Preliminaries 2 Definitions 3 Basic Properties 4 Computation of Pseudospectrum of 2 2 5 Problems
